Abstract
Breast cancer is one of the leading cancers nowadays. The genetical mechanism behind breast cancer development is an intricate one. In this review, the genetical background of breast cancer, particularly BRCA 1 and BRCA 2 had been included. Moreover, to summarize the genetics of breast cancer, the recent and ongoing preclinical and clinical studies on the treatment of BRCA-associated breast cancer had also been included. A prime knowledge is that the BRCA gene is the basis of breast cancer risk. How it mediates cell proliferation and associated mechanisms are reviewed here. BRCA 1 gene can influence all phases of the cell cycle and regulate cell cycle progression. BRCA 1 gene can also respond to DNA damages and induce responsive mechanisms. The action of the BRCA gene on associated protein has a wide consideration in breast cancer development. Heterogeneity in breast cancer makes them a fascinating and challenging stream to diagnose and treat. Several clinical therapies are available for breast cancer treatments. Chemotherapy, endocrine therapy, radiation therapy and immunotherapy are the milestones in the cancer treatments. Ral binding protein 1 is a promising target for breast cancer treatment and the platinum-based chemotherapies are the other remarkable fields. In immunotherapy, the usage of anti-programmed death (PD)-1 antibody is a new class of cancer immunotherapy that hinders immune effecter inhibition and potentially expanding preexisting anticancer immune responses. Breast cancer genetics and treatment strategies are crucial in escalating survival rates.
Introduction
Breast cancer is the most frequent cancer among women, affecting 2.1 million women each year. In 2018, it is estimated that 627,000 women died by breast cancer - that is approximately 15% of all cancer deaths among women. The breast cancer rates are higher among women in more developed regions and it is increasing alarmingly in nearly every region globally (WHO, 2018) [1]. Breast cancer is the second most common cancer worldwide after lung cancer [2]. There are several factors associated with breast cancer risk. Age, hormonal - reproductive - menstrual history, alcohol, radiation and hereditary factors are the major ones. Some of the observation related to breast cancer risk is that it is increased in early menarche, late menopause and obesity in postmenopausal women. Both oral contraceptives and hormonal therapy for menopause may increase breast cancer risk. The preventive measures include childbearing, which reduces the risk with early first birth, a larger number of births and breastfeeding are the effective ways [3]. Several studies had been conducted all over the world for controlling breast cancer risks. But the molecular basis of malignancy remains unknown. Although the general concern is that early diagnosis is the only way to decrease the death rate or prevention of malignancy. So here an attempt had been initiated to analyze genetics and treatment strategies for breast cancer.
Genetics of breast cancer
A prime knowledge of breast cancer genetics is essential for a detailed investigation. The BRCA (BReast CAncer) gene is a tumor suppressor gene that prevents uncontrolled cell division and helps in DNA breaks repair. The mutation in the BRCA gene produces BRCA1 and BRCA 2 which predisposes human breast cancer. BRCA1 and BRCA2 encode very large proteins widely expressed in different tissues during the S and G2 phases, which are localized in the cell nucleus. 55–65% of women with the BRCA1 mutation will develop breast cancer before the age of 70 and 45% of women with a BRCA2 mutation will develop breast cancer by the age of 70. Cancers related to BRCA1 mutations are more likely to be triple-negative breast cancer, which can be more aggressive and difficult to treat [4,5]. The detailed structural domains of BRCA1 and BRCA2 are analyzed (Fig. 1). Venkitaraman [5] proposed the biological functions of BRCA genes and suggested that its disruption can induce susceptibility to specific types of cancer. BRCA 1 gene is located on the q arm of chromosome 17 at position 21. BRCA 2 is on the q arm of chromosome 13 at position 12.3. BRCA 1 and BRCA 2 possess several biological roles and they can act as a caretaker of chromosome structures ie., they can control the gross chromosomal rearrangements including translocations, deletions and fusions of the non-homologous chromosomes. BRCA 2 has a main role in double strand break repair, particularly it can control RAD51 recombinase, a eukaryotic homolog of bacterial Rec A essential for double-strand break repair. BRCA 1 deficiency can cause DNA damage and thereby block cell proliferation and apoptosis [6].
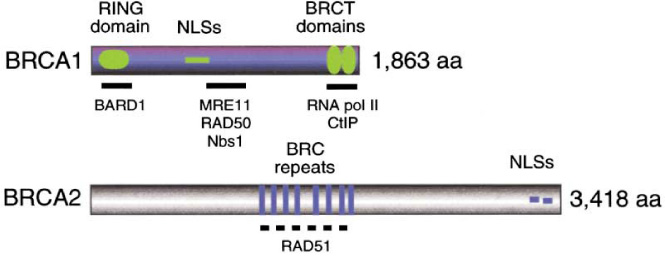
BRCA1 contains an N-terminal RING domain, nuclear localization signals (NLSs), and two C-terminal BRCT domains of 110 residues. BRCA 2 contains eight repeats of the 40 residue BRC motifs. Six of the eight motifs in human BRCA 2 can bind directly to RAD51 when expressed in vitro [5].
BRCA1 can influence in all phases of the cell cycle and regulate cell cycle progression. BRCA 1 deficiency consequently causes abnormalities in the S-phase checkpoint, G2/M checkpoint, spindle checkpoint and centrosome duplication.
Cell cycle arrests in the G1, S, and G2 phases that occur in mammalian cells after ionizing irradiation is essential to maintain normal cell homeostasis, otherwise uncontrolled cell growth and cell transformation. BRCA 1 plays an important role in cell cycle regulation. Each phase of the cell cycle is under the control of it. The G1 phase arrest occurred by the interaction of BRCA1 with hypophosphorylated RB (retinoblastoma) protein. RB is a phosphoprotein and well-known tumor suppressor. The hypophosphorylated RB interacts with E2F (transcription factor) to prevent transcription of downstream genes and inhibits cell proliferation. BRCA1 keeps RB in the hypophosphorylated state to achieve growth arrest. BRCA 1 C-terminal domains (BRCT) also form a complex with two RB-binding proteins, RbAp46 and RbAp48, and histone deacetylases 1 and 2 (HDAC1 and HDAC2) [7]. The RB and histone deacetylase complex (HDC) is thought to be suppressing transcription of E2F- responsive genes, providing additional evidence for growth inhibition through RB [8]. In the case of the G1/S phase, p53 is associated with the BRCA1 induced G1/S cell cycle checkpoint. BRCA1 can up-regulate p21 expression and prevented cell cycle progression into S phase [6].
The ataxia-telangiectasia-mutated (ATM) gene product controls overall BRCA1 phosphorylation in response to 𝛾-irradiation (IR). Tibbettes et al. [9] proved that BRCA1 phosphorylation is partially ATM-dependent in response to IR and ATM independent in response to treatment with UV light. In UV or IR radiation-induced DNA damages and the phosphorylation of p53 is mediated by BRCA1–BARD1 complex that is associated with ATM/ATR (ataxia-telangiectasia-mutated and Rad3-related). Among the IR and UV radiation-induced p53 (Ser-15) phosphorylation, only IR radiation could induce G1/S arrest, while UV radiation did not [10]. The ATM mediates IR-induced phosphorylation, while ATR mediates rapid UV induced phosphorylation. This observation suggests that the BRCA1–BARD1 complex is mediated by ATM and catalyzed by p53 (Ser-15) phosphorylation [11]. The IR radiation- induced G1/S arrest is mediated by p21 and p53 (Fig. 2) [12].
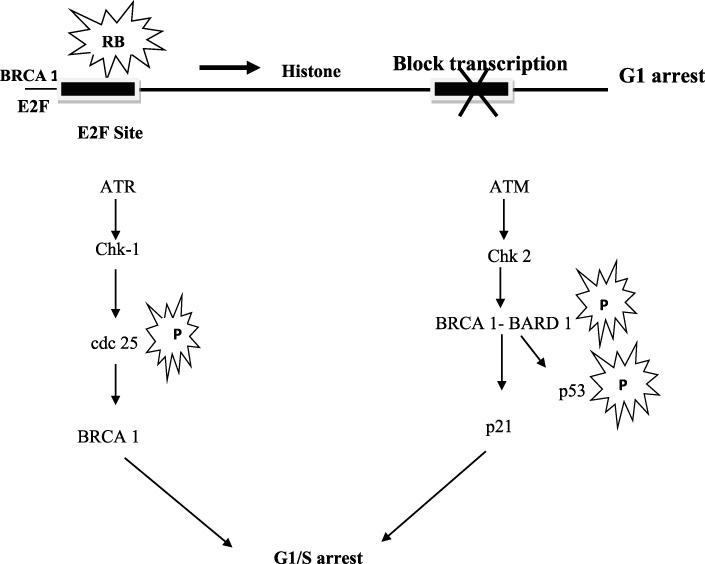
BRCA 1 mediated cell cycle arrest at G1 and G1- S phase.
Deng [6] states that BRCA1 deficiency can cause abnormalities in S-phase checkpoint, G2/M checkpoint, spindle checkpoint and centrosome duplication. The genetic instability caused by BRCA1 deficiency can interrupt the cellular responses to DNA damage and thereby blocks cell proliferation and induces apoptosis. The role of BRCA1 in cell cycle checkpoints, genome integrity, DNA damage response (DDR) and tumor evolution should be thoroughly assessed for understanding the mechanisms of BRCA1 associated tumorigenesis, as well as opening a gateway for the search of new therapeutic approaches for this lethal disease.
Hepatocellular carcinoma associated with S-phase cell cycle arrest was analyzed by Chan et al. [13]. S-phase arrest and apoptosis occur in HCC, BEL-7402 cells by the down-regulation of cyclin D1 and cdc2. Cyclins and breast cancer risks are closely associated. The mammary carcinoma induced by the neu and ras oncogenes is resistant to Cyclin D1 null mice, which indicates the essential role of Cyclin D1 in the development of some mammary carcinomas. The overexpression of Cyclin D1 and E1 are commonly seen in primary breast cancer. The functional upshot of Cyclin E overexpression in breast cancer cells is that it primarily affects cell cycle progression, whereas Cyclin D1 is targeted to transcriptional regulation [14]. The quercetin treated MCF-7 cell lines show S phase arrest by decreasing the protein expression of CDK2, Cyclins A and B while increasing the p53 and p57 proteins. The apoptotic cell death induced in MCF-7 cell lines are with decreased levels of Bcl-2 protein and increased activations of caspase-6, -8 and -9. The AIF (Apoptosis-Inducing Factor) protein released from mitochondria to nuclei and the translocation of GADD153 protein from the endoplasmic reticulum to the nuclei also occurred [15]. BRCA1 participates in cellular responses to ionizing radiation (IR) and interacts with ATM and can induce S phase and G2/M phase arrest [16]. Chen and Wong [17] state that S-phase arrest associated with breast cancer delimits the protein expression of Cyclins A, D1, D3, cyclin-dependent kinases (CDKs) 4 and 6, with concomitant induction of p21waf1/Cip1, p27Kip1 and p53. Checkpoint kinases Chk1 and Chk2 are two key components in the DNA damage checkpoint signalling cascade. The DNA damage-induced S-phase cell cycle arrest is contributed by Chk1 but G2 phase arrest is independent of Chk1 and Chk2 [18].
G2-M phase cell cycle arrest also reveals the down-regulation of Cyclin A and Cyclin B, increasing p21waf1/cip1 and P27kip1 levels and up-regulated Chk2, which leads to the increase in Cdc25C phosphorylation, inactivation of Cdc2 levels and an increase in Wee1 level [19]. In human breast cancer, MCF-7 and MDA-MB-453 cells treated with Daidzein causes cell cycle arrest at the G1 and G2/M phases. Daidzein is an isoflavone that decreases Cyclin D, CDK2, and CDK4 level, whereas the expression of CDK6 and Cyclin E was kept unchanged. The protein expression of CDK1 related to the G2/M phase decreases markedly. The expression of CDK inhibitors p21Cip1 and p57Kip2 are increased, but not that of p27Kip1 [20].
The centrosome is the architectural framework that maintains the cytoplasmic network of interphase and mitotic cells through microtubule organization. They play a vital role in cell cycle regulation [21]. The centrosome is duplicated once in a cell cycle and its regulation is coupled with cell cycle progression. Cdk/Cyclin A and E are the key regulators of centrosome duplication. The P53 mediated regulation of G1/S and G2/M phases also triggers the regulation of centrosome duplication. Some other factors behind the regulations are centriole protein, centrosome-directed kinases and phosphatases [22]. Centrosome amplification could drive tumor aneuploidy by increasing the frequency of abnormal mitoses that lead to chromosome aggregation and development of tumors [23].
Heterogeneity in breast cancer makes them a fascinating and challenging stream to diagnose and treat. Women with a BRCA1 or BRCA2 mutation are candidates for some additional risk factors. Some additional susceptibility genes have identified, including PTEN, ATM, TP53, CHEK2, CASP8, PBRL and BRIP1 [24]. The mutations of BRCA1/BRCA2 genes in breast and ovarian cancer families are mainly point mutations or small insertions and deletions. It is noteworthy that large rearrangements in the BRCA1 and BRCA2 gene accounted for less than 1% of evaluated patients suffering from hereditary breast and ovarian cancer [25]. How do BRCA1 and BRCA2 mutations cause cell proliferation? They are the key factors involved in DNA damage repair and transcriptional repair. So mutation in any of the intermediate can induce rapid cell proliferation. In addition to BRCA1 and BRCA2 mutation, several other subtypes of breast cancers are recognized, that is associated with BRCA interacting proteins (Table 1).
BRCA1 interacting proteins [5]
The elevated DNA damages are always associated with breast cancer risk as compared to the other types of cancers [26]. The role of BRCA1 and BRCA2 in the homologous recombination repair (HRR) of double-strand breaks in DNA and their ability in the suppression of breast cancer are crucial. The genetic variations associated with BRCA associated proteins are susceptible to breast cancer risk. The family of proteins RAD51 and XRCC2 are essential components of the DNA damage repair pathway. The site-directed mutagenesis of XRCC2 causes substitution or deletion of the amino acid 188 of the XRCC2 gene and it will lead to the loss of DNA damage sensitivity. It is evident that R188H polymorphism is susceptible to breast cancer risk. The variation in DNA repair capacity may influence cancer susceptibility in a population [27]. XRCC1 R399Q polymorphism also shows the risk of breast cancer in the hereditary form. In the case of dominant alleles of RAD51, TP53 and XRCC1, their combined genotypic action indicated a strong protective role against hereditary breast cancer [28]. Several reports are related to the polymorphism of DNA damage repair gene XRCC1 and its contribution to breast cancer risk [29,30].
The genetic variation in the DNA repair gene contributes to deficient DNA repair mechanisms and the risk of cancer. The three main DNA repair genes are XRCC1 Arg194Trp and XRCC1 Arg399Gln in base excision repair, XRCC3 Thr241Met in homologous recombination repair and ERCC4/XPF Arg415Gln in nucleotide excision repair. The clinical and case-based studies of Smith et al. [31] shows that variants of XRCC1, XRCC3 and ERCC4/XPF genes, particularly in combination, contribute to breast cancer susceptibility. Alli et al. [32] found that defective base-pair excision repair found in BRCA1 mutated breast cancer cells conferred sensitivity to inhibition of poly (ADP-ribose) polymerase, a DNA repair enzyme. BRCA 1 can repair DNA damages and promoting assembly of Rad5 [33].
The autosomal-recessive disorder ataxia-telangiectasia (AT) is conferring the risk of breast cancer. ATM is an intermediate breast cancer susceptibility gene. The loss of ATM function causes defects in DNA repair, cell cycle checkpoint control and is prone to cancers. BRCA1 is regulated by an ATM-dependent mechanism as a part of the cellular response to DNA damage [34]. Tavtigian et al. [35] state that the combinations of ATM truncating and splice junction variants are the main reason that leads to the development of breast cancer. The missense mutation in ATM causes breast cancer. The mutation in the p53 gene becomes a common factor in the majority of human cancers. The structural rearrangement of the DNA binding domain of the protein is damaged and leads to cancer [36].
PI3K/AKT/mTOR pathway aberrations are the common abnormalities associated with breast cancer risk. Phosphoinositide 3-kinases (PI3Ks) are a family of enzymes involved in cell growth, proliferation, differentiation and intracellular trafficking. It is a heterodimer composed of regulatory (p85) and catalytic (p110) subunits. The stimulation of receptor tyrosine kinases activating the signalling cascade through PI3K activation is followed by phosphorylation of AKT and mTOR complex 1 (mTORC1). In TNBC (Triple Negative Breast Cancer), oncogenic activation of the PI3K/AKT/mTOR pathway resulted as a function of overexpression of upstream regulators (e.g., epidermal growth factor receptor [EGFR]) and the down regulators. It also includes the mutations of PI3K catalytic subunit α (PIK3CA), loss of function or expression of phosphatase, tensin homolog (PTEN), and the proline-rich inositol polyphosphatase [37]. PI3K/AKT/mTOR pathway had become an essential tool in cancer therapy. The inhibition of the mTOR pathway in patients with cancer became more feasible after the development of Rapamycin analogs with improved pharmacologic properties [38]. The cell apoptosis induced in TNBC cells by the combined action of Gefitinib with SF1126 is mediated through the EGFR-PI3K-AKT-mTOR-p70S6K pathway [39]. In ER-positive breast cancer, PI3K/AKT/mTOR pathways had become an effective target for endocrine therapy [40].
In breast cancer clinical therapy, homologous recombination (HR) DNA repair is a relevant technique. There are three DNA based homologous recombination deficiency (HRD) scores [HRD-loss of heterozygosity score (LOH), HRD-telomeric allelic imbalance score (TAI), and HRD-large-scale state transition score (LST)] [41]. It can connect with defects in BRCA1/2, and are associated with response to platinum therapy in triple-negative breast and ovarian cancer. There is a direct relationship between the frequency of BRCA1/2 defects among different breast cancer subtypes and the HRD scores. It helps to identify breast tumors with defects in the homologous recombination DNA repair pathway. Some studies had pointed out that single nucleotide polymorphisms (SNPs) in homologous recombination repair genes can cause the incidence of breast cancer risk [42]. The high DNA damage response is connected with a low incidence of tumor development. DNA damage repair proteins are key factors for the prediction of breast cancer sensitivity to neoadjuvant chemotherapy [43].
Extensive studies are conducted on the emergence of breast cancer relating to DNA damages. These studies revealed that BRCA1 has a role in double-strand break repair mainly through HR mechanism, and initiates the response to DNA damage and thereby maintain genomic integrity. In sporadic basal-like breast cancer, the genetic instability will be collapsed by the complex mechanism of BRCA1 inactivation and other DNA repair genes that lead to the progression of tumors. In that case non-homologous end-joining (NHEJ) mechanisms take part in the DNA damage repair. Ku70/Ku80 heterodimer is the key protein involved in these repair systems. Ku70 is a protein encoded by the XRCC6 gene and Ku80 is encoded by the CRCC5 gene. Ku protein plays an important role in cell signalling, proliferation, replication, transcriptional activation and apoptosis in HR-deficient tumor [44]. In mismatch repair, enzymes MSH2 and MSH6 are proved to be the breast cancer carriers. Westenend et al. (2005) [45] confirmed the absence of MSH2 and MSH6 in tumor cells through the immunohistochemical staining method. They strongly suggested that the MSH2 gene has a key role in tumor cell development. MSH6 and PMS2 are pathogenic variants associated with LS, which can induce breast cancer [46].
Molecular subtype of breast cancer [47,48]
Estrogen and breast cancer
During puberty, estrogen is responsible for maturation of the mammary gland by mediating ductal elongation. Fillmore et al. [49] suggest that estrogen can induce the secretion of paracrine acting proteins, which in turn increase percentages of CD44+/CD24−/ESA+ populations and corresponding cancer stem-like cell properties in many breast cancer cell lines. Estrogen can upregulate FGF9/FGFR3 signalling pathways and it can also increase the amount of cancer stem cells. Estrogen and FGF signalling induces Tbx3 expression, which is another key factor of breast cancer. Tbx3 expression is correlated with ER-positive tumors, so ER-FGF-Tbx3 pathways need some careful attention. Studies of breast cancer had consistently found an increased risk associated with elevated levels of endogenous estrogen in the blood. Yager and Davidson [50] proposed new preventive and therapeutic interventions that block receptor function or drastically reduce the levels of endogenous estrogen through the inhibition of its synthesis, inactivation of the reactive quinones, and inhibition of membrane estrogen-receptor activated second-messenger pathways. Clemons and Goss [51] explained that exposure to estrogen and the risk of breast cancer has been identified in specific groups of women ie., pre and postmenopausal women. The serum estrogen concentrations, breast density on mammography, and bone mineral density prove to be useful tools for assessing a womans risk of breast cancer. The family and reproductive histories may not be given an accurate assessment of risk in individual women but provided a better way in understanding the role of estrogen in the pathogenesis of breast cancer.
Lack of HER2 expression, estrogen and progesterone receptors is another reason for breast cancer. Breast tissues are estrogen-responsive and BRCA deficient cells in the breast and ovarian tissues can escape from apoptosis and lose their damage repairing capacity. Estrogens can regulate the expression and function of c-Myc, Cyclin D1 and activate Cyclin E-Cdk2 complexes, which are the rate-limiting steps in G1–S phase progression. The activation of Cyclin E-Cdk2 by estrogen promotes the formation of high molecular weight complexes, lacking the CDK inhibitor p21 [52].
Obesity-associated with tumor burden and connection with breast cancer was reported by Berclaz et al. [53]. The patients with normal BMI (Body Mass Index) had significantly longer overall survival (OS) and disease-free survival (DFS) than patients with intermediate or obese BMI. Subset analyses showed the same effect in pre- and postmenopausal patients. Obesity proves to be the risk factor for breast cancer in post-menopausal women. The elevated estrogen level promotes body weight and breast cancer risk [54].
Deregulation of mi RNAs in breast cancer
mi RNAs are small non-coding RNA that control gene expression of mRNAs or repressing the translation or degradation of RNA molecules. The aberrant expressions of miRNAs are always associated with several pathological conditions. miRNA biogenesis takes place in the cell nucleus. The Pri-miRNA molecule is coded by RNA polymerase II enzyme. The Pri-miRNA expresses in different genomic regions. The miRNAs’ biogenesis can be regulated by the host’s gene promoters. Pri-miRNAs transforms into pre-miRNAs having 70–85 nucleotides. Pre-miRNAs have impaired stem-loop structure and gets transported to the cytoplasm, where it becomes functional double-strand miRNAs. One of these strands is the mature miRNA sequence, with about 22 nucleotides in length. Another one is the complementary strand. microRNP complex separates the two strands from each other. One of these strands turns into functionally active miRNA while the other one degrades [55]. In human breast cancer cells, deregulation of several miRNAs can be revealed through the microarray and northern blot analysis ie.,mir-125 b, mir-145, mir-21, mir-155 (Table 2). The miRNA expression is associated with breast cancer pathological features are estrogen and progesterone receptor expression, vascular invasion or proliferation index [56]. The use of miRNAs as BC biomarkers was extensively studied. The oncogenic activity of mir 27 is effected by targeting the tumor suppressor genes involved in G2-M checkpoints ie., zinc finger ZBTB10 gene, specificity protein repressor and Myt-1 [57]. The role of miRNA in tumorigenesis and its potential usage as breast cancer biomarkers are the future perspectives for the early diagnosis and treatment of breast cancer [58]. Several miRNAs have oncogenic potential which has been reported earlier. The mir 17–91 clusters show enhanced tumor induction together with c-myc was shown in the mouse B-cell lymphoma model [59]. Normal breast cells and tumor cells possess 133 miRNAs and the breast cancer subtypes show different mRNA expression profiles [60]. miRNA expression analyzed through TaqMan real-time polymerase chain reaction miRNA array methods reveals that mir 21 gene becomes an oncogenic and therapeutic target in the future that down-regulates the apoptotic gene Bcl-2 [61]. miR 125b acts as a tumor suppressor through the regulation of EST 1gene of the proto-oncogene of invasive breast cancer cells [62].
The clinical application of miRNAs
The clinical application of miRNAs
miRNAs expression can be used even as a diagnostic tool to check whether a tumor is metastatic. Elevated levels of miR-21 and miR-155 and lower levels of miR-200 are indicators of such condition. In the case of invasive cancer, miR-21 could be seen [55].
Chemotherapy, endocrine therapy, radiation therapy and immunotherapy are the milestones in the cancer treatments. Breast cancer is categorized into 3 major subtypes based on the presence or absence of molecular markers for estrogen or progesterone receptors and human epidermal growth factor 2, hormone receptor positive/ERBB2 negative, ERBB2 positive and triple-negative. More than 90% of breast cancers are not metastatic at the time of diagnosis. The systemic therapy for non-metastatic breast cancer is determined on the basis subtype. The patients with hormone receptor-positive tumors receive endocrine therapy and a minority receives chemotherapy. In the case of ERBB2-positive tumors receive ERBB2-targeted antibody or small-molecule inhibitor therapy combined with chemotherapy where as patients with triple-negative tumors receive chemotherapy alone [67].
Several gene therapy approaches for breast carcinoma have been developed. RLIP (ral binding protein 1) is a promising target for breast cancer and a good way for the survival of the patients by using an anti-neoplastic agent [68]. RLIP can regulate Akt, Myc, and ERK1/2 signalling mechanisms. A sphingolipid, ceramide has the potential to become an agent for cancer treatment. The sphingolipid, rheostat regulates sphingolipid metabolites, which in turn controls cell fate and cell death. Ceramide mediates cell death by enhancing the activity of proapoptotic membrane molecules, like CD95, and initiating apoptosis. Ceramide can actively participate in mitochondrial membrane channel formation mediated by Bcl-2 proteins and leads to the production of apoptosis-inducing protein [69].
The research on mutagen sensitivity and efficacy of DNA repair could impact the development of new diagnostic and screening strategies as well as indicate new targets to prevent and cure cancer. Moreover, the comet assay may be applied to evaluate the suitability of a particular mode of chemotherapy to a particular cancer patient [70]. The main treatments include Poly (ADP Ribose) polymerase (PARP) inhibitors. The BRCA status of a patient can predict responsiveness to platinum-based chemotherapy, as well as to inhibitors of poly (ADP-ribose) polymerase (PARP). In phase II/III studies of single-agent PARP inhibitors (PARPi) have shown that encouraging progression-free survival results in patients with BRCA1/2-mutated breast cancer, which led to the recent approval of Olaparib, the first PARPi to be approved in breast cancer. Determining BRCA1/2 mutation status in this breast cancer subgroup could potentially expand treatment options beyond the current standard of taxane and anthracycline-based chemotherapy. The detection of RAD51 foci in gBRCA tumors correlates with PARPi resistance. This is a promising biomarker, which can be used in the clinic towards the better selection of patients for PARPi therapy [71]. Radiation therapy is more effective local tumor control with negligible toxicity of surrounding normal tissues. There is the development of radio-resistance through the deregulation of the cell cycle, apoptosis, and inhibition of DNA damage repair mechanisms. Over the last decade, extensive progress in radiotherapy and gene therapy combinatorial approaches has been achieved to overcome resistance of tumor cells to radiation. The interaction between tumor cells and surrounding normal cells controls local tumor development and the radiation resistance. The intervention against the interaction between tumor cells and their microenvironment is a promising area for the development of combination of gene therapy and radiation therapy modalities. The cancer gene therapy vectors irradiated with radiations can enhance the uptake and transcription of the specific gene. The combination of such targeted cancer therapies can contribute to the growing field of personalized cancer medicine that is based on the unique genetic characteristics of the tumor as well as the patient [72].
Recent advances in chemotherapy have significantly improved the prognosis of breast cancer patients. The prediction of chemosensitivity with equivalent accuracy is currently anticipated to further improve breast cancer prognosis [43]. The estrogen and progesterone variation can induce carcinogenic growth. Effect of Hormone Replacement Therapy (HRT) can be effectively targeted to breast cancer risk. The addition of a progestin to HRT enhances markedly the risk of breast cancer relative to estrogen use alone [73]. In immunotherapy, anti-programmed death (PD)-1 antibody is a new class of cancer immunotherapy that specifically hinders immune effecter inhibition and potentially expanding preexisting anticancer immune responses. Thus, the blockade of immune checkpoints is one of the most newly investigated approaches to activating therapeutic antitumor immunity [74]. Immunotherapy with PD-1/PD-L1-blocking antibodies is clinically effective for several tumor types. PD-1 blockade that potentially enhances adaptive cytotoxic T cell potency in a human acute myeloid leukaemia was studied by Deng et al. [75]. The tumor suppressor actions of hexamethylene bis-acetamide (HMBA)-inducible protein 1 (HEXIM1) in the breast, prostate, melanomas, and AML have been reported by Montano et al. [76]. The increased HEXIM1 expression can inhibit cell proliferation and metastasis of cancer cells. HEXIM1 can inhibit the androgen receptor by the formation of anti-androgens. The anti-androgen can prevent the target specific gene expression and proliferation of cancer cells. HEXIM1 induces expression of the histone demethylase KDM5 B (lysine-specific demethylase 5B) and inhibits histone methylation, resulting in the inhibition of FOXA1 (forkhead box A1) licensing activity [77].
HEXIM1 (hexamethylene bis-acetamide inducible 1) has an inhibitory action on ERα (estrogen receptor α) because they can intercept the action of P-TEFb (positive transcription elongation factor b) which is a protein complex comprised of cyclin T1 and CDK9. ERα directly binds to the cyclin T1 domain, P-TEFb, phosphorylates the C-terminal domain of RNAPII (RNA polymerase II) and directly binds to the cyclin T1 component of this complex. HEXIM1 inhibits the co-recruitment of both ERα and cyclin T1 to the promoter region of ERα target genes. As a result, HEXIM1 inhibits the phosphorylation of the C-terminal domain of RNAPII and thereby prevents ERα-mediated transcriptional elongation in breast cells. HEXIM1 can inhibit the transcription of cyclin D1 and VEGF (vascular endothelial growth factor) [78,79]. In some clinical trial studies, it has been found that the neutrophil-to-lymphocyte ratio (NLR) and the lymphocyte-to-monocyte ratio (LMR) are effective tools for the identification of different breast cancer subtypes. Jia et al. [80] found that in triple-negative breast cancer, the NLR become superior as compared to the LMR.
Modern research in breast cancer
A new computer-aided cancer diagnosis technique was developed by Chouguard et al. [81] to rectify the difficulties of radiologists who have a hard time in mammography lesions classification. Convolutional Neural Networks (CNN) system was used to build the breast cancer screening framework and it provides 98.23% accuracy. FDG - PET CT (Fluorodeoxyglucose - Positron emission tomography/Computed tomography is a very useful tool for cancer diagnosis. An FDG PET/CT technique allows the detection of extra axillary lymph nodes and distant metastases. PET/CT also brings valuable information in the staging of clinical stage IIB and primary operable stage IIIA breast carcinoma [82]. This technique can find out the multilocus specificity of breast cancer lesions. Benign or malignant cancer can be identified through FDG PET/CT.
The interaction between cancer cells and the immune system is much important in breast cancer treatment. Now it is under the major area of consideration. The tumor-infiltrating lymphocytes (TILs) are associated with longer disease-free survival (DFS) in HER2+ breast cancer and triple-negative breast cancer. Human leucocyte antigen (HLA) class I encoded by the human leukocyte antigens HLA-A, HLA-B, and HLA-C can provide antigens to CD8+ cytotoxic T lymphocytes to recognize and eliminate infected or tumor cells. Several case studies pointed out that HLA has a potent role in HER2+ breast cancer and triple-negative breast cancer [83,84]. The prognosis of tumor induction can be determined through the pathological conditions associated with it. It mainly includes the stage, grade, human epidermal growth factor receptor 2 (HER2), and hormone-receptor status of the patients. Nowadays the tumor markers have increasing attention in breast cancer clinical studies. The cancer antigen 15–3 (CA15-3) is a member of the mucin-1 (MUC-1) family of glycoprotein over expressed in tumors. A cohort study in Chinese women proposes that pre-operative prediction of breast cancer markers is validating the cancer survival rate and offering a personalized treatment strategy in cancer subtypes [85]. The level of tumor marker is different in different breast cancer subtypes. It is lower in the triple-negative group as compared to the luminal groups [86]. Several studies have been conducted all over the world to eradicate breast cancer malignancy. The increased number of population studies on breast cancer forms an alarming signal that it continues to spread all over the world [87,88].
Conclusion
Cancer is one of the most treacherous diseases in the world. It is the second leading cause of death globally, and was responsible for 8.8 million deaths in 2015. Globally, nearly 1 in 6 deaths is due to cancer. The present review gave special emphasis on the genetic background of breast cancer. Particularly the mechanism of BRCA1 and BRCA2 in breast cancer metastasis and its role in cell cycle check points. The BRCA1 mutation can induces the altered DNA damage responses and repair mechanisms. An idea of molecular subtype of breast cancer and BRCA1 interacting proteins are discussed through the review. The deregulation mechanism of miRNA and hormonal imbalance is always associated with the breast cancer induction. Thus the given review gave an insight on the breast cancer genetics and associated mechanism of cancer induction. It will open a gateway for the molecular studies of breast cancer. The modern research on breast cancer treatment is an exploring area of research. So understanding of basic of breast cancer development is important. Thus the review will enlighten the knowledge of beginners of breast cancer research.
Footnotes
Acknowledgements
The first author gratefully acknowledges Council of Scientific and Industrial Research (CSIR), Government of India (09/043(0182)/2017-EMR-I), for the award of the fellowship.
Conflict of interest
The authors declare that there are no conflicts of interest.
Author contributions
Ramya Sree P.R. - Idea for the article; Ramya Sree P.R. & J.E. Thoppil - Literature search and data analysis; J.E. Thoppil - Drafted and/or critically revised the work.
