Abstract
INTRODUCTION:
Breast conservation is the standard of care for early breast cancer. Several oncoplastic approaches have been described for the reconstruction of partial breast defects. The multiplicity of techniques indicates that the ideal reconstructive technique is yet to be identified. There has been a recent emphasis on minimizing the magnitude of surgery. In this report, we describe our experience using the pectoralis major muscle to fill relatively large post-tumorectomy breast defects.
PATIENTS AND TECHNIQUE:
Nine patients were operated on for malignant breast lesions. Tumorectomy resection with surgical margin rendered a 40–50% breast defect. The parenchymal defect was repaired with an advancement muscle flap of the ipsilateral pectoralis major muscle.
RESULTS:
All patients experienced a non-eventful postoperative course. The early cosmetic outcome was excellent in four patients, good in another four, and fair in one patient.
CONCLUSION:
The pectoralis major flap is useful for local repair of upper half partial breast defects in non-ptosed, cup A–B breasts.
Introduction
Breast conservative surgery is the standard of care for early breast cancer patients. Evidence from large multicenter randomized trials has shown breast conservative surgery to be non-inferior to total mastectomy [1]. Oncoplastic breast surgery consists of adequate tumorectomy followed by esthetic repair of the partial mastectomy defect to achieve a satisfactory cosmetic outcome. Oncoplastic surgery is recommended for all breast conservative operations. Level-I oncoplastic surgery is used to repair minor losses of breast volume. At this level, reconstruction is limited to adequate mobilization and re-approximation of the breast parenchyma to fill the tumorectomy defect [2,3]. Larger volume loss requires level-II oncoplasty. In level-II procedures, extra-mammary tissue is mobilized to fill the breast defect (volume replacement). Alternatively, a bilateral breast reduction procedure (volume displacement) is used to obliterate the tumorectomy defect and correct any associated breast hypertrophy or ptosis [4]. With either strategy, volume replacement and volume displacement require extensive skin incisions and tissue mobilization. Thus, level-II oncoplasty achieves maximum esthetic outcome at the expense of major surgical trauma and a significant probability of operative morbidity. With the exception of level-II oncoplastic procedures, breast cancer surgery is a well-tolerated intervention that can be offered on a fast-track or day-case basis [5–7]. Recent years have witnessed a renewed interest in de-escalating oncoplastic surgical strategies. Level-I oncoplastic operations are being used more frequently to fulfill the cosmetic needs of the majority of breast conservative operations [8]. Moreover, the acceptability of a complex level-II oncoplastic procedure is variable from one patient to another [9]. Because of the above-mentioned considerations, These authors and others have worked to expand the role of level-I oncoplastic surgery. Several innovative techniques have been recommended to allow repair of wider post-tumorectomy defects with simple mobilization of breast parenchymal flaps [10–12].
The pectoralis major muscle flap is the classic workhorse of the head and neck reconstructive surgery. The muscle has been extensively used as a pedicled or free flap to reconstruct major post-ablative tissue defects [13–17]. In esthetic breast surgery, pectoralis major muscle has been used as a sling to correct breast ptosis [18].
We speculated that mobilization of glandular tissue based on the underlying pectoralis major fibers will allow repair of larger defects without the need for sophisticated level-II procedures.
Patients and technique
Participants
This report includes nine patients with malignant breast lesions. They were scheduled for conservative breast surgery based on their tumor characteristics and after a thorough discussion with the patient and the tumor board.
All patients had tumors in the upper half of their breasts. Pre-operative assessment predicted a post-tumorectomy defect of 40–50% of breast volume. The sizeable anticipated loss was due to large phyllodes mass in three cases, ill-defined borders of the tumor consequent to neoadjuvant therapy in three other cases, and a tumor-to-breast discrepancy in the remaining patients. Table 1 summarizes their most relevant preoperative information.
Showing the preoperative information of the nine patients included in this study. The patients’ age, tumor location in the breast, type of breast lesion, and breast cup size are presented. In case of a large tumor distorting the breast, the cup size of the normal side is reported. The cosmetic outcome was evaluated at three weeks using a four-point scale
Showing the preoperative information of the nine patients included in this study. The patients’ age, tumor location in the breast, type of breast lesion, and breast cup size are presented. In case of a large tumor distorting the breast, the cup size of the normal side is reported. The cosmetic outcome was evaluated at three weeks using a four-point scale
“PNA: post-neoadjuvant, IDC: infiltrating duct carcinoma, PT: phyllodes tumor, UOQ: upper outer quadrant, UIQ: upper inner quadrant, C: central tumor.”
All nine patients have provided written, informed consent for the whole management plan including the proposed reconstructive option and the available alternatives. The study has been ethically approved by the Institutional Research Board of Faculty of Medicine at Mansoura University (Protocol number: R/19.08.578). The cases reported herein constitute a part of an ongoing prospective study (clinicaltrials.gov identifier: NCT04091958).
Operative technique
Pre-operative marking of the skin incision was performed on the day of surgery. In four patients, we used a classic or a modified omega-incision (Fig. 1–3). The classic omega incision is a transverse equatorial breast incision that curves around the superior areolar margin. The modified technique included a peri-areolar dermal flap. The de-epithelialized dermal flap allowed proper re-centralization of the nipple/areola complex (NAC). A radial skin incision was used in three patients and a circum-areolar incision in another two. Resection was carried down to the plane of the pectoral fascia. The resected specimen included a three-dimensional surgical margin of grossly normal tissues. Axillary management included full dissection in three patients and sentinel lymph node biopsy in another three patients. Cases of phyllodes tumor did not require axillary surgery. Resection margins were tested for adequacy by frozen-section examination.
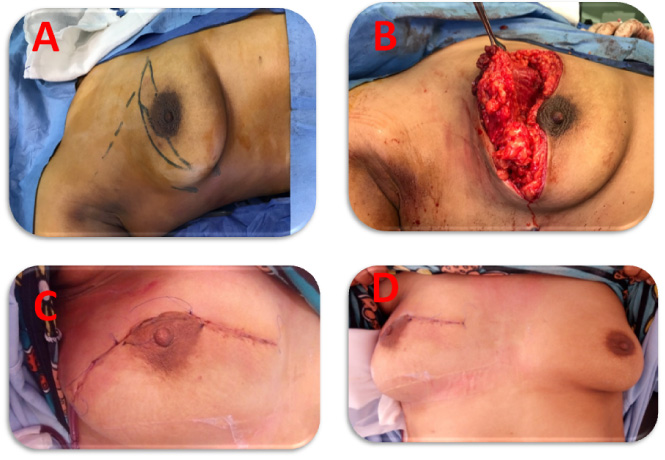
Case 2. Multifocal right breast carcinoma resected through a batwing incision.
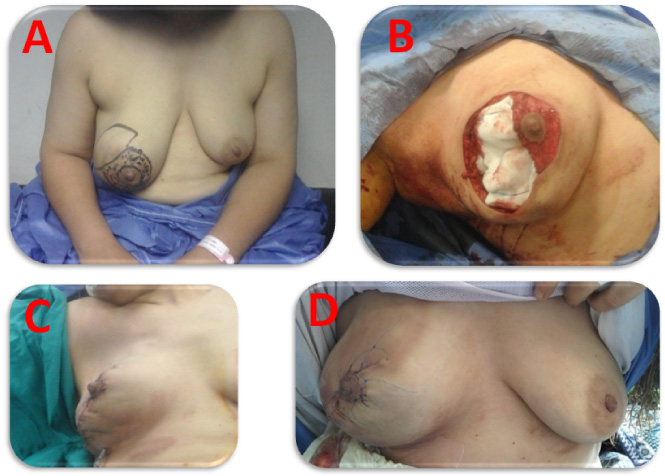
Case 3. Right malignant phylloides tumor resected through a modified batwing incision.
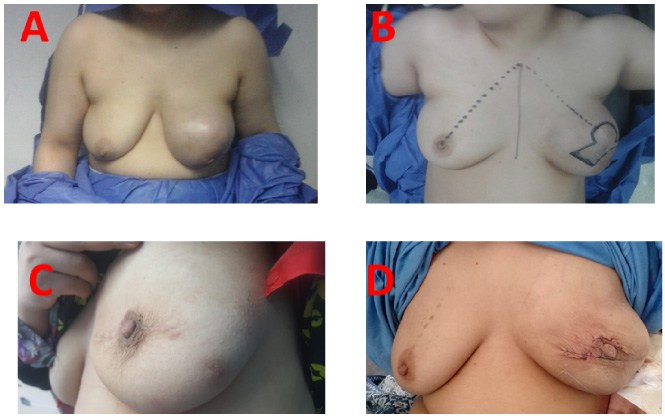
Case 4. Left malignant phylloides tumor resected through a batwing incision.
The reconstructive technique started with the routine mobilization of full-thickness skin flaps. Skin mobilization avoids postoperative skin retraction and maximizes the esthetic outcome. Contrary to the conventional techniques, we did not mobilize the glandular parenchyma off the pectoral fascia. Instead, a composite flap of the pectoral muscle and breast parenchyma was recruited to the defect using three sequential steps. First, the sternal head of the pectoralis major was dis-inserted from its humeral attachment. Second, the detached muscle was dissected free off the chest wall in the infra-clavicular region. Third, fibers connecting the muscle to the upper sternum were detached. The last step was routinely adopted after the first few cases in order to inset the flap at the recipient bed without tension. This particular maneuver leads to transaction of the perforator from the internal thoracic artery at the second intercostal space; which is a sizeable vessel supplying the breast.
Adequate hemostasis, closed-suction drainage, and closure of the skin in two layers were performed in all cases.
Postoperative assessment of the cosmetic outcome was carried out three weeks after surgery. Two observers used the four-point scale to score the results as excellent, good, fair, or poor as described [19,20].
Results
Nine patients had conservative breast surgery as a part of the management of malignant breast lesions. The extirpative surgery resulted in 40–50% breast volume loss. The breast volume loss was reconstructed using the ipsilateral pectoralis major muscle as a composite myo-parenchymal flap. The postoperative course was uneventful. The average tumor size was 6.3 cm (range 2–14) and the resected surgical specimen ranged from 88 to 1224 cm3 (median volume 600). Early postoperative results were subjectively satisfactory to the nine patients. Objective scoring by independent staff was excellent in four patients, good in four, and fair in one. The long-term outcome is not available at the time of writing. Table 2 summarizes the operative outcome of the study.
Showing the technical aspects of the surgery in the nine patients included in this report. Resection volume was derived from the final pathology report and was calculated using the ellipsoid volume formula. The cosmetic outcome was evaluated at three weeks using a four-point scale
Showing the technical aspects of the surgery in the nine patients included in this report. Resection volume was derived from the final pathology report and was calculated using the ellipsoid volume formula. The cosmetic outcome was evaluated at three weeks using a four-point scale
“UOQ: upper outer quadrant, UIQ: upper inner quadrant, C: central tumor.”
In this report, we present a potentially useful technical solution for sizeable partial mastectomy defects. Post-resectional volume loss of ≈45% of the total breast mass was encountered and satisfactory reconstruction was achieved using local flaps. Surgical manipulation was restricted to the ipsilateral breast and the underlying muscle bed. No extra-mammary donor site was involved. The proposed technique is a well-tolerated level-I oncoplasty.
The upper inner quadrant and the peripheral area (zone C) of the superior quadrant are notoriously unforgiving breast locations. Tumorectomy in these areas is particularly prone to unfavorable cosmetic outcomes because the breast parenchyma is relatively thin and the scar is potentially uncovered with the average dress. Besides tumor location, the discrepant tumor-to-breast volume is another challenge to the breast surgeon. Large resection volume is particularly anticipated after primary systemic therapy and with the presence of a phyllodes tumor. In this report, we managed the resulting significant defect using standard tumorectomy incisions. This technique was particularly useful in cup A–B breasts affected by phyllodes tumors and free of ptosis.
Batwing (omega) mastopexy is a popular oncoplastic technique [21]. The procedure involves a transverse incision at the breast equator. Generous resection is feasible and correction of breast ptosis is achieved. The resulting NAC upward shift is the downside of the technique. We combined batwing incision with a generous peri-areolar dermal flap to allow NAC re-centralization and consequently larger resection volume was permissible.
The upper part of the pectoralis major was used as an advancement (early two cases) flap or as a turn-over flap based on the internal thoracic perforator as previously described for chest wall reconstruction [22]. The described flap was based on the perforating branches of the internal thoracic artery. The superior pedicle from the acromio-thoracic artery was divided to allow muscle mobilization. Consequently, the muscle became denervated and the final tissue volume is potentially reduced. In two patients, adequate muscle mobilization was achieved with an intact innervation and superior pedicle.
The dual blood supply of the pectoralis major is consistently reliable and allows total or partial muscle flap elevation without consequences to the rest of the muscle [23–27]. The loss of the muscle function is well-tolerated because the clavicular head of the muscle as well as the pectoralis minor muscle is left intact.
Evaluation of the described technique requires a large cohort of patients and long-term outcome data. Final cosmetic results are evaluable several weeks after the last irradiation dose. The technique is not suitable for ptosed or large-cupped breasts and is best suited for breasts affected by phyllodes tumor. We suggest that using the pectoralis major myo-parenchymal flap is a useful option for breast surgeons facing a relatively large partial mastectomy defect.
Footnotes
Acknowledgements
The authors thank their patients for accepting the service and giving us the chance to serve and improve. The units of surgical oncology, medical oncology, nursing, radiology, and pathology strive to provide the best care for the patients of this study and all patients treated at Mansoura University Oncology Center.
Conflict of interest
None.
Funding
None.
Authors contribution
The first author conceived the technique and wrote the manuscript. All authors contributed to patients’ management and discussed and approved the manuscript.
