Abstract
BACKGROUND:
Radiotherapy after breast surgery decreases locoregional recurrence and improves survival. This is not without risks from radiation exposure and could have implications in clinical practice. Our study investigates the correlation between tumour location and radiation dose to the heart.
METHODS:
Left-sided breast cancer patients who had radiotherapy at Aberdeen Royal Infirmary in 2010 were identified. Tumour location was established from notes and imaging. Radiotherapy planning scans were reviewed, and cardiac doses calculated. The mean cardiac dose, maximum dose and volume of the heart in the field, along with V5-V40, were determined.
RESULTS:
40 patients had mastectomies and 118 breast conserving surgery. The median percentage of the heart in the field and the Interquartile Range was 0.59% (0.03–1.74) for all patients, with the highest for lower inner quadrant (LIQ) tumours 1.20% (0.29–2.40), followed by mastectomy 0.94% (0.02-1.82). The mean heart dose showed a higher median for mastectomies 1.59 Gy (1.00-1.94), followed by LIQ tumours 1.58 Gy (1.31–2.28), with an overall median of 1.42 Gy (1.13–1.95). The median percentage of the heart in the field, the mean cardiac dose and V5-V30 did not reach statistical significance, however, V40 and the maximum dose did.
CONCLUSIONS:
The benefits of radiotherapy after breast cancer surgery are established, but with potential harm from cardiac exposure. Our cohort showed higher radiation exposure to the heart in patients with LIQ tumours and mastectomies but reached significance only for V40 and maximum dose. This highlights tumour location as a potentially important risk factor for cardiac exposure with breast radiotherapy.
Introduction
Breast cancer is the second most common cancer worldwide [1]. The long-term effects of breast cancer treatments are of increasing importance as there has been increased survival with 78% of patients now living beyond ten years post diagnosis [2].
Breast cancer management include a wide range of strategies and the decision making is becoming more complex, often involving multiple modalities and methods. In addition to surgery, adjuvant radiotherapy is an essential part of treatment for local control, and its use is well supported in the literature. Randomised controlled trials with more than twenty-year follow-up document the significant long-term survival benefits of post-operative radiotherapy especially post Breast Conserving Surgery (BCS), and also in selected cases post mastectomy [3–7].
Whilst the benefits of adjuvant radiotherapy in breast cancer are widely accepted, some side effects especially lung and cardiac damage are acknowledged. In 2013, Darby et al., highlighted the increased risk of cardiac disease after incidental cardiac irradiation during radiotherapy in the process of treating breast or chest wall lesions, and were able to establish a linear relationship between cardiac dose and the incidence of cardiovascular disease [8]. Subsequent awareness of the associated cardiac effects of breast radiotherapy has generated improvements to radiotherapy planning, with patients receiving smaller doses of radiation to organs at risk [9]. In 2016, the Royal College of Radiologists (RCR) issued a statement establishing that cardiac sparing breast radiotherapy was to be the standard of care, by exclusion of the heart from the radiotherapy field with breath holding techniques with a census that the mean heart dose in 90% of patients shouldn’t exceed 2 Gray (Gy) [10].
There is a range of modern cardiac sparing techniques, with some aiming to lower the delivered dose of radiation to the heart area (for example, intensity modulated radiotherapy (IMR), proton therapy and partial breast irradiation) and those which attempt to use optimal positioning to eliminate as much of the heart muscle from the field (for example, prone radiotherapy). Deep Inspiration Breath Hold (DIBH) is one such technique gaining traction in recent years. During deep inspiration, the diaphragm descends and flattens, allowing a greater volume of air to enter the lungs and in doing so, the heart is forced away from the chest wall and largely out of the treatment field. In contrast, free breathing in conjunction with respiratory gating is not considered to be effective in reducing the percentage of heart in the field due to the fact that the heart does not move the required distance during a normal cycle of respiration [11]. Two methods of DIBH can be employed: voluntary DIBH (DIBHv) and moderate DIBH (DIBHm). DIBHv involves education of the patient and breath hold training whereas DIBHm (also referred to as DIBH ABC) uses active breathing control devices such as spirometer to observe and hold the flow of air in the patient’s lungs at an optimal threshold for dose delivery. A large 2013 UK based study compared the two methods in three categories: reproducibility, feasibility and ability to spare heart tissue [12]. The study concluded that whilst they are largely comparable in terms of heart tissue spared however DIBHv was more acceptable to patients, had lower set-up times and was also preferred by radiographers. DIBH is certainly a promising advance in the field of cardiac sparing radiotherapy techniques, and one which can be used alone or combined with prone positioning to achieve the best results [13].
It is known that patients with left sided breast cancers are at higher risk due to anatomical proximity and subsequent elevated incidental cardiac dose [8]. Other important factors include, anatomical variations between patients including chest wall shape or deformities such as pectus excavatum, body habitus, body mass index (BMI), breast size and shape, ptosis and the position and angle of the heart itself. All these factors contribute to the cardiac dose received [14]. Additionally, women with pre-existing cardiac risk factors are naturally at greater absolute risk also other risk factors including previous chemotherapy (anthracyclines in particular) are also important [8,15,16].
Whilst previous studies have established the link between adjuvant radiotherapy and an increased risk of cardiac disease, further research is needed to not only clarify the factors that influence cardiac doses, but also offer suggestions to minimise risk. Breast tumour location is one factor which influences the extent of cardiac doses to varying degrees. Due to anatomical proximity, it is suggested that left sided, lower inner quadrant (LIQ) tumours are associated with the highest risk of incidental heart exposure and subsequent cardiac disease [17]. Current practice ensures all patients receiving left sided breast cancer radiotherapy are counselled on the risks, including that of cardiac disease. If the risks, however, increase in association with tumour location, a question remains concerning whether patients with certain, specifically located tumours (such as left-sided LIQ) be counselled, or treated differently.
Our study aims to investigate the impact of left sided tumour location and the dose of radiotherapy received by the heart, and discuss methods to reduce this risk, in particular breath hold techniques.
Patients and methods
This is a retrospective case series that looked at all patients who had radiotherapy for breast cancer at Aberdeen Royal Infirmary between the 1st of January and 31st of December 2010. The study had been approved by the local clinical governance department. The anonymised patients’ data was recorded including the side of the tumour and the type of surgery to the breast (BCS or mastectomy). For the patients with left breast cancers who had BCS, the tumour location within the breast was identified and documented. The tumour location was established reviewing the clinical notes and confirmed using mammography, ultrasonography and MRI reports. Where there were any discrepancies, mammograms were reviewed by a consultant radiologist to establish the position of the tumours. Patients with right sided breast tumours, treated in that same period, were also identified. Recurrence and survival data were collected until January 2018.
The planning technique for treatment of breast cancers following their surgical excision in our practice involves placement of tangent pair matched gantry angle to avoid the heart whilst covering as much breast tissue as possible, with an adequate margin around the tumour bed. It is common practice to set field gantry angles to match up medial and lateral fiducial reference marks but then change the table height, gantry and collimator angles to set a limit on the maximum depth of lung in the field, whilst avoiding the heart (Figs 1,2 and 3).
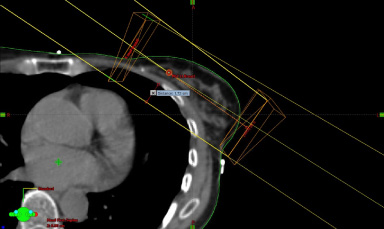
Standard field arrangement ensuring adequate tumour bed coverage with confirmatory measurement of maximum lung depth.
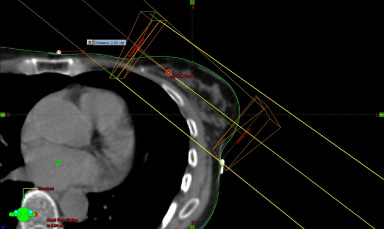
Standard field arrangement with adjusted beam arrangement to avoid heart if tumour bed is lateral (UOQ or LOQ).
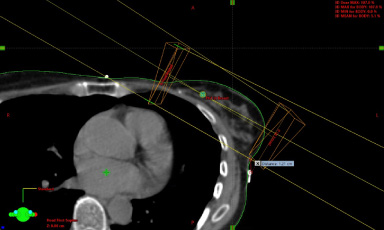
Standard field arrangement with adjusted beam arrangement to avoid heart if tumour bed is medial (UIQ or LIQ).
All patients had planning CT scans which were stored in the Varian Eclipse treatment planning system and dose calculated with the AAA algorithm. Patients received left whole breast or chest wall radiotherapy on a Varian Clinac linear accelerator with a standard dose and fractionation of 40 Gy in 15 fractions using photons. If boost dose was indicated, it was delivered using electrons. Standard immobilisation techniques with breast board and clinical tattoo mark-up were employed. Legacy treatment plans were accessed, and a heart structure was added within the treatment planning software, allowing for dosimetric analysis as shown in (Fig. 4). Each cardiac outline was contoured using a consistent and reproducible technique as illustrated in (Fig. 5). The 50% isodose was chosen as the dosimetric field size and its overlap with the cardiac contour allowed the calculation of:
Irradiated heart volume in cm3 or expressed as a percentage of the total volume Maximum heart dose in Gy Mean heart dose in Gy Percentage of the heart volume receiving 5, 10, 15, 20, 25, 30 and 40 Gy.
The overlap is demonstrated in (Fig. 6) which allowed the treatment planning software to calculate the doses delivered to the heart. Finally, the percentage volumes that received between 5 and 40 Gy (V5, V10, V25, V20, V25, V30, V40) were calculated by reference to a Dose Volume Histogram (DVH).
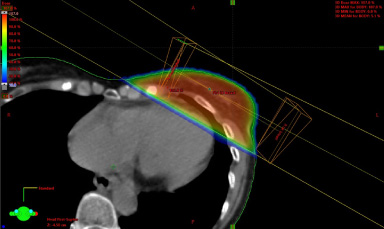
Wedged beam arrangement for left whole breast radiotherapy with a tangential pair and dose wash.
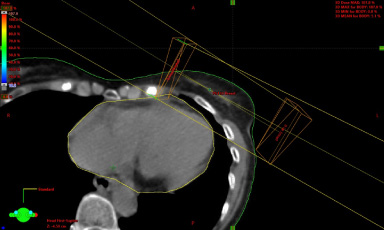
Cardiac contour outline.
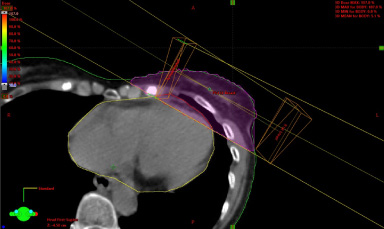
Cardiac contour overlap with tangent pair field to enable dosimetric calculations.
All the results were entered into Microsoft Excel. Statistical analysis was undertaken using IBM SPSS version 24. Median and inter quartile range (IQR) was used for the descriptive data for the analysis and also the means were calculated (Tables 1a and 1b) whilst the Kruskall–Wallis was used to test the null hypothesis comparing the different groups. P value of <0.05 was considered to be statistically significant.
Summary of dosimetric calculations stratified according to tumour quadrant geometry and mastectomy. 1a: median and IQR (Highest Bold and Lowest underlines)
Means
A total of 159 patients were identified who received post-operative radiotherapy to the left breast or left chest wall at Aberdeen Royal Infirmary in 2010. Of these, one was excluded as there was no primary tumour in the breast on imaging. In the remaining cohort of 158 patients, 40(25.3%) had undergone a mastectomy and 118(74.7%) BCS. In the BCS group, 80 had upper outer quadrant (UOQ) tumours, 14 had upper inner quadrant (UIQ), 12 had lower inner quadrant (LIQ), 11 had lower outer quadrant (LOQ) and one had a central tumour. There was no difference in local recurrence between those with right or left sided tumours.
The median percentage of the heart in the radiotherapy field and the Interquartile Range (IQR) was 0.59% (0.03–1.74) for all patients, with the highest percentage for those with LIQ tumours 1.20% (0.29%–2.40%) followed by mastectomy 0.94% (0.02%-1.82%) and the lowest for patients with LOQ tumours, of 0.45% (0.00%–0.73%). On the other hand, the mean heart dose showed a higher median for mastectomy patients 1.59 Gy (1.00–1.94) followed by LIQ tumours 1.58Gy (1.31–2.28) whilst LOQ showing the lowest results 1.17 Gy (0.90–1.52), with an overall median of 1.42 Gy (1.13–1.95). The V5, V10, V15, V25 and V30 consistently showed a higher result for both Mastectomy and LIQ with LOQ showing the lowest results (Tables 1a and 1b). The maximum dose showed that the mastectomy patients had the highest maximum dose with a median of 39.88 Gy (34.23–41.00), followed by LIQ while the lowest was the LOQ tumours.
We have also calculated the means as this was used in previous papers, but because the data didn’t follow normal distribution, median and IQR were used for the statistical analysis. It is worth mentioning that the means show that LIQ tumours have the highest results for the mean heart dose, the percentage of the heart in the field, V5-V30 and only the maximum heart dose and V40 was higher for the mastectomy. 24.4% of the patients received more that 2 Gy, we see the same trend with the highest percentage for the LIQ tumours with 33.3%, followed by 25% for the UOQ, 22.5% for the mastectomy group, 21.43% for the UIQ and 18.8% for the LOQ, and was much higher than the recommended 10% keeping in mind that the recommendation by the RCR came few years after this cohort of patients were treated.
The mean percentage of the heart and the mean cardiac dose, along with the V5 to V30 failed to reach statistical significance between the different quadrants. However, both the V40 and the maximum dose did reach significance with a P-value of <0.011 for the maximum heart dose and <0.001 for the V40.
Discussion
The advantages of radiotherapy, both in decreasing local recurrence rates and improving overall survival in breast cancer are well established [5–8]. Any survival benefit, however, may be reduced by up to 33% due to increased mortality from cardiovascular disease as a direct result of incidental cardiac exposure [7–9,15,16,18]. A case control study of major coronary events by Darby et al. in 2013 reviewed 2168 radiotherapy-treated breast cancer patients, of whom 963 women would subsequently suffer major coronary events. Overall, it was observed that any dose of radiotherapy to the myocardium is significant, with a dose dependant risk showing a linear trend demonstrating increasing risk of cardiac morbidity by 7.4% per Gy received. Additionally, the study explained that whilst the sustained damage may become evident within only a few years, it would likely to develop and continue beyond 20 years post treatment. Internationally, there is clear variation in approaches to cardiac sparing radiotherapy which was demonstrated in a systematic review in 2015 of 167 studies from 28 countries. The observed mean heart dose from left tangential whole breast radiotherapy (WBR) and chest wall radiotherapy was 4.1 Gy [9] with the highest mean dose delivered to heart reaching the value of 7.9 Gy (reported in Saudi Arabia). Taking into account these findings, there is a clear need for new service implementation to improve patient outcomes.
The literature is sparse and reveals mixed results when exploring the relationship between tumour location in the breast and cardiac dosage. While several studies have shown that tumour location in the breast can indeed be of significance [19–21], others fail to confirm this, illustrating a need for further clarification [22,23]. One larger study of 769 patients reported patients with medial tumours sustained significantly higher cardiac doses [24]. Another study of 69 patients that compared all four breast quadrants failed to show a significant difference [25]. The difference in size between the cohorts could certainly be an influencing factor in the opposing results obtained, with a larger cohort required to observe a significant trend.
Our cohort was also relatively small in size, potentially offering insight into the statistical insignificance of results observed. In 2010, however, the mainstay approach to breast radiotherapy at Aberdeen Royal Infirmary was cardiac sparing, involving placement of tangential fields in the most reproducible manner with margins of at least 1.5 cm, ensuring adequate tumour bed coverage but yet keeping organs at risk in mind. In this way, incidental cardiac doses delivered to the heart were, for the most part, well within the lower standard limit to be set by the RCR six years later [10]. This, in our opinion, is likely to have contributed to the results observed.
When comparing results for the four quadrants only, as expected, LIQ tumours had consistently higher mean cardiac doses, maximum dose and V5-V30 in addition to the highest percentage of heart in the field. LOQ tumours represented the other end of the spectrum, reporting the lowest mean cardiac doses, maximum dose and percentage of heart in the field. The lowest values observed for V5-V30 alternated between LOQ and UOQ tumours.
The post mastectomy group had high incidental cardiac irradiation, as these patients needing post mastectomy radiotherapy usually have larger tumours, more likely to be node positive with close or involved posterior margins but attention is given to irradiating the chest wall considering the anatomy of the contralateral breast. Additionally, the ipsilateral chest wall was treated to midline in the majority of cases which can help to explain the comparatively higher cardiac doses observed. While our study reports low cardiac doses with the median heart dose of 1.42 Gy and a mean of 1.64 Gy, an estimated one in four patients received a dose of over 2 Gy.
Determining potential solutions to the associated cardiac risk of radiotherapy begins with an understanding of the complex underlying pathophysiology. Recent literature suggests the primary mechanism is inflammatory, whereby an induced inflammatory state producing increasing amounts of reactive oxygen species (ROS) eventually result in impaired healing ability of the affected coronary vessel. This causes endothelial fragility resulting in the inability of the vessel to undergo proper healing making it more susceptible to the development of atherosclerotic plaques [26]. Furthermore, anatomy dictates that the heart’s Left Anterior Descending Artery (LAD) is most at risk [27]. The potential adverse effects are found to be wide-ranging, with reductions in regional myocardial perfusion [28], resulting in ischaemic changes and loss in vital elasticity of the myocardial tissue [26]. As it is frequently the case in medicine, the ostensible benefits from these treatments, in the main, may be thought of as being of greater significance than the risks of cardiac toxicity. Assuming this level of risk, interventional efforts should be directed at those patients who would sustain the most cardiac damage. In our study, this would indicate attention be on those patients in the LIQ and mastectomy groups.
Whilst tumour location is the focus of this study, it is important to consider all factors which contribute to cardiac exposure and dosage. In dosimetric calculations, the contribution of cardiac contouring is essential with specific guidance on the cardiac outline and cardiac atlases [29]. It is well recognised, however, that this is subject to both intra-operator and inter-operator variation. Whilst our study had one operator contouring all cardiac contours, hence eliminating inter-operator variation, this is another variable to consider when making efforts to standardise lower cardiac dose regimes. In addition, the choice to treat internal mammary chain (IMC) lymph nodes is a debated but undoubtedly significant factor in determining a patient's cardiac exposure and subsequent risk [11,30]. The patients in our study were exempt from IMC treatment, however, as the field directs more attention towards treating these lymph nodes to reduce recurrence, the implementation of cardiac shielding techniques becomes even more crucial.
Based on the extremely positive results and significant reductions seen with DIBH treated breast cancer patients versus non-DIBH. the radiotherapy department at Aberdeen Royal Infirmary intends to treat all left sided breast cancers with DIBH from December 2019. A significant reduction in cardiac doses is expected across all measurement points and is a major step to improving patient outcomes, and enable us to start treating the internal mammary chain in suitable patients [13]. We acknowledge that new service implementation such as DIBH exerts increased demands on health care providers and patients alike. The costs and logistics of the additional set-up time and time required to educate such a large population of patients cannot be avoided, as well as the base cost to acquire the devices and technology. However, balancing these potential barriers with the expected results would undoubtedly suggest the benefits to adopting DIBH in order to spare cardiac tissue during left breast radiotherapy is a worthwhile endeavour.
This retrospective study details a single centre experience of 158 patients treated over one calendar year and concludes that even with attention to cardiac sparing using conventional methods, tumour location in the inner quadrants of the breast, especially the lower inner quadrant (LIQ) experience increased cardiac doses. Based on the small cohort used, and the mixed literature available on the relationship between cardiac dosimetry and left-sided tumour location, there is a clear need for further prospective studies. Ideally, those with larger numbers could clarify the presence of statistical significance in trends shown. This study highlights tumour location as one of the many important risk factors in cardiac radiotherapy exposure and proposes DIBH as a new technique to reduce incidental irradiation of the myocardium and at-risk vessels. Whilst strain on resources is a potential barrier, based data shown, our study provides a strong case to adopt DIBH for subgroups where need is highest.
Footnotes
Funding
There was no funding for this project.
Conflict of interest
Yazan A Masannat, declares that he has no conflict of interest.
Gabija Lazaraviciute, declares that she has no conflict of interest.
Ian K Garbett, declares that he has no conflict of interest.
Natesh Shivakumar, declares that he has no conflict of interest.
Brittany V Brownlee, declares that she has no conflict of interest.
Tanja Gagliardi, declares that she has no conflict of interest.
Andrew Bromiley, declares that he has no conflict of interest.
Steven D Heys declares that he has no conflict of interest.
Ravi Sharma declares that he has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors. This is retrospective study analysing data on patients that have been treated in the past.
