Abstract
Sentinel lymph node biopsy (SLNB) has efficiently replaced axillary lymph node dissection (ALND) in axillary staging in node-negative breast cancer patients. Dual sentinel lymph node (SLN) localization using radioisotopes and blue dye is the standard technique for SLN mapping. Yet, nuclear medicine facilities are not widely available worldwide. In Japan, Computed tomography lymphography (CTLG) is presented as an SLN mapping technique which was first suggested in 2003 by Suga et al. Multiple subsequent studies confirmed the efficacy of CTLG in SLN mapping in breast cancer. Further applications of the technique followed; such as prediction of SLN metastasis using CTLG, the use of CTLG guided SLN biopsy after neoadjuvant therapy, video-assisted CTLG guided SLN biopsy, the use of real-time virtual sonography with 3-D CTLG, and preoperative localization of the CTLG mapped SLN using either real-time virtual sonography (RVS) guided Indocyanine green (ICG) injection or its marking using liquid charcoal and silver wire and the use of SPIO enhanced magnetic resonance imaging (MRI) for prediction of metastasis in SLNs detected by CTLG. This efficacy and variable applications open the door for conducting wide-scale randomized controlled trials to suggest using CTLG as an efficient alternative for the use of radioisotopes in SLN mapping in breast cancer patients, especially in low and middle-income countries.
Keywords
Introduction
Axillary lymph node status is the most significant prognostic factor in patients with breast cancer so far. Sentinel lymph node biopsy (SLNB) has efficiently replaced axillary lymph node dissection (ALND) in axillary staging in node-negative breast cancer patients. Since being described for the first time in 1993, SLN mapping & localization tools evoluted all over the years. Now, dual SLN localization using radioisotopes and blue dye is the standard technique for SLN mapping with the best identification rate and the least false-negative rate [1–6].
However, detection of SLN depends, after the localization method, on the surgeon’s experience & proficiency as many cases are required for mastering the technique for intraoperative detection of SLN [7].
Even though combined usage of the dye & isotope techniques can achieve satisfactory detection as well as accuracy rates, the logistical issues related to the usage of radioisotopes are still challenging. Those include the safety precautions for handling & disposal of the isotopes as well as staff training and local authorities’ regulatory issues. Besides, such material is not available easily, especially in developing countries. On the other hand, depending on blue dye alone has some disadvantages such as the relatively lower identification rate, the need for cumulative surgeon experience and the lack of possibility to locate the LN site before skin incision. This led to thinking about the need for developing other alternatives for SLN detection which can offer the same detection & accuracy rates and avoid the drawbacks of the already available methods [2,6,8–10].
In Japan, computed tomography lymphography (CTLG) is presented as an SLN mapping technique which was first suggested in 2003 with promising results [11]. This technique offers an easy and accessible tool for preoperative mapping of SLN with a very good accuracy [12,13]. Herein, we will have an overview of the history, evolution, and current applications of the use of CTLG in SLN detection in breast cancer.
The technique
The patient lies in the supine position with her arms upwards and the hands beside her head. After injection of 2 ml of 1% lidocaine hydrochloride as local anesthesia, 4 mL of undiluted iopamidol-the commercially available CT dye with a well-known very low rate of complications-are injected subcutaneously either subareolar or peritumoral, followed by one minute of gentle massage. Contiguous 2-mm-thick CT images are obtained at 3 minutes after the massage from the upper thorax to the axilla. After detection of the SLN -which is the LN enhanced with the dye-, its location can be mapped on the skin by marking the point of crossing of the vertical & horizontal lines of the red laser light beam of the CT machine. That laser beam is moved according to the SLN site on the CT [12–14].
History and evolution
The story started in 2003 when Suga and his colleagues published their first article about the preliminary results of CTLG guided localization of breast sentinel lymph nodes. The trial was conducted on fourteen female dogs. Five human female volunteers also participated. Iopamidol was used as a contrast material. The results were very encouraging; sentinel nodes could be visualized in all the 14 animals as well as the human volunteers even with low doses like 0.5 ml of Iopamidol. The lymphatic channels could also be visualized and the sentinel node could be surgically resected [11,15] Fig. 1.
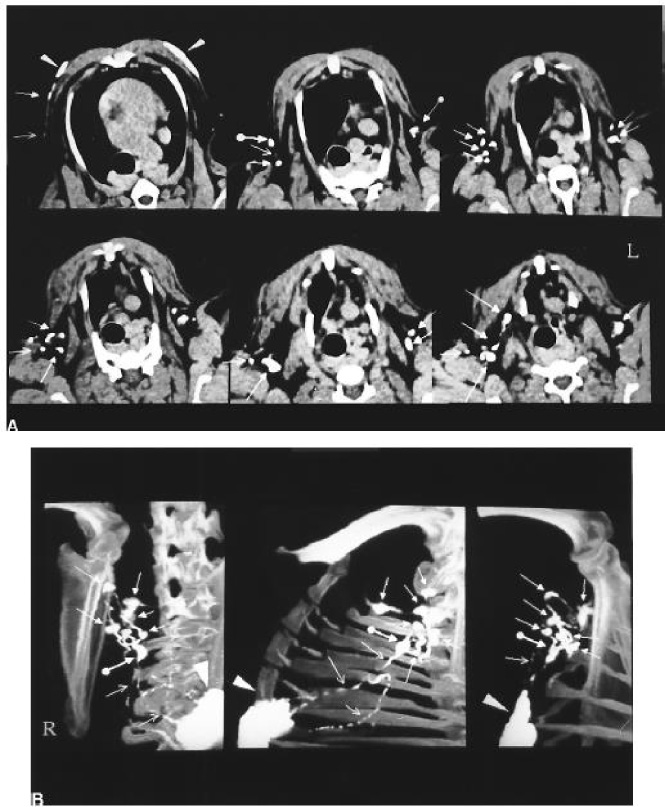
(A) CT images of lymphatic vessels and nodes draining from iopamidol injection sites. The first postcontrast CT images through the upper thorax to the axillary region after a 2-minute gentle massage after injection of 0.5 mL of iopamidol into the 2 skin areas overlying the mammary gland in a normal dog. Along the proximal lymphatic vessels (→) draining from the injection site (arrowhead), the first lymph node encountered (SLN) (∙→), other distant nodes (→), and lymphatic vessels (→) are opacified in the axillary region. (B) Multiple views of three-dimensional (3D) maximum intensity projection (MIP) CT image sets reconstructed from the first postcontrast images display the outlining of the connection between the SLN (∙→) and the proximal lymphatic channels (→) draining directly from the injection sites, and distant lymphatic vessels (→) and nodes (→) in the right axillary region, with the anatomic landmarks of bony structures [11]. Copyright permission obtained.
A few months after, the same team published their results of using this technique to localize sentinel nodes in patients with breast cancer. The study included 17 patients, where CT lymphography was performed using multi-detector CT. After the 3D reconstruction of the post-contrast images, the sentinel lymph node could be visualized and a skin mark was placed on the nearest point to its site. Sentinel lymph node biopsy was performed intraoperatively using blue dye in addition to the CTLG localized node, this was followed by axillary lymph node dissection. The sentinel nodes could be visualized by CTLG in all patients even in those with lymph flow rerouting and those with multiple SLNs. The marked nodes could be easily retrieved intraoperatively [14]. Similar results were published in the same month by another group of researchers from Japan as well [16].
Shortly after, similar results were published including a larger number of patients (40 patients) by Tangoku and his colleagues. They concluded that the CTLG can guide SLN mapping preoperatively in breast cancer patients as well as visualization of the connection between the lymphatics and SLN, and having the possibility of prediction of nodal metastasis [17].
One year later, Suga and his colleagues added further mastery to the technique. They published an article that included 68 patients, using 3 dimensional multi-detector CT images and localizing the lymph nodes by three independent experienced observers. The results confirmed those of the preceding studies on the identification rate and accuracy of the technique. Furthermore, proper visualization of the lymphatics enabled the authors to classify the patients into four categories according to the number of SLNs and the connecting lymphatics (single route/single SLN, multiple routes/multiple SLNs, single route/multiple SLNs, and multiple routes/single SLN) [18]. The concept of categorization of SLNs detected by CTLG into four subtypes was confirmed in the following studies [12,19]. A recent study added further classification of the four groups into subcategories according to the flow direction of lymphatic vessels and their branching pattern [20].
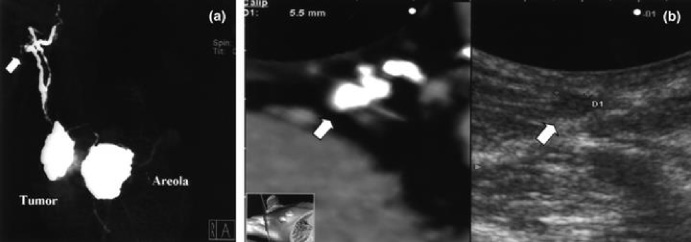
(a) A 3DCT lymphogram. A multiple routes/single SLN pattern in a 54-year-old woman with a 50 × 40-mm tumor in the right upper outer quadrant, where multiple lymph vessels drain into a single common SLN. (b) Real-time virtual sonography. A hypoechoic and globular mass shadow corresponding to an SLN detected by 3DCT lymphography (left side, axial image) in the RVS image. This SLN is positive for breast cancer metastasis. The ultrasound image (right side) corresponds to the RVS image of the SLN [30]. Copyright permission obtained.
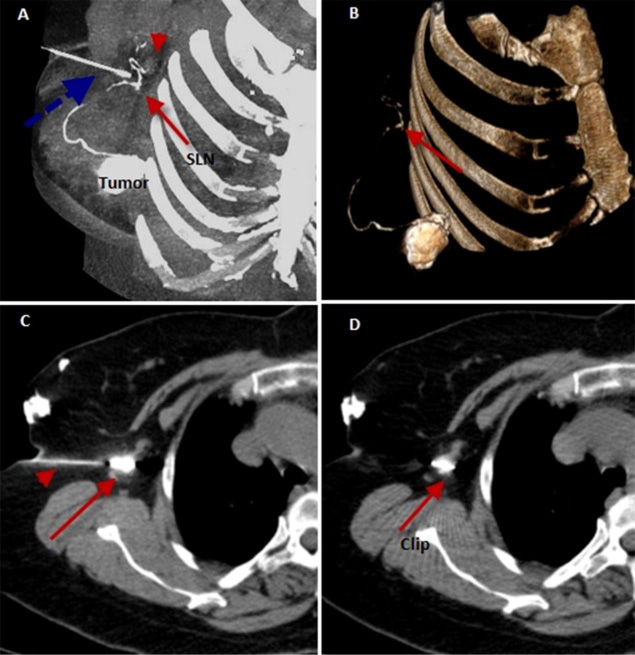
A 3D-CT lymphogram: Reconstructed MIP & SSD images (A&B) show a single afferent lymphatic channel from the sub-areolar region draining into two SLNs (arrows & arrowhead) with a rerouting lymphatic channel in-between. A spinal needle is seen around the SLN with sliver wire at its distal end (dashed arrow). Axial CT image (C) shows a spinal needle (arrowhead) inside SLN with silver wire at its distal end (arrow). The axial CT image (D) shows a silver wire inside the SLN (arrow) [33]. Copyright permission obtained.
Then the wheel turned, further subsequent larger prospective trials were performed including 218, 146, 324 & 184 patients respectively. All of them had similar results confirming the efficacy & reproducibility of the technique. They suggested a combination of CTLG & blue dye as mapping methods for SLNs, offering this combination as an alternative that is suitable for countries lacking nuclear medicine facilities [19,21–23]. The first study published in the English language from a non-Japanese research group was from China on a small number of patients (Thirty-four). It confirmed the same results as well [24]. Another study correlated between poor or non-visualization of the lymphatic vessels tracking to the SLN and the difficulty of intraoperative detection of the blue-dyed SLN. They correlated between the CTLG findings of well-visualized lymphatics and sentinel nodes and easy access to the blue-dyed lymphatics and sentinel nodes [25]. Hashimoto and his colleagues published a case report about the application of the technique in males with breast cancer [26].
In 2015, the twelve-year journey including 576 patients was narrated by Motomura and his group. The reported identification rate was 99%. Only four events of ipsilateral axillary recurrence (1%) were noted in the patients for whom ALND was omitted. They also lightened the utility of using real-time virtual sonography & ICG in conjugation with the same technique [12].
Future
Based on the previous data, the use of CTLG in SLN mapping & localization can be expected to be adopted on a worldwide scale. Furthermore, the application of deep learning & artificial intelligence systems may assist more in the prediction of the pathologic status of the sentinel lymph node. A recently published study evaluated if the deep learning features based on staging CT can predict the number & pathological features of SLNs in breast cancer [34].
Conclusion
CTLG is an easy, safe, and accurate tool for SLN mapping in breast cancer. It offers the possibility of avoiding the drawbacks of the use of radioisotopes. Further randomized controlled clinical trials are required to compare it directly to the usage of isotopes. More accurate marking and retrieval of the SLN can be achieved using ICG, liquid charcoal, and silver wire. SPIO enhanced MRI in conjugation with CTLG may offer the chance to replace SLNB in the future.
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
No funding was received.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Authors’ contributions
OH: conceptualization, data curation, formal analysis, investigation, methodology, writing-original draft. AB: conceptualization, supervision, writing-review. OF: supervision, validation. AS: conceptualization, methodology, project administration, supervision, writing-review, and editing, AD: supervision.
