Abstract
BACKGROUND:
Chronic inflammation is considered to be a risk factor for carcinogenesis, tumor development and metastasis by providing tumor-related factors.
OBJECTIVES:
We aimed to evaluate the effect of cytokine interleukin-1β (IL-1β) as a key mediator of inflammation on multidrug resistance associated protein 2 (MRP2) expression and tamoxifen toxicity in estrogen receptor positive (ER+) MCF-7 breast cancer cells.
METHODS:
The effects of IL-1β on tamoxifen toxicity following 20-day treatment of MCF-7 cells with IL-1β and/or 17β-estradiol (E2) were measured by MTT assay. Furthermore, the effects of IL-1β and/or E2 on the mRNA expression and protein levels of MRP2 and NF-κB (p65) in breast cancer cells were evaluated by QRT-PCR and Western blot analysis, respectively.
RESULTS:
Treatment of breast cancer cells with IL-1β+ E2 decreased the sensitivity to 4-OH tamoxifen compared to both E2-treated and untreated cells. The mRNA expression levels of MRP2 and NF-κB (p65) were significantly increased following treatment with IL-1β+ E2, compared to control. In addition, breast cancer cells treatment with IL-1β+ E2 increased protein expression of MRP2 and it had no significant effect on NF-κB/p65 protein expression in these cells.
CONCLUSION:
Increased expression of mRNA and protein level of MRP2 following 20-day treatment of MCF-7 cells with IL-1β + E2 might be a possible elucidation for the increased tamoxifen resistance which was observed in these cells. More researches are essential to clarify the molecular mechanisms of inflammation on drug-resistance in the tumor environment in order to reducing or eliminating chemotherapy resistance and developing more effective treatment strategies.
Introduction
Chronic inflammation is considered to be a risk factor for carcinogenesis, tumor development, and metastasis by providing tumor-related factors including cytokines, cell survival molecules and other carcinogenesis factors [1]. The cytokine interleukin-1β (IL-1β) is recognized as a main mediator of inflammation and immune response [2]. Although, the beneficial effects of IL-1β in acute inflammation have been reported, the increasing production of IL-1β in chronic inflammation can also promote tumor development and metastasis [3]. It is well documented that there is a relationship between the inflammatory mediators and the risk of drug resistance and metastasis in breast cancer [4,5].
The best therapeutic option for patients with estrogen receptor (ER)-positive breast cancer is tamoxifen (TAM). However, a considerable proportion of patients who primarily respond to tamoxifen develop acquired resistance to antiestrogen therapy [6]. Therefore, understanding the molecular mechanisms of tamoxifen resistance will be useful in developing more effective treatment strategies. The mechanism of tamoxifen resistance is complex and comes from different cellular events. It has been shown that tamoxifen might be a substrate for ATP-binding cassette (ABC) transporters [7]. Multidrug resistance associated protein 2 (MRP2/ABCC2), belonging to the MRP subfamily of ABC transporters, is expressed in cancer tissues and effluxes a wide spectrum of anticancer drugs from cancer cells [8,9]. Although many researches have been done on exploring the role of tamoxifen-metabolizing enzymes in tamoxifen resistance, there are just a few studies on the relationship between the expression of ABC transporters and tamoxifen resistance in breast cancer [10–12]. In breast biopsies, the expression of interleukin-1β in ductal breast cancer was found to be high, while it was undetectable in healthy breast tissue [13]. It has been shown that in some pathological condition including inflammation, IL-1β can modify the expression of ABC transporters [14]. Nuclear factor kappa B (NF-κB) is a main regulator of inflammation. It is one of the downstream mediators of IL-1β signaling pathways and has also been reported to be involved in the expression of ABC transporters. Aberrant NF-κB activity has been found in many types of cancer [15].
In the present study the effects of IL-1β on TAM-cytotoxicity and its effects on mRNA expression and protein level of MRP2 and transcription factor NF-κB, were evaluated in the presence of 17β-estradiol (E2) as a growth stimulator in MCF-7 (ER+) breast cancer cells for the first time. Moreover, the probable correlation between the expression of MRP2, NF-κB/p65 and IL-1β/E2 treatment was assessed.
Materials and methods
Cell culture and reagents
MCF-7 cell line was obtained from Pasteur Institute, Tehran, Iran. Cells were grown in RPMI-1640 containing 10% of fetal bovin serum (FBS) (Gibco, USA) and 1% penicillin (100 units/ml) and streptomycin (100 μg/ml) at 37 °C in a humidified 5% CO2 atmosphere.
IL-1β and E2 were provided from Millipore, USA. 4-Hydroxytamoxifen (4OH-TAM) was purchased from sigma, Germany, and ethanol 100% (v/v) was used to prepare 5 mM 4OH-TAM stock, and stored in −80 °C. MTT ((3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide)) was provided from sigma, Germany.
Viability assay
In order to evaluate the effect of IL-1β on the toxicity of tamoxifen on endocrine-responsive breast cancer cells, MCF-7 cells were exposed to E2 (10 nM) in the presence or absence of IL-1β (1 ng/ml) for 20 days. The media were replaced with fresh ones containing IL-1β and/or E2 every 3 days. At the same time control cells were treated only with culture medium. Following 20-day treatment, cells were seeded in 96-well plates (3.0 × 103 cells/well) for 24 h, and treated with different concentrations of 4OH-TAM (0–15 μM) for 5 days. Then, medium was removed and cells were incubated with 20 μL MTT (5 mg/mL) for 3 h. After discarding the medium, formazan crystals were solubilized with DMSO (200 μl/well). The spectrophotometric absorbance was measured on an ELISA plate reader (Biotek®) with a test wavelength of 570 nm and a reference wavelength of 630 nm, and the percentage of viable cells was determined.
Real-time RT-PCR
The effect of IL-1β and/or E2 on the mRNA expression level of MRP2 and NF-κB /p65 was assessed by quantitative RT-PCR. After extraction of total cellular RNA from MCF-7 cells and the synthesis of complementary DNA (cDNA) with random hexamer primers using a Yekta Tajhiz Azma kit (Iran), qRT-PCR assay was performed in a Real Time PCR instrument using a YTA two-Step SYBR Green qPCR KIT (Iran). Reactions were performed by the denaturation step at 95 °C for 3 min and PCR amplification cycles (40 cycles at 95 °C for 5 s, 60 °C for 1 min). The sequences of primers used in this study were as follows:
MRP2: 5 ′ -ACAGAGGCTGGTGGCAACC-3 ′ (forward) and 5 ′ -ACCATTACCTTGTCACTGTCCATGA-3 ′ (reverse); NF-κB: 5 ′ -CCTTATCAAGTGTCTTCCATCA-3 ′ (forward) and 5 ′ -AATGCCAGTGCCATACAG-3 ′ (reverse); β-actin: 5 ′ -TCATGAAGTGTGACGTGGACATC-3 ′ (forward) and 5 ′ -CAGGAGGAGCAATGATCTTGATCT-3 ′ (reverse). Relative expression levels for MRP2 and NF-κB were normalized to the β-actin as a reference gene and the results were shown as the ratio of target/reference of the treated samples divided by the ratio of target/reference of untreated sample.
Western blot analysis
Cytoplasmic and nuclear protein extracts from treated and control cells were prepared using nuclear protein extraction kit (abcam, USA) as mentioned previously [16]. The concentration of protein in cytoplasmic and nuclear extracts was measured by Bradford reagent. Following the separation of proteins with sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE), the proteins were transferred to polyvinylidene difluoride (PVDF) membranes. Proteins were detected using specific antibodies for MRP2, NF-κB/p65 and β-actin (Cell Signaling Technology, USA) followed by the anti-mouse or anti-rabbit IgG conjugated to peroxidase.
Statistical analysis
Results were shown as mean ± SD from three independent experiments. Statistical analyses were done with one-way analysis of variance (ANOVA) and Tukey–Kramer test using prism version 5 software and P values < 0.05 were considered significant.
Results
The effects of IL-1β on tamoxifen toxicity
Following 20-day incubation of breast cancer cells with IL-1β and or/E2, the cells were treated with various concentrations of 4-OH TAM for 5 days. Tamoxifen exhibited a concentration-dependent inhibitory effect on the growth of MCF-7 cells. As shown in Fig. 1 at concentrations of 0.1 to 15 μM of tamoxifen, mean cell viability was significantly higher in MCF-7 cells treated with IL-1β+ E2 compared to both E2-treated or untreated cells, indicating less sensitivity to tamoxifen in IL-1β+ E2 treated cells.
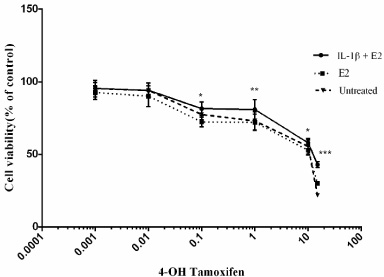
The effects of IL-1β and or/E2 on Tamoxifen toxicity in MCF-7 cells. The cells were exposed to E2 (10 nM) in the presence or absence of IL-1β (1 ng/ml) for 20 days. Then, the cells were incubated with different concentrations of 4-OH TAM (0–15 μM) for 5 days, and cell viability was measured by MTT test. Three independent experiments were performed and data are shown as mean ± S.D. (∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001).
After treatment of MCF-7 cells with IL-1β and or/E2 for 20-day, the expression level of MRP2 and NF-κB/p65 were studied using quantitative RT-PCR. The results showed a significant increase in the fold change of mRNA expression of MRP2 following 20-day IL-1β+ E2 treatment (5.34 ± 0.944), while the mRNA expression of MRP2 in E2-treated cells alone was 2.451 ± 0.2 compared to control cells (Fig. 2A). In addition, a significant increase in NF-κB/p65 mRNA expression was observed following 20-day treatment with IL-1β+ E2 (1.739 ± 0.183) compared to control. However, treating cells with E2 alone did not considerably change NF-κB/p65 mRNA expression (Fig. 2B).
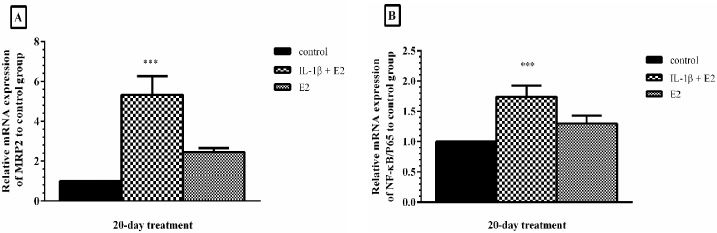
The effects of IL-1β on the mRNA expression of MRP2 (A) and NF-κB/p65 (B). Following treatment of MCF-7 cells with IL-1β and or/E2 for 20-day, mRNA expression of MRP2 and NF-κB/p65was measured by qRT-PCR. Values were normalized to the β-actin as housekeeping gene. Each experiment was repeated three times and the results were shown as mean ± S.D. (∗∗∗P < 0.001).
The protein expression level of MRP2 and NF-κB/p65 were assessed by western blot analysis. As shown in Fig. 3(A) the protein expression of MRP2 in breast cancer cells was up regulated following 20-day IL-1β+ E2 treatment compared to control (P < 0.05), while the protein expression of MRP2 in E2-treated cells alone was not significantly changed compared to control (P > 0.05). In addition, treatment of MCF-7 cells with IL-1β and/or E2 had no significant effect on NF-κB/p65 protein expression in these cells as shown in Fig. 3(B) (P > 0.05).
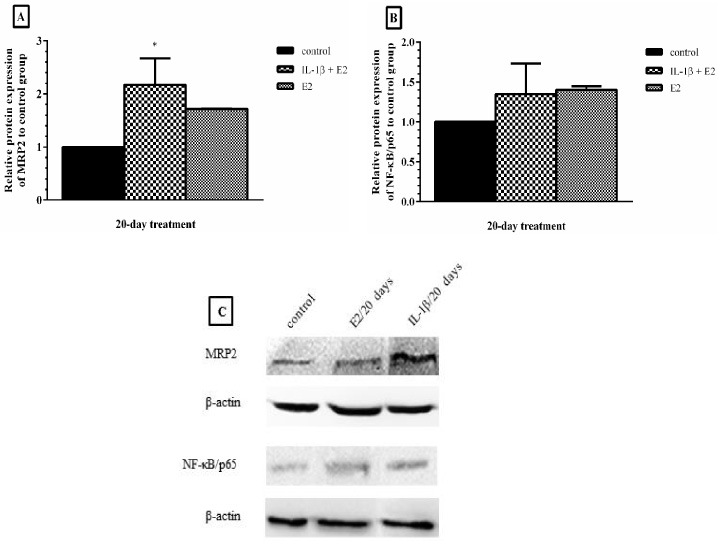
The effects of IL-1β on the protein expression of MRP2 (A) and NF-κB/p65 (B). After treatment of MCF-7 cells with IL-1β and or/E2 for 20-day, MRP2 and NF-κB/p65 protein expression were measured by western blotting and the results were presented as a ratio of the blot densities of MRP2 and NF-κB/p65 to β-actin using Alliance 4.7 Geldoc (UK) (C). Each experiment was repeated three times and the results were shown as mean ± S.D. (∗P < 0.05).
The proinflammatory cytokine IL-1β as one of the important constituents of tumor microenvironment, provides connection between inflammation, carcinogenesis and tumor development [17]. It has been reported that IL-1β may interfere with the expression and function of P-glycoprotein (P-gp) transporter in some in vivo and in vitro models of inflammation [14]. Since the overexpression of ABC transporters is an important mechanism involved in cancer drug resistance and due to the presence of proinflammatory cytokine in the breast tumor environment, we investigated the effects of IL-1β on the expression of MRP2 transporter and its effects on tamoxifen- resistance in ER+ breast cancer cells.
Our results indicated that tamoxifen reduced the proliferation of human breast cancer cell line MCF-7 in a concentration-dependent manner and long term treatment of the cells with IL-1β (1 ng/ml) + E2 decreased the sensitivity to tamoxifen in comparison to both E2-treated and control cells.
Initial studies showed that the maximum changes in ABC transporter expression occurred after 20 days of treatment with the cytokine + E2 and longer treatment times had no additive effect (data not shown). Therefore, in the subsequent experiments, the effects of IL-1β on the expression of MRP2 transporter were examined after 20-day incubation with cytokine and/or E2. Our finding revealed an increased expression of MRP2 mRNA and protein level following 20-day co-treatment of MCF-7 cells with IL-1β and E2. This alteration might provide a clue for the observed simultaneous increase in tamoxifen-resistance in these cells. Tamoxifen is a central component of the treatment for patients with ER+ breast cancers and acquired resistance to it limits the efficacy of chemotherapy in the treatment of cancer [18]. Choi et al. showed that induction of MRP2 may potentiate tamoxifen resistance in breast cancer cells [19]. Furthermore, it has been reported that the elevated expression of MRP2 may act as a predictor of the efficacy of tamoxifen-based chemotherapy in breast tumors [20]. In another study a significant clinical relation between MRP2 expression and lack of sensitivity to oxaliplatin chemotherapy have been shown in colorectal cancer patients [21]. Scientists have reported the presence of functional NF-κB binding site in the promoter of human and mouse MRP2 genes [22]. Activation of transcription factor NF-κB was correlated with a poor response to chemotherapy with doxorubicin in human breast cancer [23]. In another study, NF-κB activation through the PI3K/Akt signaling pathway was proposed as a major mechanism for tamoxifen- resistance in breast cancer cells [24]. Since one of the mediators of IL-1β signaling pathways is the NF-κB factor [25], the mRNA and protein expression level of NF-κB/p65 were assessed following treatment of breast cancer cells with IL-1β+ E2 for 20 days. In this study no changes were observed in the protein expression of NF-κB, suggesting that this cytokine may have used other signaling pathways to induce the expression of the pump, whereas in our previous study, we showed a relationship between the high expression of MRP2, high activity of NF-κB/p65 and increasing tamoxifen resistance following incubation of breast cancer cells with tumor necrosis factor alpha (TNF-α) [16]. Transcriptional activation of inflammatory response-genes is controlled by various specific transcription factors that bind to the target genes promoters and enhancers. In addition to NF-κB, other transcription factors such as C/EBPβ (NF-IL-6), STAT3, AP1 and some nuclear receptors such as PXR have been shown to be implicated in regulation of MDR transporters mediated by different cytokines [26]. According to the result of this study, it can be concluded that the signaling pathways and transcription factors used by IL-1β and TNF-α to induce MRP2 expression are probably different, which should be examined in more details in the future. In addition, more studies are needed to explain the relation between these results and their clinical implication to overcome TAM resistance in breast cancer.
Conclusion
The aim of this study was to mimic the inflammatory conditions in the tumor environment by exposing breast cancer cells to IL-1β with the purpose of studying its effects on tamoxifen toxicity and MRP2 and NF-κB (p65) genes expression. Treatment of breast cancer cells with IL-1β + E2 decreased the sensitivity of MCF-7 breast cancer cells to tamoxifen compared to control cells. The induction of MRP2 mRNA and protein expression following treatment of MCF-7 cells with IL-1β + E2 might be a possible elucidation for the observed increasing tamoxifen resistance in these cells. Further studies are required to clarify the molecular mechanisms of the effects of chronic inflammation on drug-resistance in the tumor environment in order to reducing or eliminating chemotherapy resistance and developing more effective treatment strategies.
Footnotes
Acknowledgements
The authors gratefully acknowledge Research Council in Mashhad University of Medical Sciences, Mashhad, Iran.
Conflict of interest
The authors declare that they have no conflicts of interests.
