Abstract
INTRODUCTION:
Programmed death ligand 1 (PD-L1) plays a role in tumor escape and progression by inactivating T lymphocytes. The aim of the study reported here was to determine the relationship between the expression of PD-L1 and histopathological grade, stage of disease, and the occurrence of metastasis in breast cancer.
METHODS:
The observational cross-sectional study involved analyzing the expression of PD-L1 by immunohistochemistry.
RESULTS:
PD-LI was expressed in 43 of 60 patients with breast cancer (71.6%), mostly with a moderate histopathological grade (58.3%) and at an advanced stage (50%). Associations between the expression of PD-L1 and histopathological grade (p = 0.011), stage of disease (p = 0.009), and the occurrence of metastasis (p = 0.01) were significant, with an odds ratio of 5.
CONCLUSION:
The associations between the expression of PD-L1 and histopathological grade, disease stage, and occurrence of metastasis were all significant in cases of breast cancer in the sample. Those findings suggest that the expression of PD-L1 increases the progression of breast cancer.
Keywords
Introduction
As the most common form of cancer worldwide after lung cancer, breast cancer remains a major global health problem that almost exclusively affects women. In fact, among women, breast cancer has the highest mortality rate a whopping 15.0% of all cancer [1]. More than half of all cases of breast cancer, especially in countries of low to medium income, are diagnosed at an advanced stage [2]. In Indonesia, according to data obtained from GLOBOCAN, an estimated 58,256 new cases of breast cancer (i.e., 30.9% of all cases of cancer among women) are diagnosed each year, and an estimated 22,692 (38.95%) are fatal. Thus, for women in Indonesia, breast cancer is the top-ranked cause of death [1,3]. That astonishingly high mortality rate can be reduced, however, if methods to predict the course of breast cancer, its clinical outcomes, and its response to therapy are available for use in daily practice.
Cancer cells exhibit several characteristics that distinguish them from other cells. To begin, the pathomechanism of breast cancer and cancer, in general, is induced by the interaction between genetic and environmental factors. From there, cancer occurs due to abnormalities in cell growth that include increased proliferation, increased angiogenesis, and decreased apoptosis [4]. At present, several characteristics of cancer cells have been identified, including insensitivity to growth-inhibiting signals, the ability to respond to their growth signals, changes in cellular metabolism, the inhibition of apoptosis, the maintenance of angiogenesis, the ability to invade and metastasize, genomic instability, the use of inflammatory factors for tumor growth, and the ability to evade immune response [5].
Research on cancer conducted in the last decade has often focused on immunotherapy as a treatment modality, especially in association with the expression of programmed death-ligand 1 (PD-L1). PD-L1 is a ligand for the programmed death-1 (PD-1) receptor expressed by tumor cells in an attempt to evade an immune response, such that the expression of PD-L1 triggers tumor growth and progression [6]. PD-L1 plays a role in tumor escape and progression by activating T lymphocytes, and its interaction on effector T cells with PD-1 can inhibit T cell receptor (TCR) signal transduction, thereby inhibiting the cytotoxic activity of T cells. Whereas Src homology 2 domain-containing protein tyrosine phosphatase 1 (SHP-1) can regulate the activity of T cells, the inhibition of cluster of differentiation 8 (CD8+) and SHP-1 via sodium stibogluconate increases the function of the tyrosine-based inhibitory motif and the immunoreceptor tyrosine-based switch motif (ITSM). In turn, ITSM can cause tyrosine phosphorylation on the ITSM motif due to PD-1 and recruit both SHP-1 and Src homology region 2 domain-containing phosphatase-2 (SHP-2). After that uptake, the signaling pathway can inhibit the stop signal and the interaction of T cells and dendritic cells. At that point, the blockade of TCR signal transduction causes the inhibition of phosphatidylinositol 3-kinase (PI3K) and Akt (i.e., protein kinase B) and mitogen-activated protein kinase signaling. Ultimately, the inhibition of PI3K activation suppresses the expression of extra-large B-cell lymphoma and the activation of Akt, which eventually causes apoptosis in T cells [7].
Several factors, including metastasis, can affect the prognosis of breast cancer. In fact, responsible for 90% of all deaths due to breast cancer, metastasis is the top cause of death [8]. Approximately 20–30% of patients with breast cancer experience relapse with distant metastases and an average of 5–15% of instances of breast cancers have already metastasized at the time of diagnosis [9]. In addition to metastatic factors, the prognosis of breast cancer is also influenced by tumor size and lymph node status, both of which affect the histopathological grade [10]. Histopathological grade has been identified as an important prognostic factor because a higher or more advanced histopathological stage is associated with a low long-term probability of survival [11]. The frequency of recurrence in high-grade carcinoma is also higher than in low-grade carcinoma [12].
The high morbidity and mortality rate of breast cancer indicates that the current management of breast cancer is not optimal and that other therapeutic modalities are needed, one of which is immunotherapy through the interaction of PD-L1 and PD-1. In the study presented here, the aim was to determine the relationship between the expression of PD-L1 and tumor progression in breast cancer in terms of histopathological grade, stage of disease, and the occurrence of metastasis.
Methods
The research was conducted in the Surgical Oncology Division of the Departments of Surgery at Dr. Wahidin Sudirohusodo Hospital and Hasanuddin University Hospital in Makassar, Indonesia, from August 2019 to July 2020. The expression of PD-L1 was examined using immunohistochemical methods in the Faculty of Medicine’s Anatomic Pathology Laboratory at Hasanuddin University Makassar.
Study design
In an observational cross-sectional study, histopathological grade, stage of disease, and the occurrence of metastasis were dependent variables, whereas the expression of PD-L1 was the independent variable.
Population and sample
The study’s population consisted of women diagnosed with breast cancer admitted to the Surgical Oncology Unit in the Department of Surgery at Dr. Wahidin Sudirohusodo Hospital. The inclusion criterion was being a woman with invasive ductal carcinoma breast cancer who had never received cancer therapy, whereas the exclusion criterion was having lobular carcinoma or another malignancy. The sample comprised 60 individuals who met those criteria.
Data collection
After data regarding the patient’s age, histopathological grade (i.e., based on the Modified Scarff–Bloom–Richardson grading system), tumor size, lymph node involvement, and presence of metastases were collected, the overall operation and histopathological examination procedures to be performed were described to all patients.
Immunohistochemistry
Tissue samples obtained from each patient’s cancer tissue according to the clinical stage of breast cancer were taken to the Anatomic Pathology Laboratory at Hasanuddin University Hospital for the immunohistochemical examination of the expression of PD-L1. The primary antibody was an anti-human PD-L1 rabbit monoclonal antibody (Catalog No. RM0324RTU7; Medaysis, Livermore, CA, USA), used according to the manufacturer’s instructions [13–15].
Results
Table 1 shows the characteristics of the 60 patients in the sample, more than 36 of whom (60%) were less than 50 years old. The most common histopathological grade was moderate (n = 35, 58.3%), the most common subtype was luminal A (n = 19, 31.7%), and the most common stage was advanced (n = 30, 50.0%). The positive expression of PD-L1 was found in 43 patients (71.6%).
Patients characteristics
Patients characteristics
Figure 1 offers an immunohistochemical microphotographic representation of the negative expression of PD-L1, marked by pale unstained cytoplasm, whereas Fig. 2 shows its positive expression, marked by cytoplasm that is stained brown.
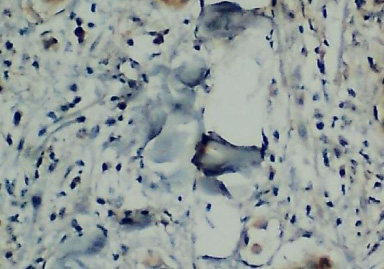
Immunohistochemical examination results with the negative expression of PD-L1.
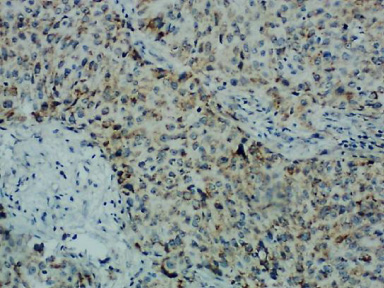
Immunohistochemical examination results with the positive expression of PD-L1.
Table 2 details the relationship between the expression of PD-L1 and histopathological grade. A significant association emerged between the expression of PD-L1 and histopathological grade (p < 0.05). The percentage of grade III breast cancer was higher in patients showing a positive expression of PD-L1 (37.2%) than in ones showing a negative expression (11.8%), while the percentages of grades I and II were higher in patients showing a negative expression (29.4% and 58.8%, respectively) than a positive one (7.0% and 58.1%, respectively).
Association between the Expression of PD-L1 and histopathological grade
Table 3 details the relationship between the expression of PD-L1 and the disease stage. A significant association emerged between the expression of PD-L1 and disease stage (p < 0.01), in which the percentage of advanced-stage cancer was higher among patients showing a positive expression of PD-L1 (60.5%) than among ones showing a negative expression (23.5%). Meanwhile, the percentages of early-stage and locally advanced stages were higher in cases of negative PD-L1 expression (35.3% and 41.2%, respectively) than in cases of positive expression (7.0% and 32.5%, respectively).
Association between expression of PD-L1 and disease stage
Table 4 shows the relationship between the expression of PD-L1 and the occurrence of metastasis. The association between the expression of PD-L1 and metastasis was significant (p < 0.05). The percentage of metastasis was higher among patients with a positive expression of PD-L1 (60.5%) than among ones with a negative expression (23.5%), while the percentage of non-metastasis was higher among cases of negative PD-L1 expression (76.5%) than among cases of positive expression (39.5%). Odds ratios reveal that patients showing a positive expression of PD-L1 had a fivefold greater risk of developing metastasis than ones showing its negative expression.
Association between expression of PD-L1 and occurrence of metastasis
The incidence of breast cancer increases in women less than 50 years of age. In research conducted in the Anatomic Pathology Department at Cipto Mangunkusumo Hospital in Jakarta, Indonesia, individuals more than 40 years old formed the largest age-based group that had developed breast cancer (78.2%) [16]. Across Asia, by comparison, 40.6% of cases of breast cancer are diagnosed among individuals 40 to 60 years old [17], whereas data obtained from the surveillance, epidemiology, and results (SEER) incidence database in the United States show that the largest age group for breast cancer is that of individuals more than 60 years old [18]. The differences may be due to differences in lifestyle, dietary patterns, and/or certain genes related to race. In the sample of the study presented here, most of the subtypes were luminal A (31.7%), which generally has a good prognosis, while less frequent was the luminal B subtype (20.0%). The American Cancer Society has reported that luminal A, accounting for 40% of cases, is the most common subtype of breast cancer [19].
A high expression of PD-L1 causes tumors to evade immune responses and, as a result, grow progressively while mitosis continues, all of which worsens the degree of cell differentiation. Histopathological grade, assessed based on tubule and glandular formation, nuclear pleomorphism, and rate of mitosis, is significantly correlated with survival from breast cancer and thus serves as an important prognostic factor in clinical decision-making [20]. Engstrom has stated that the degree of histopathology is indeed an important prognostic and predictive factor [21]. In short, the higher a tumor’s grade, the more progressive its growth. Li et al. also detected an association between the expression of PD-L1 and histopathological grade (p = 0.03) and found that a majority of cases expressing PD-L1 were cases of grade III breast cancer (54.55%) [22].
Added to that, Li et al. reported an association between the expression of PD-L1 and stage of breast cancer (p = 0.01), in which the positive expression of PD-L1 occurred mostly at an advanced stage (59.01%) [22]. Yuan et al. also found a relationship between the expression of PD-L1 and the increased stage of breast cancer due to the interaction of cancer cells with T lymphocytes following the binding of PD-1 and PD-L1 [23]. As a consequence, cancer cells were not inhibited from destroying immune cells and resulted in more progressive cancer cells that would eventually increase tumor size, lymph node metastases, and distant metastases. A high expression of PD-L1 owing to cancer cells will inevitably cause cytotoxic T cells to become exhausted, thereby reducing the proliferation and immune response of T cells, and as a result, immune cells will become unable to eliminate cancer cells.
In other research, Litvin et al. observed a significant association between the expression of PD-L1 and the incidence of metastasis [24], while Manson et al. found a positive expression of PD-L1 in 24.0% of metastatic cases and observed that PD-LI’s expression became positive in 22.4% of cases [25]. On top of that, Kronemyer reported that 23.8% of metastatic cases expressed PD-L1 [26]. In another study, one involving 614 patients with breast cancer, positive expressions of PD-L1 were observed in 75% of cases examined with the SP263 assay and in 81% of cases examined with the 22C3 assay [27]. Each stage of cancer cell metastasis interacts with both pro-metastatic immune cells such as Treg and M2 macrophages and anti-metastatic ones such as natural killer (NK) cells, cytotoxic T cells, and M1 macrophages [28]. In the process of immunoediting, the success of anti-metastatic immune cells will lead to the elimination of cancer cells, while the balance between pro and anti-metastatic immune cells will cause cancer cells to enter a dormant phase of equilibrium. Conversely, the weakening of anti-metastatic immune cells will cause tumor cells to enter the escape phase and develop into metastases. One mechanism of cancer cells is to express PD-L1, which will bind to cytotoxic T immune cells so that cancer cells are protected from destruction. In the metastatic stage, that interaction occurs following the growth of the primary tumor while in circulation (i.e., as circulating tumor cells) and during colonization at distant metastatic sites. The ability of cancer cells to manipulate the function of cytotoxic T cells increases the incidence as well as the survival of cancer cell metastases.
Conclusion
The associations between the expression of PD-L1 and histopathological grade, disease stage, and occurrence of metastasis were all significant in cases of breast cancer in the sample. Those findings suggest that the expression of PD-L1 increases the progression of breast cancer. However, the sample was small, and the investigations for determining the presence or absence of distant metastases were standard, both of which acted as limitations in the study.
Footnotes
Acknowledgements
The author would like to thank all supervisors and staff in the Surgical Oncology Division of the Faculties of Medicine at Hasanuddin University and at Dr. Wahidin Sudirohusodo Hospital, Makassar, who helped with conducting the research.
Conflict of interest
None.
Funding source
None.
