Abstract
BACKGROUND:
BRCA1/2-associated invasive breast cancer has been extensively studied. However, there are few reports of ductal carcinoma in situ (DCIS).
OBJECTIVE:
This study aimed to investigate the clinicopathological and imaging findings of DCIS in patients with BRCA1/2 mutations.
METHODS:
This was a single-institution, retrospective study. We identified patients diagnosed with DCIS with BRCA mutations between September 2003 and December 2020. Clinicopathological data and mammography (MG), magnetic resonance imaging (MRI), and ultrasound (US) findings were reviewed.
RESULTS:
We identified 30 cancers in 28 patients; 7 (25.0%) patients had BRCA1 mutations, and 21 (75.0%) had BRCA2 mutations. The median patient age was 42 years. Screening was the most common reason for the detection of DCIS (50.0%), followed by occult cancer diagnosed by pathological examination after risk-reducing mastectomy (26.7%). The nuclear grade was most often 1 (46.7%), and 93.3% were estrogen and/or progesterone receptor positive. The detection rates of MG, MRI, and US were 64.3%, 72.0%, and 64.0%, respectively. The most common imaging findings were calcification (100%) on MG, non-mass enhancement (88.9%) on MRI, and hypoechoic area (75.0%) on US.
CONCLUSION:
BRCA-associated DCIS was more strongly associated with BRCA2, and imaging features were similar to those of sporadic DCIS. Our results are helpful in informing surveillance strategies based on genotypes in women with BRCA mutations.
Introduction
Breast cancer (BC) is the most prevalent cancer in Japanese women. Approximately 5%–10% of BCs are thought to be hereditary, and BRCA1 and BRCA2 are the most relevant genes in hereditary BC. Women with BRCA1/2 mutations develop BC at a higher rate than those without. BRCA1- and BRCA2-associated BCs exhibit different characteristics. BRCA1-associated BCs are often high-grade invasive cancers that exhibit benign morphologic features (mass with smooth margins, such as fibroadenoma and cysts) on magnetic resonance imaging (MRI), ultrasound (US), and mammography (MG) images [17]. In contrast, BRCA2-associated BCs exhibit pathological features similar to those of sporadic BCs (i.e., many hormone receptor-positive cases) and imaging features of typical malignant tumors (e.g., irregular mass, spiculae, and microcalcifications). With the adoption of surveillance and risk-reducing mastectomies in women with BRCA1/2 mutations, the probability of diagnosing ductal carcinoma in situ (DCIS) in women with BRCA1/2 mutations is increasing [2,6]. However, few retrospective studies have examined DCIS, especially DCIS imaging findings, in women with BRCA1/2 mutations. Thus, it is unclear whether DCIS also exhibits these features. Knowing the characteristics of DCIS in patients with BRCA1/2 mutations may be of great help in diagnosing BC and recommending genetic counseling. Therefore, we investigated the clinicopathological and imaging findings of DCIS in patients with BRCA1/2 mutations at our institution.
Methods and materials
Patients
This single-institution, retrospective study was approved by our institutional review board. Owing to the retrospective nature of the study, the institutional review board waived the need for informed consent. We identified women who had a proven mutation in BRCA1 and/or BRCA2, underwent surgery for BC and/or risk-reducing mastectomy, and were diagnosed with DCIS at our institution between September 2003 and December 2020. We included DCIS cases in women with contralateral invasive breast cancers (IBCs). We excluded double cancer cases with IBC and DCIS in the ipsilateral breast and ipsilateral breast tumor recurrence. Demographic and clinical information collected included age at the time of DCIS diagnosis; genotype; timing of genetic testing; opportunity to detect DCIS; imaging modality that first detected DCIS; family history of BC and/or ovarian cancer (OC) in second-degree relatives; histopathologic features of tumors; expression of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor type 2 (HER2); and imaging findings (MG, MRI, and US).
Imaging assessment
MG was performed using Senographe 800T and Senographe DMR units (GE films, MIN-R EV; screen, EV150) until August 2008. Digital MG using a Selenia unit from Hitachi was introduced in August 2008. Digital breast tomosynthesis (DBT) was first introduced in 2012. In principle, DBT was performed only for those who underwent MG for the first time at our hospital.
US was performed using a LOGIQ 7 system (probe frequency used, 10 MHz) from GE, HDI 5000 (probe frequency used, 12 MHz) from Philips (until September 2007), SSA 790A (probe frequency used, 12 MHz) from Toshiba Medical Systems (from October 2007), and EUB 7500 (probe frequency used, 14 MHz) from Hitachi Medical. All elasticity images were obtained using EUB 7500. A breast technologist first performed whole-breast US on all patients, followed by a double check by radiologists specializing in breast imaging.
We used 1.5T to 3.0T MRI scanners (Signa EXCITE HD, Optima MR450w, Discovery MR750w; GE Healthcare, Chicago, IL, and MAGNETOM Skyra fit and MAGNETOM Verio; Siemens Healthineers, Erlangen, Germany) with dedicated 4- or 8-channel (GE Healthcare) and 32-channel (Siemens Healthineers) breast array coils. The patients underwent MRI in the prone position. The imaging protocol consisted of high-resolution pre-contrast imaging and dynamic contrast-enhanced imaging in the axial plane before and 60, 180, 300, and 420 s after starting an intravenous injection of meglumine gadopentetate.
MG, MRI, and US scans that were performed as screening or preoperative evaluations were retrospectively reviewed by a radiologist with >30 years of clinical experience in breast radiology. We assessed the findings on MG (calcification, mass, architectural distortion, and focal asymmetric density), MRI (non-mass enhancement, mass, and focus), and US (mass, hypoechoic area, calcification, distortion, and duct dilatation). The term hypoechoic area is described in the Japan Association of Breast and Thyroid Sonology classification system and refers to a subtype of non-mass abnormalities found on US examinations. A hypoechoic area in the mammary gland is defined as a lesion that differs from the surrounding tissue and cannot be recognized as a mass. Although the American College of Radiology BI-RADS Atlas [1], which is a widely used guideline for diagnostic imaging, does not use the term hypoechoic area, we found it useful in our practice. Its value was confirmed by Watanabe et al., who reported that this concept was useful for detecting DCIS lesions [22].
Pathology and immunohistochemistry
All pathological data were based on pathological reports. Patients who were included in the analysis underwent definitive surgery, and their pathological specimens were reviewed by breast pathologists. Pathological data were reviewed, including the type, size, nuclear grade (NG), presence of comedo necrosis, and ER, PR, and HER2 status. The NG assessment was based on nuclear pleomorphism and mitotic counts. A numerical scoring system of 1–3 was used to ensure that the two factors were independently assessed. ER and PR expression was assessed by immunohistochemistry (IHC) and scored using the Allred method, with Allred scores of 3 or greater considered positive. HER2 status was assessed using IHC and scored as 0–3+.
Statistical analysis
Statistical analyses were performed using SPSS version 24 software (SPSS Inc., Chicago, IL, USA). The chi-square or Fisher’s exact tests were used to analyze the associations between BRCA mutations, clinicopathological data, and imaging findings. The Mann–Whitney U test was used to compare lesion size and age between the BRCA1 and BRCA2 groups. Differences were considered statistically significant at a p value of 0.05.
Results
Clinicopathological characteristics
We identified 291 BRCA1 and/or BRCA2 mutation carriers, and 199 patients underwent surgery for BC and/or risk-reducing mastectomy in our hospital. In total, 196 BC cases were diagnosed in 168 women. Of these, 30 DCIS cases were diagnosed in 28 women. Thirty-one women were found to have no cancer after risk-reducing mastectomy (Fig. 1).
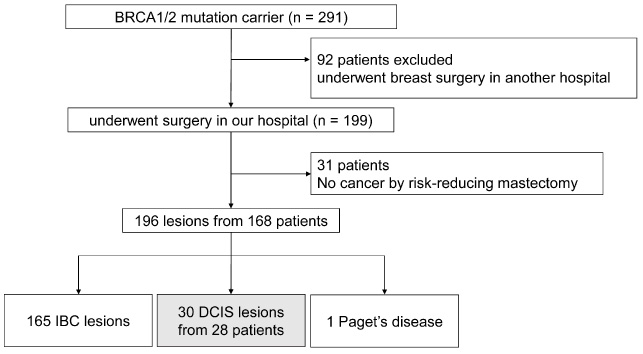
Flowchart of the study population. Multiple DCIS and IBC lesions on ipsilateral breast cancers were classified as IBC. DCISs from ipsilateral breast tumor recurrence and additional resections were excluded. IBC Invasive breast cancer, DCIS Ductal carcinoma in situ.
Table 1 shows the clinicopathological characteristics of the 30 DCIS lesions and 28 patients; 7 (25.0%) patients had BRCA1 mutations, and 21 (75.0%) had BRCA2 mutations. The mutation sites in BRCA are shown in Table 2. The median patient age was 42 (range, 30–63) years, and 24 and 5 patients had family histories of BC and OC, respectively. Twenty-two patients (78.6%) underwent genetic testing for DCIS prior to surgery. All seven women with BRCA1 mutations underwent genetic testing for DCIS before surgery. Screening was the most common reason for DCIS detection (15 lesions, 50.0%). Five of eight (62.5%) lesions in BRCA1-associated cancers were diagnosed as occult cancer by pathological examination of the risk-reducing mastectomy specimen. One (3.3%) lesion was identified as palpable. The median size of DCIS tended to be smaller for BRCA1-associated (5 mm) cancers than for BRCA2-associated (27 mm) cancers; however, the difference was not statistically significant (p = 0.067). Fourteen (46.7%) lesions were classified as NG1, and this grade was the most common in BRCA2 lesions; NG1 and NG3 were equivalent in BRCA1 lesions. Comedo necrosis was more common in BRCA2-associated cancers than in BRCA1-associated cancers. The year of surgery was three lesions until 2009, 10 lesions from 2010 to 2014, and 17 lesions after 2015.
Characteristics of patients and lesions
DCIS ductal carcinoma in situ, FH family history, BC breast cancer, OC ovarian cancer, CBC contralateral breast cancer, NG nuclear grade, MG mammography, US ultrasound, MRI magnetic resonance imaging, ER estrogen receptor, PR progesterone receptor, HER2 human epidermal growth factor receptor type 2. ∗Statistically significant.
BRCA1/2 mutations in DCIS patients: clinical aspects and molecular description
A adenine, T Thymine, C Cytosine, G Guanine, dup duplication, del deletion, Cys Cysteine, Arg Arginine, Leu Leucine, X unspecified amino, Ser serine, Val Valine, fs frame shift, Glu Glutamic acid, Ter termination, Ala Alanine, Ile Isoleucine, Tyr Tyrosine, Lys Lysine, Trp Tryptophan, His Histidine, ∗Termination. †Results from companies (Myriad genetics, FALCO biosystems, Laboratory Corporation of America, Ambry Genetics Corporation). Empty field: No protein changes due to changes in nucleotides.
Table 3 shows the imaging findings for DCIS with BRCA1/2 mutations. Thirty DCIS lesions were identified in our study. Among these, 28, 25, and 25 were examined preoperatively using MG, MRI, and US, respectively. MG was able to correctly identify malignancy in 18 (64.3%) of the 28 lesions, of which 2 (11.1%) were BRCA1-associated and 16 (88.9%) were BRCA2-associated. BRCA2-associated DCIS had more lesions identified by MG than BRCA1-associated DCIS; however, the difference was not statistically significant (p = 0.063). Malignant calcifications were observed in all 18 lesions. Mass and architectural distortion with calcifications were observed in one lesion. The most common type of calcification was amorphous (11/18 lesions, 61.1%). Grouped calcifications were found in 9 (50.0%) of 18 lesions, and segmental calcifications were found in 8 (44.4%) of 18 lesions. DBT was performed in 11 of 30 cases. There were no new cases in which malignant findings were detected using DBT. Breast density on MG was extremely dense in 2 cases (7.1%), heterogeneously dense in 20 cases (71.4%), and scattered fibroglandular in 6 cases (21.4%), and no case was entirely fatty. The detection rates of malignancy were 100% (2/2 cases), 80% (16/20 cases), and 0% (0/6 cases), respectively. The malignancy findings for each breast density were all calcification in extremely dense, 15 calcifications, and 1 mass in heterogeneously dense.
Imaging findings of DCIS
Imaging findings of DCIS
DCIS ductal carcinoma in situ, US ultrasound, MRI magnetic resonance imaging. a The presence or absence of each finding was evaluated for each lesion, and each finding may overlap. ∗Statistically significant.
MRI was able to correctly identify malignancy in 18 (72.0%) of 25 lesions, of which 2 (11.1%) were BRCA1-associated and 16 (88.9%) were BRCA2-associated. BRCA2-associated DCIS were easier to identify by MRI than BRCA1-associated DCIS, and a significant difference was noted (p = 0.007). Of the 18 lesions, 16 (88.9%) showed non-mass enhancement. Background parenchymal enhancement on MRI was marked in three cases (12.0%), moderate in six cases (24.0%), mild in seven cases (28.0%), and minimal in nine cases (36.0%). The malignancy detection rates were 66.7%, 50.0%, 85.7%, and 77.8%, respectively.
US was able to correctly identify malignancy in 16 (64.0%) of 25 lesions, of which 2 (12.5%) were BRCA1-associated and 14 (87.5%) were BRCA2-associated. Hypoechoic areas were observed in 12 lesions, which was the most frequent finding. Calcifications were observed in nine lesions.
The detection rates of the three modalities were comparable. The detection rate for BRCA1-associated DCIS was lower than that for BRCA2-associated DCIS in all three modalities. Twenty-four cancers were examined using three modalities: MG, MRI, and US. Figure 2 shows the number of cancers identified using each imaging modality. Twelve lesions were detected using all three modalities. Figure 3 shows one such case. Two lesions were detected only by MRI and were from women with BRCA2 mutations, which were classified as NG1, ER- and PR-positive, and HER2-negative. The DCIS sizes were 28 and 35 mm, respectively. Both lesions showed non-mass enhancement on MRI. Figure 4 shows a 35-mm lesion. One lesion was detected only by MG (Fig. 5). It appeared as an increase in calcifications in a woman with a mutation in BRCA2. Its DCIS size was 85 mm, and it was classified as NG2, ER- and PR-positive, and HER2 2+. One lesion detected by US was also associated with BRCA2. It appeared as an indistinct oval mass with calcifications. Calcifications were detected by MG. However, these calcifications were coarse and considered benign. Its DCIS size was 6 mm, and it was classified as NG1, ER- and PR-positive, and HER2 2+.
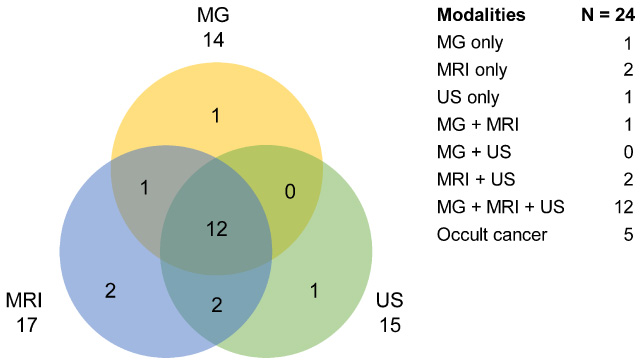
Breakdown of the modalities used to detect the cancers. A total of 24 lesions were evaluated by three modalities, MG, MRI, and US. The Venn diagram shows the lesions detected by each modality as well as those detected by multiple modalities. The information is also summarized on a list on the right. MG mammography, MRI magnetic resonance imaging, US ultrasound.
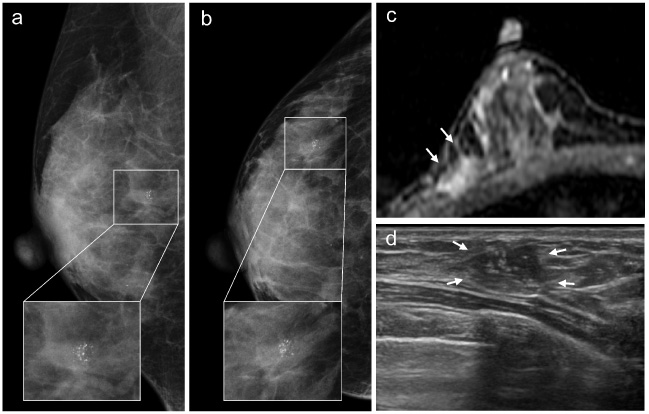
Case detected by MG, MRI, and US. This lesion was detected in a woman with a BRCA2 mutation by a screening MG. The DCIS was 25 mm in size and NG1, ER, and PR-positive, and HER2 1+. (a,b) Right medial-lateral oblique and cranio-caudal MGs showed amorphous-grouped calcifications. (c) Axial contrast-enhanced T1-weighted MRI showed a focal, non-mass enhancement. (d) US showed a hypoechoic area with calcifications. MG mammography, MRI magnetic resonance imaging, US ultrasound, DCIS ductal carcinoma in situ, NG nuclear grade, ER estrogen receptor, PR progesterone receptor, HER2 human epidermal growth factor receptor type 2.
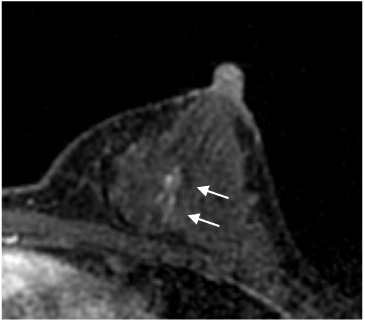
Case detected by MRI only. This lesion was detected in a woman with a BRCA2 mutation by a preoperative examination for contralateral breast cancer. The DCIS was 35 mm in size and NG1, ER and PR-positive, and HER2 1+. Axial contrast-enhanced T1-weighted MRI showed a focal, non-mass enhancement. MRI magnetic resonance imaging, DCIS ductal carcinoma in situ, NG nuclear grade, ER estrogen receptor, PR progesterone receptor, HER2 human epidermal growth factor receptor type 2.
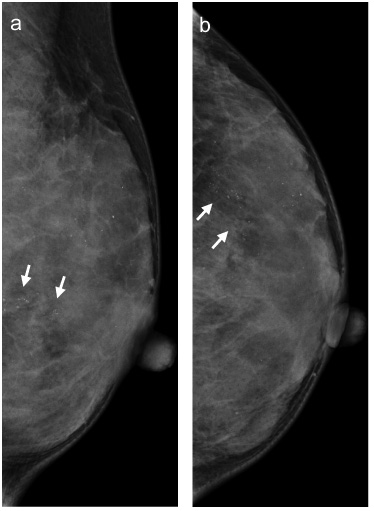
Case detected by MG only. This lesion was detected in a woman with a BRCA2 mutation by a screening MG. In addition to the previously existing calcifications, new segmental calcifications are detected in MG. On the MRI, the lesion was not detected because of the basement parenchymal enhancement marked. The DCIS was 85 mm in size and NG2, ER and PR-positive, and HER2 2+. (a) Mediolateral oblique view and (b) craniocaudal view in right breast mammography. MG mammography, MRI magnetic resonance imaging, DCIS ductal carcinoma in situ, NG nuclear grade, ER estrogen receptor, PR progesterone receptor, HER2 human epidermal growth factor receptor type 2.
Eight cases of DCIS were identified using risk-reducing mastectomy. Two patients had malignant findings on MRI. In one case, preoperative MRI was not performed. The other five cases showed no malignant findings on any of MG, US, and MRI examinations, and these were regarded as true occult DCIS. Table 4 shows the pathological features of the five occult DCIS cases. All five cancers were BRCA1-associated. The largest occult cancer was 6 mm in size. More than half of the occult cancers were NG1 or NG2, and hormone receptor positive and HER2 negative. Two lesions were NG3. One of them was triple negative (TN), and the other was hormone receptor positive and HER2 positive.
Characteristics of occult DCISs
Characteristics of occult DCISs
DCIS ductal carcinoma in situ, NG nuclear grade, ER estrogen receptor, PR progesterone receptor, HER2 human epidermal growth factor receptor type 2.
In this study, we investigated the clinicopathological and imaging findings of BRCA-associated DCIS. Understanding the clinicopathological and imaging characteristics of BRCA-associated DCIS is helpful in informing surveillance in women with BRCA mutations.
BRCA-associated DCIS was more commonly detected in women with mutations in BRCA2 than in those with mutations in BRCA1. Previous reports have reported similar results [5,9,17]. In a single-center BRCA-associated DCIS survey conducted by Liu et al. [9], there were nine DCIS cases among BRCA1/2 mutation carriers. Of these, only one BRCA1-associated DCIS was observed, and it was characterized as NG3 and TN. Warner et al. reported that one of eight pure DCIS cases diagnosed in patients with BRCA1/2 mutations was BRCA1-associated [20].BRCA1-associated cancers have aggressive characteristics (TN type and high grade) and easily become invasive, making the occurrence of BRCA1-associated DCIS rare. In our study, 8 of the 30 DCIS cases were BRCA1-associated, and this prevalence was higher than that reported in previous studies. This may be because five of the eight cases were occult cancers. Six (75%) of the eight BRCA1-associated DCISs were ER positive, and only two were TN DCIS. Among DCIS cases, unlike in IBC cases, the TN type was not common. This may also be due to the rarity of high-grade DCIS cases.
The most common imaging findings of the cancers were calcification on MG, non-mass enhancement on MRI, and hypoechoic area on US. These imaging features were similar to those of sporadic DCIS. Some studies have found that BRCA1-associated IBC shows benign imaging features more often than sporadic IBC. In contrast, the imaging features of BRCA2-associated IBCs are typical malignant findings [4,17]. This has also been observed in Japanese women [10]. The imaging features of IBCs with BRCA mutations reflect the pathological findings. That is, many BRCA1 cancers are high-grade, such as TN, whereas BRCA2 cancers are often hormone receptor positive. In this study, BRCA-associated DCISs were often hormone receptor positive and had low NG. These pathological findings influenced the BRCA-associated DCIS imaging findings. We found no differences in DCIS imaging findings between the BRCA1 and BRCA2 cases.
The detection rates of MG, MRI, and US in our study were 64.3%, 72.0%, and 64.0%, respectively. Previous studies have reported that the detection rate of IBC in high-risk women and sporadic DCIS by MRI was higher than that of MG and US [8,13,14,17,21]. Reports of the detection rate of BRCA-associated DCIS vary [4,13,19,20]. Warner et al. [20] reported that the detection rates of MG, MRI, and US were 10%, 100%, and 0%, respectively, whereas Ha et al. [4] reported detection rates of 50.0%, 85.7%, and 78.6%, respectively. MRI is useful for BC surveillance of women with BRCA1/2 mutations because of its high sensitivity. Annual MRI surveillance for high-risk women is recommended by some guidelines, such as the American Cancer Society Guideline for Breast Screening with MRI as an Adjunct to Mammography (2007) (ACS 2007) [15], National Comprehensive Cancer Network Guidelines Version 2. 2022 for Genetic/Familial High-Risk Assessment (http://www.nccn.org), and Guidelines for Diagnosis and Treatment of Hereditary Breast and Ovarian Cancer 2021 by the Japanese Organization of Hereditary Breast and Ovarian Cancer. The addition of MG and US to MRI screening may have limited utility for high-risk women because of the lower sensitivities of MG and US than that of MRI. However, in this study, the detection rates of MG, MRI, and US were comparable for BRCA-associated DCIS. One of the reasons for discrepancies in detection rates between modalities is the difference in patient backgrounds. This is because patient characteristics such as age, breast density on MG, and background parenchymal enhancement on MRI can lead to different rates of lesion identification [11,16,18]. In addition, as a reason for the high detection rate of US, there is data that the US was useful for early detection of BC in Japanese women [12]. Another possible explanation for the observed high sensitivity of MG and US may be related to diagnostic suspicion bias in retrospective studies. It is necessary to further study the optimal examination modalities for each situation.
In our study, there were differences in the detection rates between genotypes. The detection rate of BRCA1-associated DCIS on MRI was significantly lower than that of BRCA2. A similar trend was observed in MG (28.6% and 76.2%, respectively) and US (28.6% and 72.2%, respectively); however, no statistically significant difference was observed. All five occult cancers that underwent the three examinations (MG, MRI, and US) were BRCA1-associated. Yamauchi et al. showed a relatively higher incidence rate of occult cancer (11.3%) in risk-reducing mastectomy specimens from Japanese women [23]. Of the five occult DCISs detected in this study, four were associated with BRCA2. Our results differ from these findings. In our study, occult cancers were <6 mm in size, and the size of DCISs detected by any modality was ≥6 mm. Size is considered to be related to lesion detection. We suggest that it is difficult to detect DCIS in women with BRCA1 mutations because of the small size of these lesions. This may be because BRCA1-associated cancers easily progress to invasive carcinoma owing to their aggressive nature.
Currently, individualization of surveillance methods according to BRCA genotype is being discussed because BRCA1- and BRCA2-associated IBCs have different characteristics [7,19]. In BRCA-associated DCIS, there were not many differences between BRCA genotypes in pathological and imaging findings; however, the detection rates of BRCA1-associated DCIS were lower than those of BRCA2. In BRCA1 mutation carriers, it was difficult to detect cancer in the DCIS state before it became invasive. The results of this study may change the weighting of options for BRCA mutation carriers when choosing between imaging surveillance and risk-reducing mastectomy, depending on the mutation type: BRCA1-positive patients are less likely to have DCIS detected by routine imaging surveillance and, therefore, may be more likely to elect for risk-reducing mastectomy; for BRCA2, routine imaging surveillance may detect BC at an earlier stage and, therefore, may prefer surveillance over risk-reducing mastectomy. The survival benefits of early DCIS detection in BRCA1/2 mutation carriers are unclear, and further investigation is required to determine the prognostic impact of surveillance and risk-reducing mastectomy for each genotype. However, for women who are more likely to have high-grade BC, diagnosing early DCIS may be more beneficial because it avoids additional therapy. The results of this study may provide a more tailored option for BRCA mutation carriers when considering the individual surveillance methods.
This study has some limitations. This retrospective study was conducted at a single institution. Not all patients underwent MG, MRI, or US, which could potentially lead to a selection bias. However, we believe that the effect on the results is minimal because the missing data for each method represented only a small percentage of the lesions. Because the examinations were performed over a long period, the results may be somewhat different due to the use of less technologically advanced imaging in earlier cases. All diagnostic imaging equipment have undergone technological innovations over the past 17 years, from 2003 to 2020. For the equipment used in this study, the MG pixel size was changed from 100 to 70 μm, and digital conversion was also changed from indirect conversion to direct conversion. DBT has been introduced since then. DBT has been performed at our hospital since 2012, and there is a possibility of detection bias depending on whether DBT was performed. However, there was no difference in the detection of malignant findings among the 11 cases in which DBT was performed in the current study. This is due to the fact that most of the MG findings were calcified lesions. This may be because DBT is superior in detecting masses and architectural distortion but is comparable with MG in detecting calcifications [3]. MRI has progressed from 1.5T to 3.0T and from 4 to 8 and 32 channels. For US systems, the frequency of the probe has been improved from 10 to 14 MHz, and the spatial resolution has been improved as a result of the detection capability of superficial US. It is possible that changes in these imaging techniques may be related to lesion detection. However, the quality control of the images from the early years was also well maintained. In addition, only 3 of the 30 cases were older than 2009, suggesting that the image quality from the early years was similar or not significantly different for the purposes of our study.
Conclusion
Our study showed the clinicopathological and imaging features of BRCA-associated DCIS at our institution. BRCA-associated DCIS was more strongly associated with BRCA2-positive, NG1, and hormone receptor-positive cases. The imaging features were similar to those observed in sporadic DCIS. The detection rates of DCIS in MG, MRI, and US were comparable. Our results are helpful in informing surveillance in women with BRCA mutations.
Footnotes
Acknowledgements
The authors thank all staff from the Breast Surgical Oncology, Radiology, Pathology, and Clinical Genetics departments of St Luke’s International Hospital for their help in collecting clinical data.
Funding
This work was supported by AMED (20ck0106555h0001).
Conflict of interest
Hideko Yamauchi received grants from AMED during the conduct of the study and grants from AstraZeneca outside the scope of the submitted work. The other authors have no conflicts of interest to declare.
Ethical approval
For this type of study, formal consent is not required.
Informed consent
Owing to the retrospective nature of the study, the institutional review board waived the need for informed consent.
