Abstract
BACKGROUND:
A recently introduced CD4+ T subset that mainly secretes interleukin (IL-) 22 has been reported to be associated with a variety of tumors, including colon, gastric, hepatocellular, and small- and large-cell lung carcinoma. Both tumor-promoting and - suppressing roles have been suggested for these cells. In the present study, we aimed to investigate the frequency of IL-22-producing subsets in tumor-draining lymph nodes (TDLNs) of the patients with breast cancer and determine their association with the clinicopathological characterizations of the disease.
METHODS:
Thirty untreated women diagnosed with breast cancer were enrolled and their axillary lymph nodes were dissected during surgery. Mononuclear cells were isolated using Ficoll density gradient, activated, permeabilized, and stained by fluorochrome-conjugated antibodies against CD4, IL-22, IL-17, and IFNγ. The cells were then acquired on the FACSCalibur flow cytometer, and raw data was analyzed by the FlowJo software package (V10).
RESULTS:
Our results demonstrated that 2.39% ± 0.39 of CD4+ lymphocytes in TDLNs of patients with breast cancer produced IL-22. Among them, 0.64% ± 0.8 just produced IL-22 but were negative for IFNγ and IL-17. Statistical analysis indicated that the frequency of CD4+IL-22+ cells was significantly higher in the patients with stage III and the ones with 3–9 tumor involved lymph nodes (N2) compared to those with stage II and those having 1–3 tumor involved lymph nodes (N1) (P = 0.008 and P = 0.004, respectively).
CONCLUSION:
The higher frequency of IL-22-producing cells in draining lymph nodes of patients with more advanced tumors (higher stage (stage III) and more involved lymph nodes) suggests a role for IL-22-producing cells in the tumor progression and invasion. However, further studies with larger sample size and more functional studies are needed to clarify the role of IL-22-producing cells in breast cancer pathogenesis.
Introduction
Regional Lymph nodes (LNs) are the first place of tumor metastasis in human breast cancer. LNs normally contribute in generation and regulation of immune responses, while tumor-draining lymph nodes (TDLNs) develop into an immunotolerant milieu which is an important step in immune suppression and cancer progression [1]. Breast cancer has been considered as an immunologic tumor, and the association between the immune system components and tumor cells has been quite challenging [2]. In addition to tumor eradiation, the host immune system may contribute to the progression, invasion and metastasis of cancer.
Up to the present, various CD4+ T helper (Th) cells with different functions including Th1, Th2, Th9, Th17 and Th22 have been discovered. Current researches show that the balance between these cells and their cytokines serve as the main factor in maintaining homeostasis and tumorigenesis [3,4]. In this regard, Th22, a newly described CD4+ subset by the production of interleukin (IL)-22 may play an important role in cancer [5]. Although other CD4+ cells such as Th1 and Th17 can also produce IL-22, Th22 can be marked by presenting high level of aryl hydrocarbon receptor (AHR) and low level of T-bet and ROR𝛾t. Accordingly, Th22 lymphocytes are unique in IL-22 production, while they do not produce IL-17 and IFN-γ [5–7]. IL-22 is emerged as a dual-function cytokine in cancer and contributes to both tumor promoting and tumor suppressing base upon its concentration and the duration of over-expression [8]. Recent studies have shown that increased IL-22 expression and its receptor complex formation lead to tumor progression and subsequent decrease in the overall survival rate in some cancers such as gastric, colorectal and hepatocellular cancers [9–11]. Although the mechanism is not fully understood, it is conjectured that IL-22 acts through its downstream mediator STAT3 which increases cellular proliferation [9]. However, the role of Th22 and IL-22 in breast cancer has not well been investigated. Thus, the present study aimed to investigate the mean frequency of IL-22-expressing CD4+ subset in TDLNs of patients with breast cancer and determine their association with the clinicopathological characteristics of the disease.
Experimental
Patients
Thirty patients with breast cancer (48.2 ± 2.14 years old) who did not receive any pre-operative chemotherapy, radiotherapy and/or immunotherapy were enrolled in this study. All patients were undergone surgical resection in public hospitals affiliated to Shiraz University of Medical sciences (Shiraz, Iran). The study was approved by the Ethics Committee of Shiraz University of Medical Sciences, Shiraz, Iran (IR.sums.med.rec.1396.s205) and informed consent was obtained from each subject before sampling. After pathological examination, a part of axillary lymph nodes (ALNs) was transferred to cancer immunology laboratory for further investigation.
Lymph Node Mononuclear Cells (LNMCs) isolation
Mononuclear cells were isolated from lymph nodes as previously described [12,13]. Briefly, fresh lymph nodes were mechanically minced into small specimens in RPMI 1640 medium (Biosera, France) containing 2% fetal bovine serum (FBS). Mononuclear cells were then isolated using Ficoll-Hypaque (Biosera, France) gradient centrifugation. The number of viable cells was assessed using the Trypan blue vital staining test (Biosera, France).
Cell staining
To determine the frequency of IL-22-producing subsets, the mononuclear cells were stimulated and the cytokine secretion was blocked simultaneously. To do so, the cells were suspended and incubated in a RPMI 1640 medium (Biosera, UK) containing 50 ng/ml Phorbol myristate acetate (PMA) and 1 μg/ml Ionomycin (Both from Sigma-Aldrich, Germany) in the presence of 0.7 μl Brefeldin A (GolgiPlug), 0.7 μl Monensin (GolgiStop) (Both from BD Bioscience, USA) and 2 μl DNase (1000 Unit/μl) for 5 h at 37 °C in CO2 incubator. To remove the activation medium, the cells were washed twice with staining buffer (1 × PBS + 2% FBS). The cells were fixed with 1% cold Paraformaldehyde for 15 min, and then permeabilized with BD 1 × Perm/Wash TM buffer for 15 min at room temperature and stained with PerCP/Cy5.5–anti-CD4 (clone: RPA-T4), PE – anti-IL-22 (clone: 2G12A41), Alexa Fluor® 647–antiIL17a (clone: SCPL1362) and FITC–anti-IFNγ (clone: 4S.B3). In separate tubes, matched isotype control antibodies were also used (all antibodies from BD Bioscience, USA).
Data acquisition and analysis
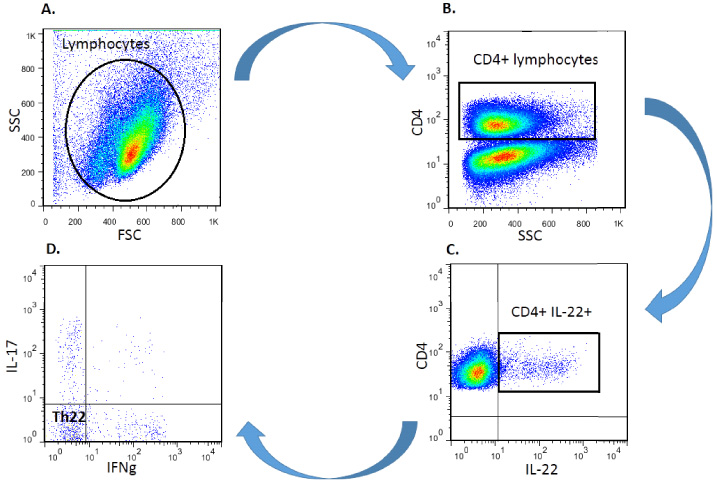
Gating strategy for determining IL-22-expressing CD4+ cells’ frequency in lymph nodes draining from patients with breast cancer. Following the lymphocytes were gated based on morphological parameters on a forward vs. side scatter (A), CD4+ lymphocytes were first gated among lymphocytes (B), CD4+IL-22+ subsets (C) and IFNγ-IL17-IL-22+ CD4+ subsets (D) were then determined in CD4+ lymphocyte gate.
The stained cells were acquired on four-color FACSCalibur flow cytometer (BD Bioscience, USA). More than 200,000 events per each run were obtained during acquisition. The collected raw data were analyzed by FlowJo software version 10. Geometric mean florescent intensity (MFI) was also used as an estimation of IL-22 expression per cell. As illustrated in Fig. 1, to determine the mean percentage of IL-22-expressing CD4+ cells, following separation of lymphocytes population based on their forward and side scatters (Fig. 1A), CD4+ lymphocytes were first gated (Fig. 1B) and then the frequency of CD4+IL-22+ cells (Fig. 1C) and IFNγ-IL17-IL-22+CD4+ lymphocytes (Fig. 1D) were determined and their frequencies were reported in CD4+ gate.
SPSS software package version 16 (SPSS Inc, USA) was used for statistical analyses. The non-parametric Mann–Whitney U test and Kruskal–Wallis H were used to determine statistical differences between two or more different groups, respectively. Correlations between the percentage of IL-22 producing cells, Th22 lymphocytes, age of patients, and tumor size were assessed by non-parametric Spearman correlation rank test. The data are reported as mean ± SEM. P-value less than 0.05 was considered statistically significant. Bonferroni-correction was considered in case of multiple comparisons. GraphPad Prism software package version 6 (Inc; San Diego CA, USA, 2003) was used for drawing the statistical graphs.
Results
Clinicopathological characteristics of the patients with breast cancer
Most patients (22/30, 75.9%) were diagnosed with invasive ductal carcinoma (IDC) as their tumor type (Table 1). Based on the TNM staging (American Joint Committee on cancer classification and stage group; AJCC, seventh edition) patients were classified as stage I (5/30, 16.7%), stage II (19/30, 63.3%), and stage III (6/30, 20%). None of them developed distance metastasis at the time of diagnosis (stage IV). In most cases (22/30, 73.3%), at least, one involved lymph node (LN) was seen (LN+ patients). Clinical and pathological characteristics of patients with breast cancer are summarized in Table 1.
Clinical and pathological characteristics of patients with breast cancer
Clinical and pathological characteristics of patients with breast cancer
As shown in Table 2, the frequency of CD4+IL-22+ cells and Th22 lymphocytes with IFNγ-IL-17-IL-22+CD4+ phenotype in the draining LNs of patients with breast cancer were 2.39 ± 0.39 and 0.64 ± 0.9, respectively.
CD4+ IL-22+ cells and Th22 lymphocytes in tumordraining lymph nodes of patients with breast cancer
CD4+ IL-22+ cells and Th22 lymphocytes in tumordraining lymph nodes of patients with breast cancer
aMean florescent intensity.
Our results demonstrated that the frequency of CD4+IL-22+cells was significantly higher in the patients with stage III (4.25 ± 0.98) compared to those with stage II (1.71 ± 0.45; P = 0.008; Bonferroni corrected P < 0.017, Fig. 2). The frequency of these cells was also observed to be significantly higher in TDLNs of patients with late stage (III; 4.25 ± 0.98) than those in early stages (I/II; 1.93 ± 0.39, P = 0.015). However, the frequency of IFNγ-IL-17-IL-22+CD4+ cells, showed no significant difference among different stages (Fig. 2).
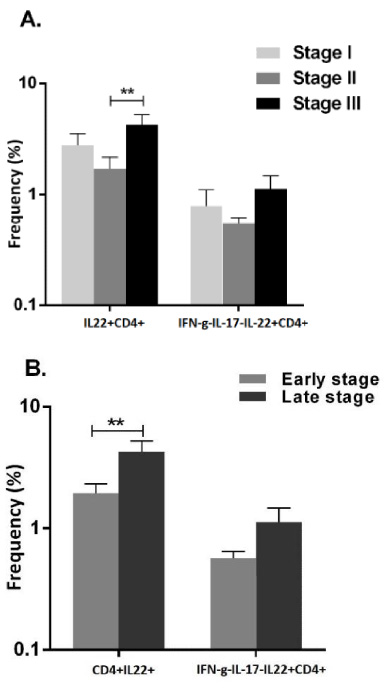
Mean percentage of IL-22-producing CD4+ subsets in different stages of patients with breast cancer. Mean percentage of IL-22+CD4+ T cells was significantly higher in stage III as well as late stage. The data are presented as mean ± SEM. ∗∗ P < 0.017.
More than 70% of patients with breast cancer were reported to have at least one tumor-involved node (introduced as LN+ patients) and about 27% had no metastasis to draining LNs (introduced as LN− patients). Sixteen and five patients were present in N1 (with metastasis to 1–3 axillary lymph nodes) and N2 groups (5–9 involved lymph nodes), respectively. Because of low frequency (n = 1), N3 group (with metastases to more than 9 lymph nodes) was omitted from the analysis. No significant difference was found in the frequencies of IL-22+CD4+ and IFNγ-IL-17-IL-22+CD4+ subsets when LN+ patients were compared to LN−. However, the comparisons demonstrated that the percentage of IL-22+CD4+ lymphocytes had significantly higher frequency in TDLNs of patients in N2 (4.8 ± 0.99) than N1 (1.56 ± 0.5) group (P = 0.004, Bonferroni corrected P < 0.017). The frequency of IFNγ-IL-17-IL-22+CD4+ cells were also significantly increased in TDLNs of N2 patients compared to N1 (1.36 ± 0.44 vs. 0.54 ± 0.08, P = 0.029, however P values did not resist Bonferroni correction (P > 0.017) (Fig. 3).
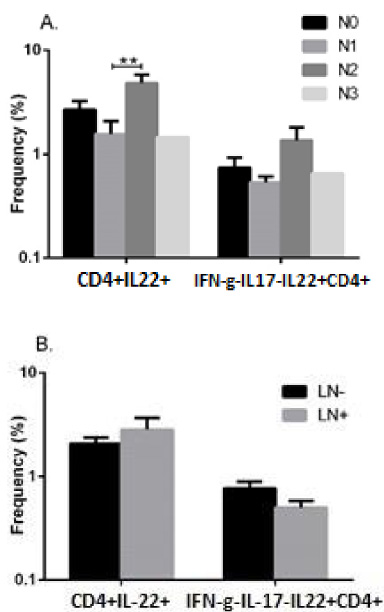
Mean percentage of IL-22-producing CD4+ subsets in TDLNs of patients with breast cancer with different nodal statuses. Mean percentage of IL-22+CD4+ subsets was significantly higher in N2 group than N1 group. However, no significant difference was found when LN+ patients were compared to LN-patients in terms of IL-22+CD4+ and IFNγ-IL17-IL-22+ CD4+ subsets frequency. The data are presented as Means ± SEM. ∗∗ P < 0.01.
No further significant differences were found between the frequency of IL-22-producing CD4+ subsets and mean expression of IL-22 (based on MFI) in these cells in TDLNs of patients with breast cancer with other clinical and pathological characteristics including histological grade, lymphatic invasion, vascular invasion, perineural invasion, peritumoral vessels’ invasion, estrogen receptor, progesterone receptor, and Her-2 expressions (P > 0.05). Furthermore, no significant correlation was observed among IL-22-producing CD4+ subsets and quantitative parameters such as patients’ age and tumor size (data are not shown).
Discussion
In the present study, we investigated the percentage of IL-22-producing CD4+ subsets (CD4+IL-22 and IFN-γ-IL-17-IL22+CD4+) in tumor draining lymph nodes of patients with breast cancer, as the primary site of antigen presentation and immune system activation. Our results indicated that in average more than two percent of CD4+ lymphocytes in tumor draining lymph nodes (TDLNs) of the patients with breast cancer produced IL-22. However, most of them also produced IFN-𝛾 and IL-17, but in smaller amounts. Smaller amount (0.64%) of CD4+IL-22+ cells (2.39%) produced IFN-𝛾 and IL-17. The frequency of IFNγ-IL-17-IL-22+CD4+ cells (CD4+ lymphocytes positive for IL-22 but negative for IFN-γ and IL-17 was about 0.6%. Statistical analysis indicated that the mean percentage of IL-22-producing CD4+ cells showed a significant increase in tumor draining lymph nodes of the patients with higher stage (stage III vs. stage II and late stages vs. early stages. Our results additionally revealed an elevated level of IL-22-producing CD4+ cells in TDLNs of the patients with higher numbers of tumor involved lymph nodes. The mean percentage of IL-22-producing CD4+ was found to be higher in the patients with higher stage and N2 patients in comparison to the ones with early stage as well as N1 patients. However, the difference was not statistically significant. These findings suggest a role for IL-22-producing CD4+ subsets in tumor progression of patients with breast cancer and may provide a new therapeutic approach for breast cancer.
To the best of our knowledge, this is the first study which investigated IL-22-producing cells in TDLNs of patients with breast cancer. A recent study in the patients with triple negative breast cancer showed higher frequency of IL-22+ IL-17− CD4+ T cells in tumor tissues compared to normal tissue and para-tumoral compartments. The study also revealed that IL-22 can promote cells migration and paclitaxel resistance [14]. Another study on breast cancer showed that increased level of IL-22 in serum and over-expression of IL-22 and its receptor in tumor tissues promote the migration and invasion of breast cancer cells through a long non-coding RNA located in HOX gene cluster named HOXB-AS5. These process might be associated with clinical stage and tumor progression [15]. It has been demonstrated that cancer cells could directly induce production of IL-22 and increase the activity of the IL-22 transcription factors in T cells [4,8]. These observations are in line with tumor-promoting properties of IL-22 (enhancing tumor-cell proliferation, protecting from apoptosis, recruiting suppressive immune cells or releasing pro- and anti-inflammatory cytokines). IL-22 also promotes neo-angiogenesis and epithelial-to mesenchymal transition, which are hallmarks of cancer development [16–18]. In vitro analysis in triple negative breast cancer cells showed that IL-22 promotes tumor cell migration and induces paclitaxel resistance by activating JAK-STAT3/MAPKs/AKT signaling pathways [14]. Similar study indicated that IL-22 participates in epithelial cell transformation through MAP3K8 activation [18]. In addition, the presence of IL-22-producing cells was associated with a more aggressive phenotype in a variety of cancer types including lung, gastric, skin and colon cancer, indicating a role for IL-22 in cancer progression [16]. In contrast, a murine model of breast cancer has been shown that IL-22 is able to reduce tumor growth by inhibiting signaling pathways such as ERK1/2 and AKT phosphorylation, situation that was associated with a cell cycle arrest in the G2-M phase [19]. Dual function of IL-22 might be a consequence of the environment in which the tumor formed and the type of cancer studied. However, more investigation is required to determine exact role of IL-22 and IL-22-producing cells in breast cancer.
Taken together, our findings indicated higher frequency of IL-22-producing cells in draining lymph nodes of patients with advanced stages and with more involved nodes. Based on our observation and the observation of others, IL-22-producing cells may play a critical role that IL-22-producing cells may be involved in the progression and invasion of breast cancer. However, further studies with larger sample size and more functional studies are needed to better clarify the role of IL-22-producing cells and Th22 lymphocytes in pathogenesis of patients with breast cancer and other cancers.
Footnotes
Acknowledgements
This study was financially support by Shiraz University of Medical Sciences, Shiraz, Iran (Grant NO: 95-01-01-12890) and Shiraz Institute for Cancer Research (ICR-100-500). The project was submitted as the MD thesis of Ali Salmanpour.
Ethical statement
Ethical approval: All procedures performed in studies involving human participants were in accordance with the Ethics Committee of Shiraz University of Medical Sciences, Shiraz, Iran (IR.sums.med.rec.1396.s205) and informed consent was obtained from each subject before sampling.
Compliance with ethical standards
All procedures involving people comply with the ethical standards of the institutional and/or national committee for research ethics and the 1964 Helsinki Declaration and its subsequent changes or comparable ethical standards.
Informed voluntary consent was obtained from each of the participants.
Conflict of interst
The authors revealed no conflict of interest.
Funding
This study was financially support by Shiraz University of Medical Sciences, Shiraz, Iran (Grant NO: 95-01-01-12890) and Shiraz Institute for Cancer Research (ICR-100-500).
