Abstract
INTRODUCTION:
Primary breast sarcoma (PBS) was first described in 1887 as a rare heterogeneous neoplasm arising from the mesenchymal tissue of the mammary gland accounting for less than 5% of all body soft-tissue sarcomas and less than 0.1% of all malignant tumors of the breast.
CASE PRESENTATION:
A 31-year old lady presented with left breast mass which she felt four years before during which the mass increased in size from 1 to 6 cm in the largest diameter, diagnosed clinically as a benign fibroadenoma without any further cytological or histopathological confirmation. Histopathological examination of the excised breast mass reveals undifferentiated, primary stromal sarcoma of the breast (PSSB), which was followed by mastectomy three weeks later with reconstructive breast surgery with a total duration of follow-up of 3 years thereafter. This is the first case of PSSB reported in Jordan.
DISCUSSION/CONCLUSION:
PSSB is the generic term given to malignant breast tumors thought to arise from the specialized mesenchymal stroma of the breast but lacking an epithelial component with a phylloides pattern. PSSB is difficult to diagnose preoperatively due to its rarity and inadequate imaging methods to establish an exact diagnosis. The histology of the patient mass may be the leading factor for the management of these tumors. Even in very young patients, a progressively growing breast mass should alert the clinician to investigate for malignancy and verify the results by biopsy. Surgery with adequate resection margins represents the only potentially curative modality with prognostic significance. Adjuvant chemotherapy and radiotherapy are not very beneficial. The prognosis is dismal for patients with lymph node involvement and the size of the tumor has a lesser bearing on the outcome.
Introduction
Primary breast sarcoma (PBS) first described in 1887 is generic term given to malignant breast tumors thought to arise from the specialized mesenchymal stroma of the breast but lacking an epithelial component with a phylloides pattern [1,2] on thorough histopathological sampling, with exclusion of sarcomas arising from the skin, muscle, and adjacent bone [3]. Primary breast sarcoma accounts for less than 5% of all body soft-tissue sarcomas [4] and less than 0.1% of all malignant tumors of the breast [5].
It must be borne in mind that the breast is a rare site for primary presentation of sarcoma [6] and those that are seen are either the extremely rare primary pure breast sarcomas [7] or those that are seen as an advanced breast sarcomas which may follow radiotherapy, which forms the great majority of cases, due to increasing use of breast radiation after breast-conserving treatment [8], with about 1 of 300 patients receiving radiotherapy for breast cancer can be expected to develop sarcoma [9].
At the Mayo Clinic, 27 881 malignant breast tumours were seen between 1940 and 1999 (C Adem, personal unpublished data) and 18 breast sarcomas were diagnosed accounting for 0.0006 rate of breast malignancies [5].
Morales-Miranda [10] report 0.0002 rate (8 patients out of 40,000) in a retrospective review of PBS using Breast Clinic data from the National Cancer Institute in Mexico from 2000 to 2010.
The annual incidence of sarcomas of the breast among women in the United States, between 1973 and 1986 was 44.8 new cases per 10 million women [11].
Only a few hundred cases of breast sarcomas have been reported in the literature up to 2014, making the diagnosis difficult [8].
We report a case of 31-year-old female in which excision of a clinically diagnosed breast benign fibroadenoma of 4 years’ duration without any further cytological or histopathological confirmation reveals undifferentiated primary stromal sarcoma of breast.
Case report
A 31 year-old lady presented in January 2018 with a left breast mass which she discovered during 2014, 4 years before, when she was 27 years old after her 1st childbirth. At that time, the mass was well circumscribed, freely mobile, painless, about 1 cm in diameter. She was reassured that the mass was clinically benign fibroadenoma, without any further cytological or histopathological confirmation. She had no family history of breast cancer and there was no history of breast trauma, implants or irradiation.
Three years later (June 2017) and during her 2nd pregnancy, she noted significant increase in the size of the mass which became painful. Ultrasound examination revealed well circumscribed hypoechoic mass 4 cm in diameter, but unfortunately, no further confirmation was done at that time and she planned for a follow up 6 months after her second delivery.
At follow up in January 2018, examination reveals large, mobile rounded, firm, well defined palpable mass in the left breast. There was no left nipple discharge, no palpable axillary lymphadenopathy with normal contralateral breast. All vital signs and chest X-ray were normal.
The clinically diagnosed benign fibroadenoma (with no further confirmation) was excised and sent for pathological examination. Gross examination reveals 78 grams oval, well-encapsulated, whitish soft mass (softer than the usual firm fibroadenoma), and measuring 60 × 50 × 30 mm. On section it looks homogenous, uniformly pinkish-white and soft. Histopathological examination showed poorly differentiated sarcomatous tumour, with predominance of atypical elongated to plump spindle-shaped cells showing considerable cytological pleomorphism with areas showing storiform architecture (Fig. 1) and numerous bizarre multinucleated tumor giant cells (Figs 2, 3). Normal and abnormal mitotic figures were seen in the range of 10–15/high-power field with focal areas of necrosis. Neither epithelial carcinomatous cells elements nor entrapped breast acini or ducts were seen within the tumor. No focal ossification or mucinous degeneration was seen.
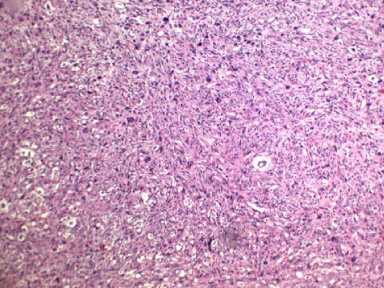
Microscopic image of the tumor showing the storiform architecture (H&E).
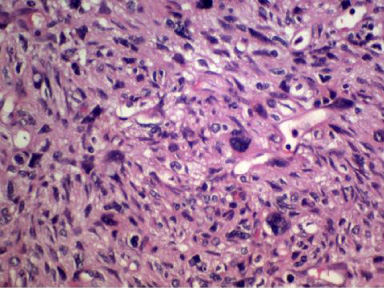
Microscopic image of the tumor showing the spindle shaped cells, numerous bizarre multinucleated tumor giant cells with normal and abnormal mitotic figures (H&E).
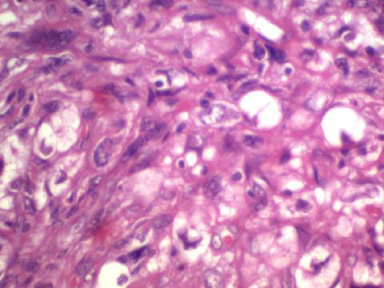
Microscopic image showing atypical elongated to plump spindle-shaped cells showing considerable cytological pleomorphism (H&E).
The Cancer Staging Manual (8th Ed, 2017) [12] issued by the AJCC include the Breast Sarcoma staging with the Soft Tissue Sarcoma of the Trunk and Extremities, and accordingly the TNM tumor stage of our patient is T2N0M0 (T2 = Tumor more than 5 cm and less than 10 cm in greatest diameter).
Immunohistochemistry showed spindle cell tumor with a mesenchymal phenotype Positive for Vimentin only and for mitoses Ki-67; and Negative for all epithelial markers as well as all other mesenchymal markers tested, including S-100, CD 68, myogene, desmin, leukocyte common antigen, Keratin, smooth muscle antigen, CD34, HMB45, EMA, CD10, p63 and Cytokeratin IHC (CKHMW) (Fig. 4).
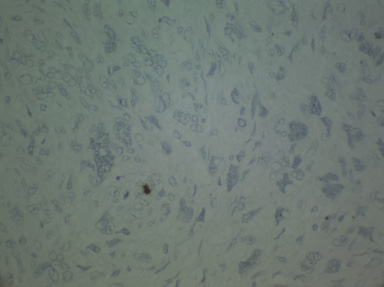
Microscopic image showing Negative CKHMW immuno-stain (Dafco Company) of the tumor cells with positive stain of very few intervening breast epithelial cells (×400).
The tumor was diagnosed as undifferentiated primary stromal sarcoma.
Staging workup including computed tomography (CT) of chest, abdomen, pelvis and bone scan were all free from secondaries. Three weeks after lumpectomy, completion mastectomy with sub pectoral expander reconstruction was carried out, microscopic examination of the mastectomy specimen showed residual foci of sarcomatous tumor cells in the wall of the lumpectomy cavity, with marked foreign body granulomatous reaction to suture material. The nipple, all lines of resection and the rest of the breast tissue were within normal.
The postoperative course was uneventful. Adjuvant treatments with chemotherapy or radiotherapy were not given. She delivered her third child in July 2019. She was well and her follow up Pet CT scans, up to 36 months later (December 2020) reveals no evidence of metastasis.
Subtypes of breast sarcoma
In USA Cancer Statistics 2010, the breast cancer remains the heading type of cancer with an estimate of 28% in American women [13]. Breast cancer is the most common cancer diagnosed among US women (excluding skin cancers) and is the second leading cause of cancer death among women after lung cancer; with approximately 13% of women (1 in 8) will be diagnosed with invasive breast cancer in their lifetime [14].
Most invasive breast neoplasms are epithelial tumors, while mesenchymal breast neoplasms are rare. Stromal sarcomas of the breast were first defined by Berg et al. in 1962 [15] who excluded malignant phyllodes, lymphoma and angiosarcoma from his study and described 25 cases of homogeneous tumours with fibrous, myxoid and fatty patterns seen on light microscopy. The tumours were named stromal sarcoma of the breast because they were considered normal variants of mammary duct stroma. If the morphological features of a specific type of sarcoma are related to its cell of origin, e.g. “liposarcoma, fibrosarcoma” a more meaningful classification for prognosis and therapy may be proposed.
The incidence, histological type and clinical course of sarcomas of the breast are not well established. This is due to the rarity of these tumours and a lack of definition in various reports. These neoplasms, therefore, pose a significant diagnostic and therapeutic challenge.
Adems et al. [5] mentioned 16 published series in the literature between 1967 and 2000 that have included many different entities under the title of “sarcomas of the breast” such as malignant cystosarcoma phyllodes, lymphosarcoma and even carcinomas with pseudo-sarcomatous changes. In his review, Adems et al. choose to categories primary breast sarcomas in histogenic terms, similar to other soft-tissue sarcomas, thus including angiosarcomas, and excluding malignant cystosarcoma phyllodes, as reported by another 12 authors mentioned in his review of the literature in 2004 [5].
The pathological classification of primary breast sarcoma produces controversy because the disease consists of a heterogeneous group of malignancies, as seen in soft-tissue sarcomas in other parts of the body. The whole complement of sarcoma sub-types have been reported to occur in breast. The most common sub-types are malignant fibrohistiocytoma (MFH) (Myxoid), fibrosarcoma, angiosarcoma and spindle cell sarcoma. Several other sub-types (leiomyosarcoma, liposarcoma, rhabdomyosarcoma, haemangiopericytoma, synovial sarcoma, osteosarcoma, chondrosarcoma, neurosarcoma and stromal sarcoma) have been described as smaller percentages of case series or as case reports [9]. Angiosarcomas appear to be the commonest histological subtype in radiation-induced sarcomas of the breast [16,17].
Advances in diagnostic methods, such as immunohistochemistry and electron microscopy, have enabled more accurate identification of the cellular origin of specific tumors [18]. Histological grading of PBS correlates poorly with its clinical behavior [19]. Since only case reports or small series are available in literature, PBS is not well-understood [20]. PBS occurs over a wide age range; Table 1 shows that the youngest patient was 14 years old and the oldest at 80 years.
A literature review of primary stromal sarcoma of breast (PSSB)
A literature review of primary stromal sarcoma of breast (PSSB)
The etiology of breast sarcoma is largely unknown [3]. The majority of breast sarcomas present without an identifiable etiologic factor [9]. The predisposing risk factors for breast sarcoma in general are not clearly known. A main risk factor for the development of breast sarcomas is previous radiation treatment for a breast carcinoma. An additional risk factor is chronic lymphedema. Professional exposure to vinylchloride and artificial implants constitute debatable risk factors [21–23]. Preexisting fibroadenomas, and as sarcomas are part of the hereditary LieFraumeni syndrome tumour spectrum, thus, breast sarcomas may develop in these patients carrying a mutation in p53 [9,24]. Radiation-induced sarcomas of the breast develop after a wide range of time from radiation treatment. A cumulative incidence of 0.3% at 15 years after radiation treatment has been reported in a series of patients diagnosed between 1973 and 1997, some of whom had been treated before this interval [16]. About 1 patient in 300 receiving radiotherapy for a breast cancer can be expected to develop a sarcoma during the 15 years that follow [9].
In our case, no adenomatous or epithelial elements were seen within the tumor to support the possibility that this tumor was associated with or developed on top of preexisting fibroadenomas.
The related single case reports about primary stromal sarcoma of breast (PSSB) are rare, they include (11) reports by Fuyama (1995) [25], Hefny (2004) [26], Cila (2008) [27], Tekbas (2012) [28], Gesakis (2014) [8], Nagaraju (2015) [29], Kumar (2016) [30], Inaba (2016) [31], Amanda Lino de Faria (2018) [32], Alkhudari 2019 [33], with an additional one series of six stromal sarcomas of the breast analyzed in a retrospective study of 53 years, from 1954 to 2007, in one hospital in China by Cong (2009) [34]. Our case report is an additional one.
Treatment
The treatment for PBS is planned by a multidisciplinary team following the treatment model of sarcomas in other locations [8]. However, there is still no definitive consensus regarding the treatment of primary breast sarcomas [20]. Surgery represents the only potentially curative modality [31,35]. Surgical resection is first line of treatment, mastectomy without axillary lymph node dissection, is treatment of choice for primary breast sarcoma, because the axillary lymph nodes are seldom involved. Axillary dissection should be avoided unless there are clinically positive nodes [36]. Few selected patients may be treated with wide local excision [20]. An adequate resection with negative surgical margins is the most important determinant factor of local recurrence and long-term survival in such cases [3,35]. Adjuvant radiotherapy should be considered in high-risk cases and has been recommended especially for large or high-grade tumors. The role of chemotherapy, however, is unclear and can be proposed to patients with the worst prognosis [20,37].
Prognosis
In local disease such as primary early breast sarcoma, complete microscopic resection of the primary tumor is an important factor for local disease control, overall survival, and the disease free survival at 3-year, as the disease is characterized by its tendency to recur locally. In our patient the postoperative disease free survival was 3-years (January 2018 to December 2020), which is similar to that reported by others; Johnstone 1993 [38] report that with a median potential follow up of 99 months postoperatively, seven patients remain alive and without evidence of disease 142, 119, 82, 48, 45, 28, and 19 months postoperatively.
Pandey et al. [36] study concludes that the margin of surgical resection was found to be the most important prognostic indicator, and all attempts should be made to achieve a negative margin as this appears to be the only factor influencing survival in these patients. Pathological prognostic factors play a considerable role in soft tissue sarcomas, and the tumor grade was reported to be the single most important pathologic prognostic factor in soft tissue sarcomas [36]. Overall, the prognosis of PBS is poor [39] and it is dependent on the tumor grade, size and histological type (angiosarcoma vs others) and for most patients, therapy can be similar to that administered for soft tissue sarcomas of other sites [20]. Tumors with size measuring less than 5 cm are associated with better outcome [36].
Table 1 shows wide variation in the reported PBS tumor size at the time of presentation, ranging from smallest 16 mm in greatest diameter in an 80 years old women reported by Inaba et al. [31] to a massive 350 × 250 mm in a 36 years old women reported by Hefny et al. [26]. In our patient the tumor mass was 60 mm in diameter. Like in breast carcinoma, delay in primary breast sarcoma diagnosis has important clinical and treatment implications [39]. The reported 5-year survival rates for patients with primary breast soft tissue sarcomas range from 14% to 91% [1,3,38,40].
Conclusion
In conclusion, well defined breast masses are generally interpreted as benign, especially in young females. Stromal sarcoma of the breast is difficult to diagnose preoperatively. Due to the rarity of breast sarcoma and inadequate imaging methods to establish an exact diagnosis, radiologists and clinicians may misdiagnose and merely follow these tumors. The histology of the patient mass may be the leading factor for the management of these tumors as was the case in our patient. Even in very young patients, a progressively growing breast mass should alert the clinician to investigate for malignancy and verify the results by biopsy.
Footnotes
Acknowledgements
The work was conducted at Prince Hamzah Hospital, Amman, Jordan.
Our gratitude to Prince Hamzah Hospital for the technical support they provided to accomplish this paper.
Statement of ethics
Ethics approval was granted from the institution review board (IRB) of Hashemite University and Prince Hamza Hospital. The patient has given a written informed consent to publish her case and supplementary material.
Conflicts of interest
The authors declare that there is no conflict of interests.
Funding
This project is not funded.
5.
Mohammad Al-Wiswasy: study design, manuscript preparation, literature search.
Mahmoud Al-Balas: data collection, manuscript preparation, literature search.
Raith Al-Saffar: manuscript revision, literature search.
Hamzeh Al-Balas: data collection, manuscript revision, literature search.
