Abstract
Sentinel lymph node biopsy (SLNB) is the standard of care for staging the clinically node-negative axilla in early breast cancer. Evidence guiding current practice describes dual localization technique using Patent blue dye and radioisotope (99mTc). Adverse effects of blue dye include 1:1000 risk of anaphylaxis, skin staining and loss of plane visibility, which may increase operative time and reduce resectional accuracy. The risk to a patient posed by anaphylaxis may be greater when operating in a unit without on-site ITU support – a situation more common with recent restructuring during the COVID-19 pandemic. Aim is to quantify the benefit of blue dye above radioisotope alone in identifying nodal disease. This is a retrospective analysis of prospectively collected sentinel node data including all consecutive sentinel node biopsies in a single center during the period 2016–2019.
In terms of results, 760 sentinel nodes were taken in 435 patients. 59 nodes (7.8%) were detected by blue dye alone; 120 (15.8%) ‘hot’ only, 581 (76.5%) hot and blue. 4 of the blue only nodes contained macrometastases but 3 of these patients had further hot nodes excised that also contained macrometastases. 1 out of 435 patients (0.2%) had macro metastatic disease identified as a result of blue dye alone which would have been missed had it not been used. In conclusion, the use of blue dye carries risk and offers little benefit in terms of staging in SLNB and its use may be unnecessary in the hands of the skilled surgeon. This study supports the omission of blue dye, which may be advisable if operating in units without ITU support. If larger studies support these figures, it may become as outdated.
Introduction
Blue dye was first described in 1992 for lymph node mapping in melanoma using intradermally injected Patent Blue [1]. Guiliano et al. later described the use of blue dye in the staging of breast cancer in 1994 [2]. Work with the use of radiolabeled colloids in sentinel node detection was also going on around this time, and the first significant publication of this work came about in 1993 by Krag et al. with their use of a gamma-ray detection probe [3].
The landmark ALMANAC trial [4], published in 2006 demonstrated reduced morbidity and better quality of life outcomes in sentinel lymph node biopsy (SLNB) versus axillary clearance/ four node sample, with no significant difference in rate of local recurrence. This trial was fundamental in driving current practice. In ALMANAC, 2 millilitres of radiolabeled colloid in the form of technetium-99 (Tc99) was injected intradermally into the diseased quadrant of the breast on the day of surgery and massaged for 5 minutes prior to lymphoscintigraphy. Then, in theatre 2 ml of Patent Blue V dye was diluted to 5 ml with 3 ml of saline and injected peritumorally 3–5 minutes before surgery. All stained and ‘hot’ nodes (radioactive counts >10× background count) were excised up to a total count of 4 nodes.
The NEW START surgical training programmed altered this protocol slightly – blue dye was injected subdermally either 2 ml 2.5% undilute or diluted to 5 ml with saline at a single periareolar site in the tumour quadrant [5]. NICE guidelines published in 2009 recommended SLNB as the preferred method for staging the axilla. Current UK practice is still largely based on the NEW START technique. Some units no longer use lymphoscintigraphy.
The results of the ALMANAC trial were further supported by the NSABP B-32 trial [3] in 2010. This was a large randomized controlled trial of 5611 women with invasive breast cancer who were randomized to SLNB plus axillary clearance (group 1) or SLNB alone (group 2) provided excised nodes were negative. Overall survival, disease free survival and regional control were equivalent in each group despite the fact that 3.8% of women in group 1 had cancer detected in a non-sentinel node. This provided further evidence that SLNB was an oncologically safe alternative to primary axillary clearance in clinically node negative-disease.
Despite never receiving a license for use in sentinel node biopsy, Patent Blue V dye in combination with Tc99 has since formed the mainstay of sentinel node identification in the UK. An important exception to this is in pregnancy where, if SLNB is performed, Tc99 is used without blue dye as recommended in guidelines from the Royal College of Obstetricians and Gynaecologists [6]. Uterine radiation measurements during lymphoscintigraphy indicate that this is safe [7] but the effects of blue dye (which would pass the placenta) on the foetus are unknown. In these patients there is a precedent for using Tc99 alone for SLNB.
The main concern with using blue dye has been allergic reaction, mediated through type I immediate hypersensitivity. The reported rates vary between 0.1 and 1.1%, in NEW START and ALMANAC anaphylaxis rates were 0.06% [8]. The Association of Breast Surgery produced a document relating to blue dye reactions [9] with a number of recommendations regarding the consent process plus investigation and management of patients exhibiting an allergic response. Although acknowledging no reported mortality the document recognizes the burden of adverse events (200–300 per annum in the UK) and states: “Every effort must be taken to minimize patient risk”.
This study seeks to investigate whether we could best minimize patient risk by eliminating the use of blue dye altogether without significantly affecting the sensitivity of SLNB for detecting disease. Evidence exists to show success rates and false negative rates using separate versus combined techniques in the past but there is a paucity of studies looking at the relative cancer detection rates and the likely impact of missing metastatic disease by omitting blue dye.
Materials and methods
The authors report a retrospective cohort study. All consecutive sentinel lymph node biopsies for breast cancer spanning a 3-year period between 1/1/2016 and 1/1/2019 within a single District General Hospital was included. Patients included were clinically node negative at time of surgery and patients post neoadjuvant chemotherapy were excluded. Data was collected prospectively for all sentinel nodes removed, allocating a score to describe the macroscopic appearance of the blue dye uptake in the node (blue score) and radioactivity count. The blue score was allocated by a single surgeon, with 0 denoting no blue staining, and 3 to denote deep blue staining. The method for identifying sentinel nodes was as described in NEW START and standardized within the department [5]. All nodes with a radioactivity count of >10% of the hottest excised nodes were considered sentinel nodes as were all nodes with blue score 0–3. These data were collated and cross-matched with electronic histology records for each node (each node was assigned a number at excision and which was stated in the histology report). Histological node status was defined as micrometastasis (deposit of tumour cells less than 2 mm in diameter), macrometastasis (deposit of tumour cells spanning more than 2 mm) or grossly positive.
Results were analysed to identify proportions of sentinel nodes detected by blue dye alone (blue only), radioisotope alone (hot only) or both (hot and blue). Within these groups histological status was analysed. All patients with positive ‘blue only’ nodes had the hot, blue and histological status of any other excised nodes analysed in order to identify patients whose staging and treatment may have been affected had blue dye been omitted.
Ethical approvals were not required as per the NIHR online decision tool (http://www.hra-decisiontools.org.uk/research/).
Results
760 sentinel nodes were excised from 435 patients (Fig. 1). All patients had breast cancer with clinically negative lymph nodes at the time of surgery. The mean number of nodes excised per patient was 1.7 in 2016, 1.8 in 2017 and 1.7 in 2018. Over the 3-year period this averaged at 1.7 sentinel nodes per patient. Mean age of patients was 64 years (median 65; range 23–98; standard deviation 11.9).
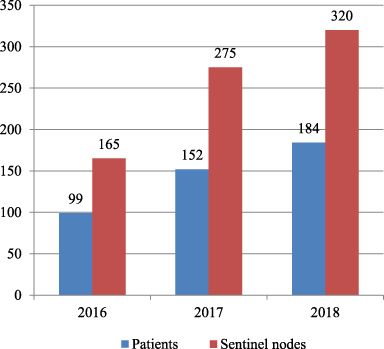
Number of patients undergoing sentinel node biopsy and number of sentinel nodes excised 2016–2018.
There were 760 sentinel nodes removed from 435 patients. 581 (76.5%) of the sentinel nodes excised were both hot and blue, 120 (15.8%) were hot only and 59 (7.7%) were blue only (defined as a blue score of more than or equal to 2) (Fig. 2).
A total of 4 nodes belonging to 4 patients which were blue only were identified as containing a macro metastasis which would be clinically significant and influence treatment pathways. Of these 4 patients, just one (0.2%) had no other positive nodes identified as ‘hot’.
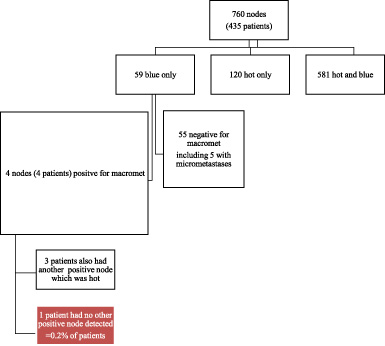
Flow chart to demonstrate proportion of node findings at sentinel node biopsy. ‘Hot’ is defined as a count of more than 10 times the background rate of gamma radiation. Blue is a subjective user scale, on a scale of 0–3 in intensity of staining macroscopically within the node.
This retrospective cohort study has demonstrated that within skilled hands, the false negative detection rate of sentinel node biopsy without the use of blue dye (technetium 99 only) is 0.2%. Balanced consideration needs to be made when comparing the risk of serious anaphylaxis from blue dye at 1%, along with the increased difficulties in dissection and increased operative time when using this unlicensed product. For surgical hubs without intensive care support on site that is required if using blue dye, it is an important consideration in terms of choosing the modality for sentinel node localisation.
There is no doubt that the visual aid given by blue dye in identifying sentinel nodes makes identification easier. A study by Derossis et al. demonstrated that isotope localization technique improves with experience (i.e. requires more skill) and the marginal benefit of blue dye declines simultaneously [10]. As skilled surgeons we should strive be able to identify sentinel nodes without the use of blue dye, just as we do for our pregnant patients. Concern exists regarding potentially missing positive sentinel nodes if we omit blue dye. This may be unfounded, given that a large meta-analysis of sentinel node mapping concluded sentinel lymph node identification success rates were higher with the combined technique versus radioisotope alone (odds ratio 2.03 with significant heterogeneity) but dual technique did not confer any advantage in lowering false negative rates [11].
This cohort study provides additional support in omitting blue dye, in order to provide safer patient care during times when the provision of intensive care support is limited, especially during the COVID pandemic and in consideration of maximizing resources such as setting up clean or “green” sites where intensive care may not be available in post pandemic recovery and ongoing provision of surgical services.
There are limitations to this study as it is a single center experience only and it has been conducted in a retrospective manner. There are also emerging sentinel node localization techniques, namely the use of superparamagnetic iron oxide mapping agents, that has been demonstrated to be non-inferior to the standard dual radioisotope and blue dye technique [12]. In view of the emerging technology, radioactive isotope and blue dye localization may become outdated especially in terms of resource utilization in terms of nuclear medicine and intensive care support if using the dual localization technique. However, this study supports radioactive colloid identification of nodes alone and supports it as single agent localization technique. There can also be concerns raised in terms of the 1 patient missed. However, in the advent of tailored adjuvant therapies such as multigene assay analysis of the tumour, the chance of this patient having an adverse oncological outcome and being “missed” is very low.
Conclusions
In conclusion, the report of this study is important as localizing sentinel lymph nodes with radioactive isotope is a safe method, missing only a small percentage of blue nodes that will not affect survival. Despite newer methods and innovation in sentinel lymph node localization considering the traditional, reliable, tested and an effective method is useful and should be considered.
Footnotes
Acknowledgements
None further.
6.
The authors declare that there is no conflict of interest regarding the publication of this paper.
7.
Data is available on request from UD.
8.
There was no external funding for this project.
9.
None
