Abstract
Background:
Breast cancer is the commonest cancer among women. India along with United States and China collectively account for one third of the global burden. The present study reports the clinico-epidemiological data of our patient population. This may help in better understanding of the disease in our population and also form ground for conducting further breast cancer research in India.
Methods:
The study was conducted at an apex teaching and medical research institution in India from September 2013 to April 2015 as a retrospective review of prospectively collected data of breast cancer patients. The socio-demographic characteristics, reproductive risk factors, clinical presentation, TNM staging and histopathological characteristics for breast cancer in these patients were recorded. The data was recorded on an Xcel spreadsheet and analyzed using IBM SPSS 21.
Results:
The study comprised of 1310 breast cancer patients with males comprising 1.1%. The median age of presentation was 47 years, and menarche 14 years. Most of women were married and multiparous. More than half of the women were postmenopausal at presentation. All patients were symptomatic at presentation with median duration of symptom of 5 months and median lump size of 5 cm. Most common stage at presentation was Stage II and most common histopathology was Invasive ductal carcinoma. 61.9% tumors were hormone receptor positive. Triple negative cancers formed one third of all tumors.
Conclusion:
Breast cancer in the Indian scenario is a disease of younger woman who lack the characteristic reproductive and demographic risk factors. This calls for a need to study the clinico-demographic risk factors and characteristics of our own population.
Keywords
Introduction
Breast cancer is the commonest cancer among women with an estimated 1.67 million new cancer cases diagnosed in 2012, constituting about 25% of all cancers [1,2]. India along with United States and China collectively account for one third of the global burden. India has witnessed an 11.54 % increase in incidence and 13.82 % increase in mortality due to breast cancer during 2008–2012 [1,2]. It is now the most common cancer in woman in five out of six population based cancer registries (PBCRs) in India [3].
The available Indian literature [4,5] and our clinical experience suggest that the presentation, risk factors, outcome and behavior of most of our breast cancer patients differ from that reported in the west. However, most of the understanding of breast cancer, its management protocols and survival data is based on the studies from the west. There is a lack of data on various epidemiological aspects of breast cancer in India and the only credible data is available from the various urban and rural PBCRs and hospital based cancer registries, which work under the national cancer registries program of the Indian Council of Medical Research [6].
Urbanization and adoption of westernized lifestyle may explain the rising incidence in the urban PBCRs in India, however, the statistically significant increase in age adjusted rate over time (1982–2014) of 1.87% seen even at the rural PBCRs at Barshi makes a case for studying the patient profile in detail in this ethnic and geographic region of the globe [3]. The present study reports the clinico-epidemiological data of the breast cancer patients visiting an apex medical teaching and research institution in Northern India. This may help in better understanding of the disease in our population and also form ground for conducting further breast cancer research in India.
Material and methods
The study was conducted at an apex teaching and medical research institution in Northern India from September 2013 to April 2015 as a retrospective review of prospectively collected data of breast cancer patients after approval from the Institute ethics committee. The study included breast cancer patients who were enrolled in a single surgical unit run breast cancer clinic from January 1993 to December 2014. Patients having malignancies other than carcinoma or those who underwent definitive surgery for breast cancer outside the index surgical unit were excluded. The data was recorded from the breast cancer files, which were maintained for each patient from the time of inception of the clinic. The review of these case files was carried out to obtain information on various socio-demographic characteristics like age, gender, religion, marital status, reproductive risk factors (age at menarche, menopause, first child birth, parity, breast feeding), clinical presentations (presenting complaint, clinical examination findings), American Joint Committee on Cancer (AJCC) Tumor, Node, Metastasis (TNM) staging and histopathological characteristics. The treatment received by the patients, and their survival outcomes measured by overall (OS) and disease free survival (DFS) was also noted. Attempt was made to complete the missing data during routine clinic and telephonic follow-ups. However, still information for some variables wasn’t available for all patients. Hence, the total number of patients for whom complete information was available for a particular parameter are indicated at respective places. During this time span of over two decades, the staging of breast cancer underwent revisions and all cases prior to publishing of AJCC VII were restaged according to this system. The final clinical and pathological data is presented here in accordance to the AJCC VII edition [7]. Early Breast Cancer (EBC) included T1N0, T2N0, T1N1 AND T2N1 tumors. Locally advanced breast cancer (LABC) included lesions more than 5 cm in size or those with chest wall involvement, skin fixity or N2/N3 disease without evidence of distant metastasis. All patients with distant spread were labeled as metastatic. Hormonal receptor expression (Estrogen receptor-ER, Progesterone receptor-PR) is reported in this study as positive or negative as differing methods of receptor positivity have been used for reporting over the past decade. Her2neu receptor positivity was taken as positive for 3+ on Immuno-histochemistry (IHC) or amplification positive result on Fluorescence in situ hybridization (FISH), when available. Grading of the tumors was done using the Nottingham modified Scarff-Bloom-Richardson system and were assigned from I to III.
Data was entered into Microsoft Excel 2011, following which data analysis was done using IBM SPSS 21. The graphs were generated using a data analysis application Wizard. For the various continuous variables, descriptive statistics involved mean, median, range and standard deviation when appropriate. Means were compared by unpaired t test for two categories or the Mann Whitney test when non normal distribution of the variable was encountered. When comparisons were to be made amongst groups more than two in number, the ANOVA test was used or the KruskalI Wallis in case distribution free method was needed. For the categorical variables, proportions were noted and compared between groups using the Chi square test and Fischer exact test. To find correlations between two variables Pearson’s correlation was used when data was normally distributed. Otherwise Spearman’s rank correlation was used. Linear and logistic models for univariate and multivariate analysis of continuous and categorical variables calculated regressions respectively. We compared agreements between few variables using the Cohen’s kappa and IntraI class Correlation Coefficient (ICC). We evaluated two ‘time to event’ variables viz. Disease free survival (DFS) and Overall Survival (OS). DFS were calculated from the time of intervention to the time of first recurrence. Time of first recurrence was equated to time of death if the interval between them was not known. OS was calculated from the time to intervention till the time of death. These ‘time to event’ variables were described using the life tables and Kaplan Meier product limit estimator. The log rank test was used to compare to Kaplan Meier curves. For calculation of Hazard ratios for different variables that impacted survival, the Cox proportional hazard model was used for univariate and multivariate analysis. For all calculations, estimates were considered significant if
Results
The study comprised of 1310 breast cancer patients enrolled from 1993 to December 2014. The majority of our patients were females with males comprising 1.1 %. The median age of presentation was 47 years. 114(9.4%) of patients were less than 35 years and 18% were above 60 years. The male patients were significantly older than female patients (mean for males 60.5 years versus 47.88 years for females). The demographic characteristics are summarized in Table 1.
Demographic characteristics
Demographic characteristics
The reproductive risk factors presented in Table 2. Nine (0.7%) of our patients were unmarried. Median age at menarche was 14 years. Only 103 patients were nulliparous out of 1203 on whom the data on parity was available. Median age at first childbirth was 21 years. 1173 (89.54%) had breast-fed. The median age of menopause was 46 years (range 27–59). There were more postmenopausal women (54.57 % versus 45.43%;
Reproductive risk factors
Amongst the other risk factors analyzed, 67 patients had history of undergoing surgery or biopsy on breast for a previous history of benign breast disease. Twenty-six women (2%) had history of taking oral contraceptives or hormone replacement therapy. Data on family history was available for 1204 patients, and 54 (4.5%) of these patients had a first or second-degree relative affected with breast cancer. Thirty-nine patients had family history of other solid organ malignancies, most common being lung followed by cervix.
All patients were symptomatic at presentation. The most common symptom was an isolated breast lump (88.09%). The median duration of symptoms was 5 months (mean 7.8 + ∕− I9 months, range 10 days-07 years). There was a significant difference in the duration of symptoms between EBC (range: 03 days – 7 months; median 4 months) and LABC (range: 5 days to 07 years; median 6 months;
Characteristics of patient presentation
Mean clinical size of the lump was 5.6 cm with a median of 5 cm. The most common T stage was T2 (35% approx), followed by T4 stage with 345 patients (27 %). Complete information on axillary lymph nodes was available in 1246 cases. Clinically, most of our patients had a mobile axillary node (N1), followed very closely by N0 status (48% versus 40.29%). Fifteen percent of patients had metastasis at time of presentation. Visceral metastasis was seen in 35%, and skeletal and soft tissue metastasis in 30% of those with metastasis.
Less than five percent of patients had stage I disease. Thirty three percent (433/1310, 33%) patients had EBC, 484 (37%) had LABC and 199 (15% were metastatic at presentation. Another 15% patients presented after having lumpectomy/excision biopsy performed elsewhere rendering staging difficult. Table 4 depicts the TNM classification of the tumors as per AJCC VII.
Clinical TNM classification (AJCC VII)
The most common pathological type was Invasive Ductal Carcinoma (IDC) of the No Specific Type (NST) accounting for 93% of the total.
Histopathology
The data on hormone receptors was available for 768 patients as the testing for the same became more readily available in our institution from 2004 onwards. Out of these 768 patients, 61.9% were hormone receptor positive (either ER or PR). 38.9% of the patients were both ER and PR positive and nearly equal proportions of patients (38.2 %) were negative for both steroid receptors. 61.8% of tumors in patients less than forty had hormone receptor expression as compared to 86% in patients older than seventy years. The HER-2neu status was available in 504 tumors and 39.08% of these were positive for the same. Triple negative Breast Cancers accounted for 33.9% of these tumors. These are detailed in Table 5.
The initial treatment offered to our patients was surgery in 67% and neoadjuvant chemotherapy in 33% of the non metastatic patients. In patients with early breast cancer, 6.9% received neoadjuvant chemotherapy to increase chances of breast conservation. 32.7% of locally advanced breast cancer patients had upfront surgery compared to 93.1% in the EBC group.
Complete information on surgical management of primary was available in 1049 patients. 77% of these underwent mastectomy, which included skin sparing mastectomy in 1%, radical mastectomy in 3% and palliative mastectomy in 3%. The remaining 23% patients underwent breast conservation surgery. Only 21 patients opted for breast reconstruction after mastectomy. When stage wise comparisons were done, conservations were significantly more in EBC patients rather than in LABC (31.4% versus 12.6 %;
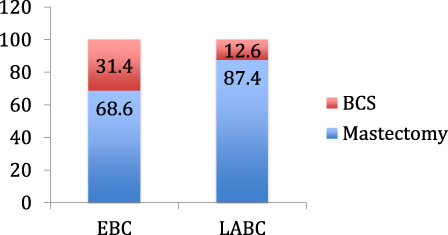
Stage wise distribution of surgery for primary lesion (EBC: early breast cancer, LABC: Locally advanced breast cancer, BCS: breast conservation surgery).
There was rising trend in the proportion of patients opting for conservation over the years, from 11.9% before the year 2000 to 30.7% in the past five years. This is shown in Fig. 2. The number of LABC patients undergoing breast conservation surgery (BCS) post Neo-adjuvant chemotherapy (NACT) increased from 5.2% before the year 2000 to 17.2% after that.
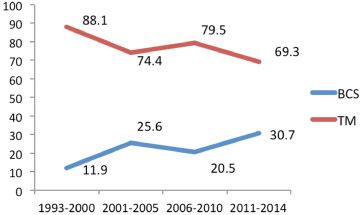
Time trends in surgical treatment of primary lesion. (BCS: breast conservation surgery, TM: Total mastectomy).
The data on management of axilla was present in 1032 patients, 81% of whom underwent axillary lymph node dissection (ALND). Sentinel Lymph Node Biopsy (SLNB) was done in 19% as a standalone procedure or followed by ALND if SLNB was positive, no sentinel node found, clinical suspicion of non-sentinel metastatic axillary nodes or as a part of validation phase of SLNB. Stagewise, more SLNB were performed in EBC patients (26 versus 9.3%;
Figure 3 depicts the time trends in the proportion of patients undergoing SLNB. There has been more than fourfold increase in the percentage of patients undergoing SLNB.
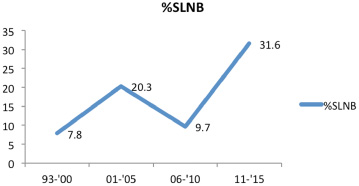
Time trends in SLNB (sentinel Lymph node biopsy).
The overall survival (OS) for all stages at 5 and 10 years were 61.5 % and 49.5% respectively. Disease free survivals (DFS) at 5 and 10 years were 59.5% and 47.3%. The relative hazard ratios for overall survival considering EBC group to have a hazard of 1 were 2.414(1.944–2.998;
In our study of 1310 breast cancer patients, the median age was 47 years. This younger age at presentation is in concordance with most of Indian studies, which report most common age to be between 46 and 49 years [4,8]. Mir et al reported mean age to be 42 years [9]. This is in contrast to reports from SEER data, which show median age in the US to be 61 years [10]. We also noted almost 10% of patients were aged less than 35 years, similar to previously reported figure of 11% [11]. The reason for younger age at presentation in Indian patients remains enigmatic and may be due to younger population structure, differences in tumor biology, genetic makeup or ethnic differences. Male breast cancer was seen in 1.1% cases, which is comparable to the published Indian and American literature [12–16].
The median age of menarche was 14 years. While early menarche at age <12 years constitutes a high risk for developing cancer [8], only 0.82% of our woman attained menarche at age younger than this. In other Indian studies, the mean age of menarche has been shown to be 12.9 years [9] and 13.2 years [17] respectively. Our result is not corroborative with most of the Indian literature with our patients having a median age of menarche as 14 years.
Nulliparous women constituted only 8.3% and median parity was three. In a similar study by Saxena et al, only 1.1% of patients were unmarried and 0.7% nullipara [18]. Thomas et al reported that none of their patient was nullipara [19]. The mean age of menopause was 45.6 years with over 45 % of our patients being pre-menopausal at presentation. Previous study by Raina et al reported 50% of patients as pre-menopausal [20]. Thomas et al reported around 70% of their patients to be pre-menopausal [19]. This is in contrast to the western data where premenopausal breast cancer constitutes around 15–20% of the cases only [18]. Despite the absence of most of reproductive risk factors, we continue to see an increasing incidence of breast cancers in our country, which calls for the need to assess other factors contributing to this.
All our patients were symptomatic with most common symptom being painless breast lump, similar to many other Indian studies [17,20]. Screen detected cancers are a rarity in India and even isolated nipple discharge or pain without a palpable lump are uncommon presentation [21]. Left sided predominance and higher incidence of disease in the upper outer quadrant were seen, similar to that reported by other authors [22–25].
Family history of breast cancer was noted in 4.5% of our patients. Agarwal et al also reported 5% cases having positive family history [6]. Studies from outside India have also reported familial breast cancers to comprise 5–10% of the total. In a review by Kochhar, studies report varying prevalence of family history [26]. The variations in the reported incidence of family history in the literature may be related to detail in which history is taken.
Only 4.65% of our patients had stage I disease. This is in contrast to SEER data showing 62% of patients having stage I disease [10]. In UK and Sweden, stage I and II make 90% [27,28]. Kemperman et al have reported 58% of the patients Stage I disease [29]. In most Indian studies, less than 10% of the tumors of EBC group were less than 2 cm [30–32] while more than 70% of the tumors in the U.S (2010 statistics) were are less than 2 cm [10]. Even in NSABPB32 trial, 84% tumors were less than 2 cm [33]. The lack of breast cancer awareness, breast self-examination practices, formal screening program, unfavorable socioeconomic circumstances, and poor access to health care facilities are some of the factors which contribute to Indian women presenting with advanced disease. Over 57% of our patients had palpable axillary nodes. At another Indian center, axillary nodal metastasis was seen to be present in >50% of patients with T1 and T2 disease and 2/3rd of LABC patients [6]. In another Indian study, 80% cases treated over a decade had disease in axillary lymph node basin [18]. In contrast to this, more than two thirds of the patients in west are node negative [10]. It may be pertinent to mention that most treatment recommendations and guidelines emanate from the west where patient profile and disease burden of patients is quite different from our patients. Extrapolating their results onto our patient population doesn’t always translate into similar results. The outcome of trials such as Z-0011 [34] where average tumor size was 1.7 cm in the ALND group (and 1.6 cm in SLND group) and where all patients were T1 or T2 tumors may not be applicable to early breast cancer patient population in our setup. In our study, only 6% of patients had tumors less than or equal to 2 cm in size. Thus caution should be exercised while counseling the patients based on western trials.
The most common histopathological type of breast cancer was infiltrating ductal carcinoma, no specific type, which comprised 87.3%. Saxena reported 88% of all tumors to be IDC [18]. Other studies report an incidence varying from 40–75% of total breast cancers [35]. Rosai et al. report an incidence of 2% for colloid carcinoma, which was seen in 1% of our cases [36]. In our study only 2% of all tumors had
We observed Estrogen receptor positivity in 53.2% of our patients. This was comparable with the results ranging from 49–57% reported by other studies [37,38]. These results are much lower than the western data where ER positivity is observed in 80% of the patients [10]. We observed that there was a trend of increasing steroid receptor expressing tumors as the age at presentation advances. 61.8% of tumors in patients less than forty had hormone receptor expression as compared to 86% in patients older than seventy years. This may also explain the higher receptor positivity in patients of the west since their breast cancer patients are older than ours. Of tumors on whom results of all three receptors were known, 33.9% were TNBC. In the U.S SEER population only 15% had triple negative breast tumors [10]. This difference may be explained on the basis of age at presentation, as higher proportions of younger patients are known to have triple negative breast cancers.
Most of our patients (77%) underwent mastectomy. This is similar to another study reporting that only 11.3% cases underwent BCS, while MRM was performed in 88.7% [20]. The scenario is different in the west where mostly patients undergo BCS. The trend is now changing in major centers in India, including ours, where the number of patients opting for BCS is increasing, though still lower than the west [5]. Similarly the trend of using SLNB, which has become the standard of care worldwide for non-metastatic clinico-radiologically negative axilla, is now increasing as shown in our results. However, this trend is not reproducible for a large number of Indian centers as the facility to perform the SLNB using dual agent technique with a blue dye and radio-colloid is available at selected centers only in India.
The survival outcomes of our non-metastatic patients are comparable to other Indian literature. However, shown in table 8, the stage wise survival rates for our patients are still lower than those of the west. This may stem from the fact that our tumor size and overall disease burden mostly falls in upper range of each stage. The low survival value in stage IV disease in our study as compared to the literature may be because of the fact that ours is a surgical unit and almost all upfront metastatic patients are referred to oncology unit for further care. This often leads to suboptimal data updation and loss to follow up of patients with us despite best efforts.
Survival outcomes (OS: overall survival)
Survival outcomes (OS: overall survival)
This study is one of the largest studies available in the Indian literature stressing upon the patient profile of breast cancer along with the tumor characteristics like pathology and treatment trends. Though lack of complete data in all the fields is inherent to any retrospective study, our study still manages to provide most of the data in all the fields as the data has been prospectively maintained in the clinic files and all efforts are made to ensure regular follow up of patients is there. All attempts are made to contact patients by letters/phone calls/email (as appropriate) in case patients consecutively miss multiple follow up visits.
Breast cancer in the Indian scenario is a disease of younger woman who lack the characteristic reproductive and demographic risk factors. Our patients present with larger tumors, advanced stage and a higher proportion have triple negative disease. These facts must be taken into account before extrapolating the results of western trials to our patient population. There is a strong need to study the clinico demographic risk factors and characteristics of our own population.
Funding
None.
Conflicts of Interest
None.
