Abstract
Breast cancer (BC) is the 2nd most common cause of cancer-related deaths. Antibody-drug conjugates (ADCs) are monoclonal antibodies linked to cytotoxic agents and are directed towards a specific tumor protein. Therefore, they are more potent and can have relatively less toxicity. In this meta-analysis, we assessed the efficacy and safety of ADCs in breast cancer. We searched PubMed, Cochrane, Web of Science, and clinicaltrials.gov for relevant studies and included 7 randomized clinical trials (N = 5,302) and 7 non-randomized clinical trials (N = 658). R programming language software was used to conduct this meta-analysis. In 4 RCTs on HER-2 positive BC (N = 2,825), the pooled HR of PFS and OS was 0.72 (95% CI = 0.61–0.84, I2 = 71%) and 0.73 (95% CI = 0.64–0.84, I2 = 20%), respectively in favor of ADCs versus chemotherapy. In RCT on triple negative BC (N = 468), HR of PFS and OS were 0.55 (95%CI = 0.51–0.61) and 0.59 (95% CI = 0.54–0.66), respectively, in favor of saci-gov versus chemotherapy. In RCT on HER-2 positive residual invasive BC, HR of recurrence/death was 0.61 (95% CI = 0.54–0.69) in favor of ADC versus chemotherapy. In an RCT (N = 524), the HR of PFS and OS were 0.28 (95% CI = 0.22–0.37) and 0.55 (95%CI = 0.36–0.86), respectively, in favor of trastuzumab-deruxtecan (T-der) as compared to trastuzumab-emtansine (T-DM1). Anemia, rash, diarrhea, fatigue, hypertension, thrombocytopenia, and elevated aminotransferases were the common ≥grade 3 adverse events reported in 4%, 1%, 2%, 1%, 2%, 9%, and 3% of the patients, respectively. ADCs were more effective than single and double agent chemotherapy in patients with HER-2 positive or triple negative BC. Among ADCs, T-der was more effective than T-DM1.
Keywords
Introduction
The second leading cause of most cancer-related deaths in women is breast cancer. It remains the most common non-cutaneous invasive cancer in women [1]. Molecular profiling shows that approximately 75% of all breast cancers exhibit the high activity of estrogen receptor (ER+) [2], 15–20% of breast cancer patients, overexpress the human epidermal growth factor receptor-2 (HER-2) [3], and another 15–20% constitute triple-negative breast cancer (TNBC) [4]. TNBC lacks estrogen and progesterone receptor expression and manifests the usual number of gene copies and expressions of the HER-2 receptor gene [5].
Neo-adjuvant chemotherapy has become the mainstay of treatment in TNBC and HER2-positive early breast cancer [6]. The therapeutic cornerstones include chemotherapy and anti-HER2 targeting for HER-2-positive disease; endocrine therapy for hormone receptor-positive disease; poly adenosine diphosphate ribose polymerase inhibitors and bone stabilizing medications for breast cancer gene (BRCA) mutation carriers; and, quite recently, immunotherapy [7]. However, in metastatic breast cancer (mBC), therapy aims to prolong survival and maintain quality of life [6]. Nevertheless, systemic treatment has some limitations due to poor selectivity of the target, these drugs often cause dose-limiting toxicity in addition to resistance to the anti-cancer agents. Therefore, more precisely targeted drug therapies can improve efficacy and reduce toxicity [8].
Antibody-drug conjugates (ADCs) is considered one of the most rewarding targeted drug strategy. Multiple ADCs have been approved for cancer by the United States Food and Drug Administration (FDA) and multiple ADCs including trastuzumab emtansine (T-DM1) and trastuzumab deruxtecan (T-der), sacituzumab govitecan-hziy (Saci-gov), trastuzumab duocarmazine (T-duo), BAT8001, ALT-P7, PF-06804103, patritumab deruxtecan, ladiratuzumab vedotin, glembatumumab vedotin, etc. are tested in breast cancer patients [8]. ADCs are monoclonal antibodies conjugated to the cytotoxic payload via a chemical linker, directed towards cell-surface proteins specific to tumor and, therefore, are tumor-specific and potent and less toxic, interests not achievable with conventional drugs [9]. Prerequisites for effective and safe ADCs development for cancer therapy are selecting a suitable target molecule, a monoclonal antibody directed to the target, a potent cytotoxic effector molecule, and the monoclonal antibody conjugation to the cytotoxic agent [9,10] (Fig. 1).
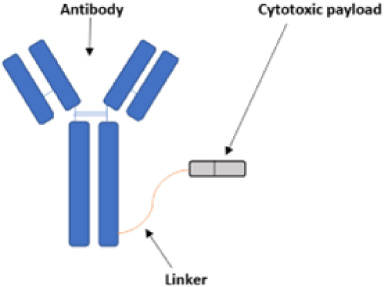
Structure of a typical Antibody-drug conjugate (ADC).
Since taxanes were introduced as adjuvant therapy, comparatively few therapeutic advancements have happened in treating TNBC. However, enormous research is now occurring in this area [5]. This review aims to sum up the efficacy and safety of different antibody-drug conjugates in patients with triple-negative and HER-2 positive breast cancer.
This systematic review and meta-analysis was performed according to the pre-specified guidelines registered on Prospero (CRD42022329529). Cochrane [11] and PRISMA [12] guidelines were followed to establish these prespecified guidelines.
Search strategy
Databases included for literature search were Embase, PubMed (Medline), Web of Science (WoS), and Cochrane, and registry of clinicaltrials.gov. MeSH and Emtree terms “breast neoplasms” and “immunoconjugates” used. The literature search was performed from the inception of literature till 04/25/2022 by following the PICO framework (Supplementary Table S1) [13].
Inclusion and exclusion criteria
Only clinical trials providing safety and efficacy data were included, i-e, clinical response, survival rates, and adverse events. All preclinical studies, case reports, retrospective studies, review articles, meta-analyses, and clinical trials without breast cancer, ADC use or prespecified efficacy and safety outcomes were excluded.
Study selection
Articles were reviewed by three researchers and selected them based on pre-specified inclusion and exclusion criteria. The differences were addressed by a fourth researcher.
Data extraction
Data was extracted for the baseline characteristics of the study (treatment drug, patient population, metastatic location, age of patients, and prior therapy), efficacy measures (overall response rate (ORR), complete response (CR), progression-free survival (PFS), overall survival (OS), hazard ratios (HRs) of survival rates and toxicity (≥ grade 3 adverse effects) by three authors.
Primary outcome of meta-analysis was hazard ratio (HR) of PFS. OS and ORR were the secondary outcomes for efficacy and pooled incidence of ≥ grade 3 adverse effects were measured as outcomes of safety.
Risk of bias assessment
The risk of bias (ROB) was assessed by using Rob-2 tool [14]. Two researchers (XX and XX) assessed the ROB independently and the differences were addressed by the third researcher (XX).
Statistical analysis
We used R programming language software for meta-analysis in this study. “Meta” package in R was used [15]. Random-effects model for meta-analysis to keep our results consistent and applicable broadly. Dersimonian–Laird estimator was used to calculate between-study variance. The Mantel–Haenszel method was used to pool the risk ratios. A continuity correction of 0.5 was used for studies with zero events in any of the arms. To estimate the heterogeneity, I2 was used. 95% confidence interval was used in these calculations.
Results
3,936 articles were identified from PubMed (1,560), Embase (472), WOS (1,782), and clinicaltrials.gov registry (122). Seven randomized clinical trials (RCTs, N = 5,302) [16–22] and seven non-randomized studies (nRCTs, N = 658) [23–29] were included (Fig. 2).
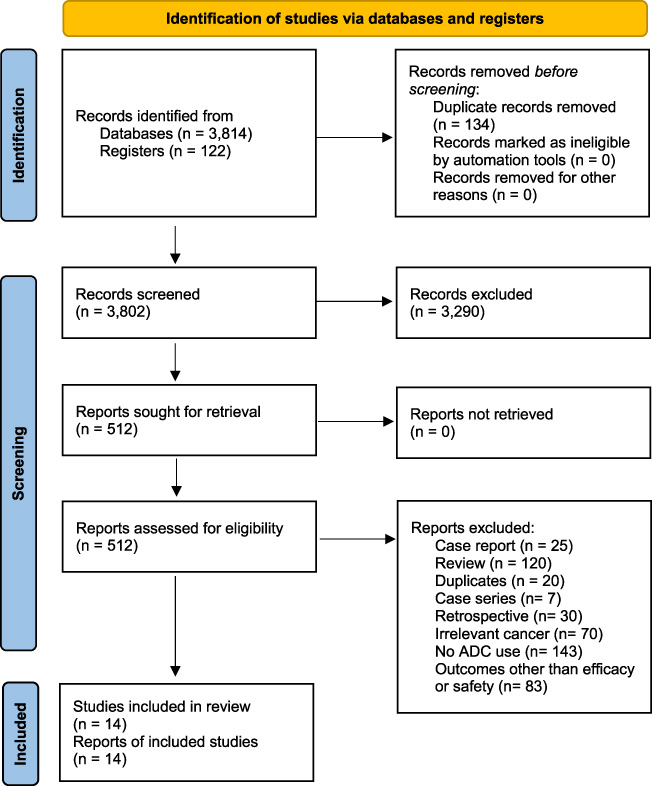
Prisma flow sheet of selection of articles.
The risk of bias was high in all included randomized studies due to lack of blinding. Risk of bias was low due to missing outcome data, selection of reported outcome results, randomization process, or measurement of outcome in RCTs. Risk of bias was high in nRCTs due to lack of randomization and blinding (Fig. 3).
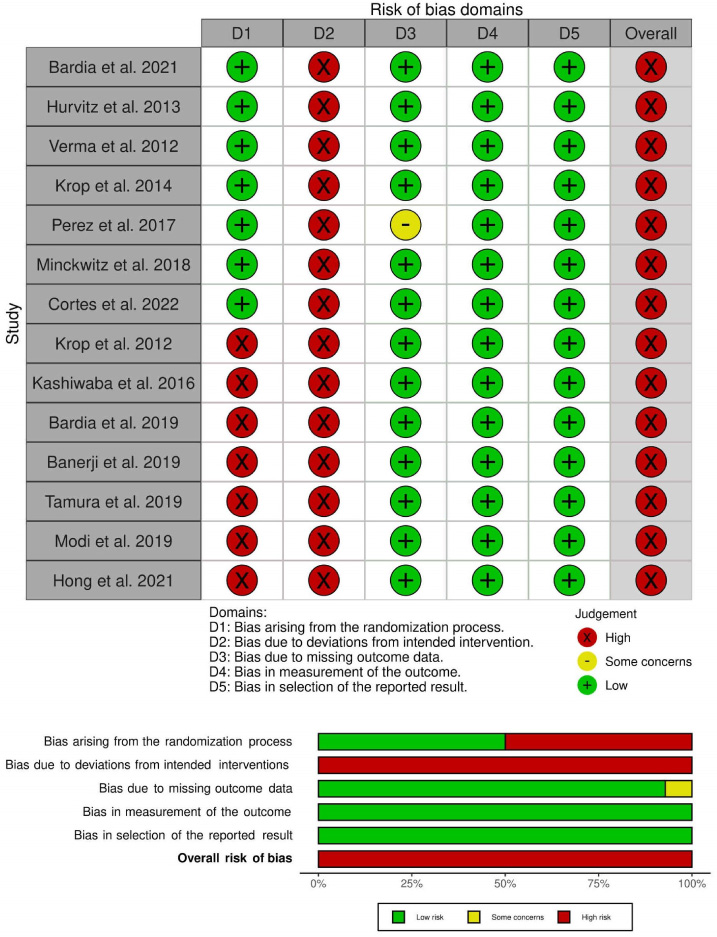
Risk of bias in included clinical trials.
Efficacy of ADCs versus chemotherapy
6 RCTs including 4,779 patients with breast cancer were conducted to assess the efficacy of ADC versus chemotherapy. 468 patients had triple-negative breast cancer (TNBC) while 4,311 patients had HER-2 positive breast cancer. 3,293 patients had advanced metastatic disease while 1,486 patients had residual invasive disease. 2,311 patients were treated with ADC (235 with sacituzumab govitecan (saci-gov) and 2,076 with T-DM1), 431 patients were treated with single agent chemotherapy, 743 patients were treated with single agent tastuzumab, and 931 patients were treated with two drug chemotherapy regimens (Table 1).
Baseline characteristics of patients included in clinical trials
Baseline characteristics of patients included in clinical trials
In 4 RCTs on HER-2 positive advanced breast cancer patients (N = 2,825), the pooled HR of PFS and OS was 0.72 (95% CI = 0.61–0.84, I2 = 71%) and 0.73 (95% CI = 0.64–0.84, I2 = 20%), respectively, in favor of ADC as compared to chemotherapy (Fig. 3A and B). Risk of ORR was 1.48 (95% CI = 0.84–2.60, I2 = 91%) in favor of ADC (Fig. 3C). In RCT on triple negative breast cancer (N = 468), HR of PFS and OS were 0.55 (95% CI = 0.51–0.61) and 0.59 (95% CI = 0.54–0.66), respectively, in favor of saci-gov (ADC) as compared to chemotherapy. In RCT on HER-2 positive residual invasive breast cancer, HR of recurrence/death was 0.61 (95% CI = 0.54–0.69) in favor of T-DM1 (Fig. 4 A, B, C).
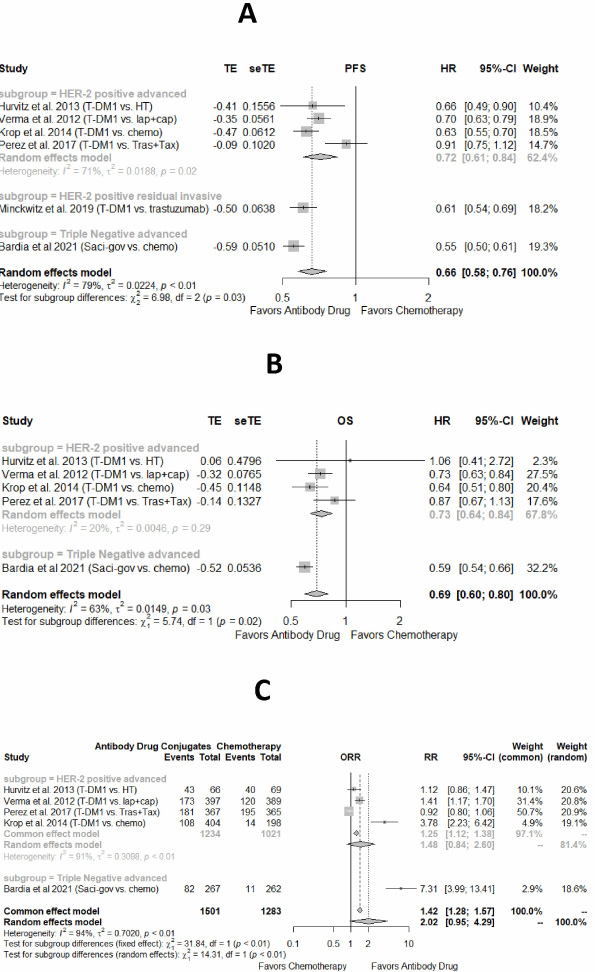
Efficacy of antibody drug conjugates as compared to chemotherapy in randomized clinical trials. (A) Hazard ratio of progression free survival, (B) Hazard ratio of overall survival, (C) relative risk of overall response rate.
To address heterogeneity, sensitivity analysis and subgroup analysis were performed. Perez et al. 2017 was detected as an outlier. Further subgroups were made based on the number of drugs in chemotherapy group. In the subgroup of ADC versus one drug chemotherapy regimen, pooled HR of PFS and OS were 0.59 (95% CI = 0.55–0.64, I2 = 22%) and 0.60 (95% CI = 0.55–0.66, I2 = 0%), respectively, in favor of ADC. In the subgroup of ADC versus two drug chemotherapy regimen, pooled HR of PFS and OS were 0.73 (95% CI = 0.63–0.85, I2 = 0%) and 0.70 (95% CI = 0.63–0.78, I2=0%), respectively, in favor of ADC. (Supplementary Figs S1--S3)
In an RCT by Cortes et al. (N = 524), 261 HER-2 positive mBC patients were treated with T-der, and 263 patients were treated with T-DM1 (Table 1). The HR of PFS and OS were 0.28 (95% CI = 0.22–0.37) and 0.55 (95% CI = 0.36–0.86), respectively, in favor of T-der as compared to T-DM1. The RR of ORR and CR were 2.33 and 1.28, respectively, in favor of T-der as compared to T-DM1 (Supplementary Figs S4–S5).
Non-randomized clinical trials
Efficacy of newer ADCs in trial single arm comparison
In a clinical trial by Banerja et al., 39 advanced HER-2 patients were treated with trastuzumab duocarmazine (T-duo). CR, ORR, and mPFS were 0%, 33% (95% CI = 20%–48%), and 7.6 months (95% CI = 4.2–10.9), respectively. In a clinical trial by Hong et al., 29 advanced HER-2 patients were treated with BAT8001. CR, ORR and mPFS were 3% (95% CI = 0–0.18), 41% (95% CI = 0.24–0.61) and 4.3 months (95% CI = 2.51–6.09), respectively. Pooled incidence of ORR in pretreated/refractory breast cancer patients treated with T-DM1 (N = 1188), T-der (N = 556), and saci-gov (N = 375) were 36% (95% CI = 32%–41%), 68% (95% CI = 56%–78%), and 31% (95% CI = 27%–36%), respectively. (Supplemental Figs S6--S7)
≥Grade 3 adverse events of ADCs
In pooled analysis of 3 RCTs (N = 1,827), RR of ≥grade 3 adverse events was 0.69 (95% CI = 0.51–0.94, I2 = 85%), in favor of ADC as compared to two drug chemotherapy regimen. In pooled analysis of 3 RCTs (N = 2,529), RR of ≥grade 3 adverse events was 1.2 (95% CI = 0.74–1.92, I2 = 93%), not significantly different among ADC versus single agent chemotherapy (Supplementary Fig. S8).
Pooled incidence of ≥ grade 3 adverse events were 43% (95% CI = 38%–49%, I2 = 79%), 75% (95% CI = 46%–91%, I2 = 94%), 35% (95% CI = 27%–54%, I2 = 0), 49% (95% CI = 44%-54%, I2=20%), and 48% (95% CI = 29%-67%, I2 = 0) of the patients treated with T-DM1, saci-gov, T-duo, T-der, and BAT8001, respectively. Anemia, rash, diarrhea, fatigue, hypertension, thrombocytopenia, and elevated aminotransferases were the common ≥grade 3 adverse events reported in patients treated with ADC. The incidence of each adverse event is given in (Table 2, supplemental Figs S9-S17).
Incidence of ≥Grade 3 adverse events in patients treated with ADC
Incidence of ≥Grade 3 adverse events in patients treated with ADC
Interim results of two trials presented in conferences [30,31](N = 62) showed that ALT-P7 and PF-06804103 were safe and effective in HER-positive breast cancer. Five phase III randomized clinical trials (N = 3,643) are in progress to assess the efficacy and safety of T-der, T-duo, and sac-gov. 14 early phase trials (N = 1,769) are in progress to assess the efficacy and tolerability of novel immune-drug conjugates and their combinations: patritumab deruxtecan (U3 1402), DS-8201a, ARX788, sacituzumab govitecan+talazoparib, alpelisib + Sacituzumab deruxtecan, nivolumab + trastuzumab-deruxtecan, pembrolizumab + trastuzumab-deruxtecan, MRG002, and ladiratuzumab vedotin (Table 3).
Ongoing clinical trials registered on clinicaltrials.gov
Ongoing clinical trials registered on clinicaltrials.gov
Multiple ADCs are being studied to assess their efficacy and safety in the treatment of breast cancer in combination with or after failure of other therapies. However, only a handful of ADCs were found to be effective in RCTs in breast cancer treatment including, T-DM1, T-der and Saci-gov.
T-DM1 combines HER2–targeted delivery of the potent maytansinoid antimicrotubule agent DM1 with the antitumor activity of trastuzumab and uses a very stable thioether linker which plays a role in its favorable safety profile [32].
In the first in human phase I nRCT (N = 24) done by Krop et al. (2010) [33], T-DM1 was used thrice weekly in patients with HER2-positive mBC with a median of four prior chemotherapeutic agents for metastatic disease. Maximum tested dose of 3.6 mg/kg was well-tolerated in 4 patients for a year with minimal adverse effects, reversible thrombocytopenia being the most common. In the phase II study by Krop et al. (2012) [23] on HER 2-positive mBC patients, (N =110) patients were previously treated with all standard HER2-directed therapies and chemotherapies. The results confirmed the effectiveness and tolerability of T-DM1 with a clinical benefit in terms of ORR, prolonged PFS of 6.9 months and duration of response of 7.2 months. The safety profile of T-DM1 in this study was encouraging. The only ≥ grade 3 AE reported in >5% of patients were thrombocytopenia.
In 2013, T-DM1 was approved as second-line therapy for HER2-positive mBC after the RCT EMILIA trial by Verma et al. (N = 991) [18]. The study supported the therapeutic potential of T-DM1 (N = 495) as compared with lapatinib plus capecitabine (N = 496). T-DM1 showed significantly improved PFS by 3.2 m (HR 0.65, 95% CI = 0.55–0.77), ORR (43.6% versus 30.8%); and OS by 4 months (HR 0.75, 95% CI = 0.64–0.88) across a heterogeneous population of patients with HER-2-positive mBC that had progressed during or after treatment with trastuzumab and a taxane.
In 2013, Hurvitz et al. [17] conducted the first Phase II RCT of T-DM as first line treatment versus trastuzumab + docetaxel in patients with HER-2+ mBC. T-DM1 provided a clinically meaningful and statistically significant (41%) reduction in the relative risk of disease progression versus first line HT therapy as well as superior PFS (5 month longer), safety, and quality of life. Notably, grade 4 AEs and treatment discontinuation due to toxicity were significantly lower with T-DM1 compared to HT (grade 4 AEs = 5.8% versus 57.6%, discontinuation = 7.2% versus 40.9%). The TH3RESA Phase 3 study by Krop et al. (2014) [22] assessed the efficacy and safety of T-DM1 versus treatment of physician’s choice in patients with progressive HER2-positive breast cancer after trastuzumab-based and lapatinib-based therapy for advanced disease and previous treatment with a taxane. The results of TH3RESA study were consistent with EMILIA study and showed that T-DM1 was associated with a significant improvement in OS, PFS, and safety in such patients. Therefore, T-DM1 showed better results than chemotherapy drugs.
The study evaluating the feasibility and cardiac safety of T-DM1 in patients with early TNBC (N = 148) was done by Krop et al in 2015 [34] and concluded that after anthracycline-based chemotherapy, approximately 1 year of (neo)adjuvant T-DM1 was feasible and well tolerated by most of the patients. 17.6% of the patients were not able to complete the T-DM1 treatment. On the other hand, in the joint analysis of the National Surgical Adjuvant Breast and Bowel Project (NSABP B-31) trial and North Central Cancer Treatment Group (NCCTG N9831) trial, 31.4% of the patients discontinued trastuzumab before completing 1 year of therapy. The overall rate of cardiac events in the heterogeneous patient population was low. 2.7% patients had asymptomatic decline in LVEF, 2 patients had serious atrial fibrillation, and T-DM1 was discontinued in one patient with drug related cardiac complications.
Kashiwaba et al. (2016) [24] explored the efficacy, safety, and pharmacokinetics of T-DM1 in a Phase II study in heavily pretreated Japanese patients with HER2-positive mBC. The efficacy results, safety profile and the pharmacokinetics from this multicenter study were also consistent with the findings from the EMILIA and TH3RESA Phase III trials.
In the phase 3 RCT (KATHERINE) by Von Minckwitz et al. (2019) [20], it was concluded that the risk of recurrence of invasive breast cancer or death was 50% lower with adjuvant T-DM1 than with trastuzumab alone. However, recurrence in the central nervous system remained a persistent problem. Reversible peripheral sensory neuropathy and thrombocytopenia were more with T-DM1 than with trastuzumab alone while cardiac events were fewer in the T-DM1 group. Some factors limiting clinical activity of T-DM1 include cross resistance of the DM-1 moiety (targeting the microtubules) in taxane pretreated patients. T-DM1 was approved as post-neoadjuvant therapy for patients who were refractory to trastuzumab and taxane-based therapy and is a useful option for this patient population. However, double blinded long term follow- up studies on T-DM1 and T-DM1 combinations would provide more useful information.
The phase 2 study (DESTINY-Breast01) on T-der by Modi et al. (2020) [35] showed durable antitumor activity in patients with HER2-positive mBC who were treated with T-DM1. T-der retained efficacy in patients with low HER2 expression as well. Gastrointestinal and hematologic toxic effects were reported. The most concerning adverse effect was interstitial lung disease (in 13.6% of the patients) requiring careful monitoring and even death in some patients. It was approved by FDA and EMA as third line agent for HER-2 positive mBC.
Recently, the efficacy and safety of T-der was compared to T-DM1 in the study by Cortes et al. (2022) [21] in second line setting in DESTINY-Breast03 phase 3 RCT in patients with HER2-positive mBC previously treated with trastuzumab and a taxane. The data showed significantly superior reduction in the risk of progression or death with T-der as compared to T-DM1. The estimated median PFS of 6.8 months with T-DM1 was lower than that reported in the EMILIA trial which may result from differences in prior pertuzumab use in this trial. It is notable that, median PFS with T-DM1 was considerably longer among patients who had received no or one line of previous therapy than those who had received two or more (8.0 and 5.6 months, respectively). Almost twice as many patients in the T-der group demonstrated a CR compared to the T-DM1 group (16.1% versus 8.7%). The incidence of adverse events after the start of treatment was similar both groups (99.6% and 95.4%, respectively). T-der was associated with increased risk of pneumonitis and interstitial lung disease. However, incidence of interstitial lung disease and pneumonitis associated with T-der was lower in this trial than the previous ones. Despite causing pneumonitis, T-der caused significant improvement in efficacy therefore should be considered in the treatment of breast cancer. However, long term follow ups and large scale double blinded clinical trials are needed to further investigate the side effects.
Sacituzumab govitecan-hziy (saci-gov) is an antibody–drug conjugate with Trop-2 as the target of recognition. TNBC has a high expression of Trop-2 and is associated with a poor prognosis [36]. Bardia et al in 2019 [25] studied saci-gov (IMMU-132) in patients with mTNBC who were pretreated with at least two previous therapies for metastatic disease and the drug was well tolerated by most of the patients with significant improvement in median survival rates. Discontinuation rate was low. The randomized phase III study ASCENT by Bardia et al. (N = 529) [16], compared saci-gov to selected mono-chemotherapies in patients with at least 2 prior chemotherapies for metastatic disease. The trial was stopped early due to sufficient evidence of efficacy. Saci-gov reduced the risk of disease progression or death and significantly prolonged the OS. The efficacy of saci-gov was independent of BRCA status. The study also enrolled patients only 1 prior chemotherapy if they progressed rapidly after (neo)adjuvant therapy and these patients derived the same benefit compared to the overall population.
In the phase I study by Hong et al 2021 [29], BAT8001 (a novel ADC targeting HER2) was well tolerated at a dose of 3.6 mg/kg. BAT8001 showed promising efficacy and therapeutic potential in heavily pretreated patients with HER2- positive mBC. ORR and disease control was reported in 41.4% and 82.8% of the patients, respectively. One patient achieved CR at the MTD cohort. Reported adverse effects included thrombocytopenia and elevated liver transaminase that was reported as a dose-limiting toxicity in the 6.0 mg/kg cohort.
T-duo is an ADC with a DNA-alkylating duocarmycin payload. In 2019, a phase 1 study on T-duo in heavily pretreated patients with locally advanced or metastatic solid tumors showed important and relevant clinical activity and a manageable safety profile with a few grade 3 or 4 adverse events [26]. Ocular toxicity was the most common side effect reported in 71% of the patients (7% garde 3). T-duo showed meaningful single-agent clinical activity not only in HER2-positive, and HER2-low (IHC 1+ or IHC 2+ ISH-negative) hormone receptor-negative mBC, but also in non-breast HER2-expressing metastatic cancers, which have few treatment options and poor prognoses (e.g., urothelial and endometrial cancers). The TULIP randomized phase 3 study (NCT03262935) comparing T-duo with standard-of-care chemotherapy combinations in patients with HER2-positive breast cancer is ongoing.
Bendell et al. in 2014 [37] conducted a Phase I/II study of the antibody-drug conjugate glembatumumab vedotin in patients with locally advanced or mBC. Results of this trial suggested that glembatumumab vedotin had an acceptable and manageable safety profile with limited anti-tumor activity but can be more in patient with gpNMB tumor expression. EMERGE and METRIC trials were conducted to assess the efficacy and safety of glembatumumab vedotin in RCTs in patients with advanced, gpNMB-expressing, pretreated breast cancer. However, the trials didn’t show significant improvement in disease progression.
The first-in-human study of PF-06647263, a novel ADC consisting of the anti-EFNA4 mAb PF-06523432 conjugated to a calicheamicin payload was conducted in 2019 by Garrido-Laguna et al. [38]. The study enrollment was terminated due to the limited response to adequate exposure of PF-06647263 in patients with TNBC.
Conclusion
ADC were well tolerated by most of the patients. T-DM1 was significantly more effective than chemotherapy in patients with relapsed/refractory or treatment naïve HER-2 positive breast cancer. T-DM1 was significantly more effective than trastuzumab in HER-2 +ve residual invasive disease after neoadjuvant therapy. Saci-gov was significantly more effective than single agent chemotherapy in patients with refractory mTNBC. T-der was significantly more efficacy than T-DM1 in patients with relapsed/refractory advanced breast cancer. Among novel ADCs, trastuzumab-duocarmazine, MM302, and BAT8001 have shown promising efficacy results in early phase trials. More randomized double blind clinical trials are needed to confirm these results.
Limitations
The risk of bias was high in the included studies due to lack of blinding in the included studies. Novel agents were only tested in There was limited data available on combination regimens of ADCs. Long term follow up studies for adverse effects and long-term improvement in survival rates were not reported in the included studies. Despite these limitations, the article was able to provide an overview of efficacy and safety of ADCs in breast cancer.
Footnotes
Acknowledgements
None.
Authorship statement
Wajeeha Aiman, Muhammad Ashar Ali, and Fatima Afzal designed the study. Hafsa Zahoor, Ayifa Riaz Bajwa, Syeda Hafsa Kazmi, Aqsa Anwar, Sana Rashid did the database screening. Hina Zubair, Tooba Kashif, and Muhammad Yasir Anwar did the data extraction. Wajeeha Aiman and Muhammad Yasir Anwar analyzed the data. Muhammad Ashar Ali, Wajeeha Aiman, Fatima Afzal and Hafsa Zahoor wrote the manuscript. Wajeeha Aiman critically reviewed the paper for the final submission.
Conflict of interest
All authors declare no conflict of interest.
Funding
No funding or sponsorship was received for publication of this article.
Financial disclosure statement
This manuscript has not been previously published and has not been submitted for publication elsewhere while under consideration.
Ethical approval
This article is based on previously conducted studies and the information is publicly available and does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Not applicable.
