Abstract
INTRODUCTION:
Inflammatory Breast Cancer (IBC) is a distinct and rare type of breast cancer accounting for up to 6% of all breast cancer cases in Europe. The aim of this study was to investigate diagnostic methods, treatments, and outcome after IBC in patients treated at a single institution in Denmark.
METHOD:
All patients treated for IBC at Aarhus University Hospital between 2000 and 2014 were identified and included in the cohort. Survival was assessed using Kaplan-Meier curves and log-rank statistics.
RESULTS:
A total of 89 patients were identified with a median follow up of 3.6 years. The overall survival at 5 and 10 years were 41% and 18%, respectively. The disease free survival at 5 and 10 years were 47% and 27%, respectively. Thirty-four percent had distant metastasis at time of diagnosis. Patients with ER positive tumors had a significantly better overall survival than patients with ER negative tumors (p = 0.01).
CONCLUSION:
Despite a more aggressive systemic and loco-regional treatment today, IBC is still a very serious disease with a high mortality.
Introduction
Inflammatory Breast Cancer (IBC) is a distinct type of breast cancer which accounts for up to 6% of all breast cancer cases in Europe [1,2]. Despite of its rareness IBC represents a significant burden of morbidity and mortality.
Sir Charles Bell was the first to draw attention to the condition of IBC in 1814. He described the clinical presentation with the words: “Like a purple colour on the skin over the tumor accompanied by shooting pains”.
IBC tends to grow and spread rapidly, often with exacerbation of symptoms within months, weeks or even days. Approximately 30% of patients with IBC have distant metastases at the time of diagnosis [3]. IBC is a clinical diagnosis characterized by warmth, edema (peau d’ orange), and with discoloration affecting at least one third of the breast. A palpable distinct mass is often not found [4,5].
The lack of uniform and precise diagnostic criteria for IBC makes it difficult to investigate the prognosis of IBC. Previous studies indicate that patients who are able to complete a full course of treatment, including neoadjuvant chemotherapy, mastectomy, and radiation have better overall survival (OS) [6].
Studies have found that IBC most often affect middle aged women, have worse outcome compared to early breast cancer, more ER negative tumors, overexpression of Rhoc-GTPase oncogene, loss of expression of WISP3 gene, high lymph-angiogenesis, high chemokines, and persistent expression of E-Cadherin [5].
There are no particular pathological diagnostic criteria or specific histological subtype for IBC. There are, however, some important pathological findings that in conjunction with the clinical characteristics can help to support the diagnosis IBC [5,7].
The classic histological finding in IBC is the presence of dermal lymphatic invasion, often localized in the papillary and the reticular dermis of the skin overlying the breast, with numerous dilated dermal lymphatic vessels filled with small tumor emboli [5,8]. However, only 75% of patients with the clinical characteristics of IBC have dermal tumor emboli [5,8], and therefore dermal tumor embolism is not a mandatory diagnostic criteria. The emboli typically consist of high-grade tumor cells with a ductal phenotype (Fig. 1). The IBC tumor may or may not form a distinct mass, consequently in many cases there are difficulties assessing the size of the tumor [7]. Furthermore, the relationship between the size, the number of the emboli, and the clinical inflammatory skin involvement is weak [5,9].
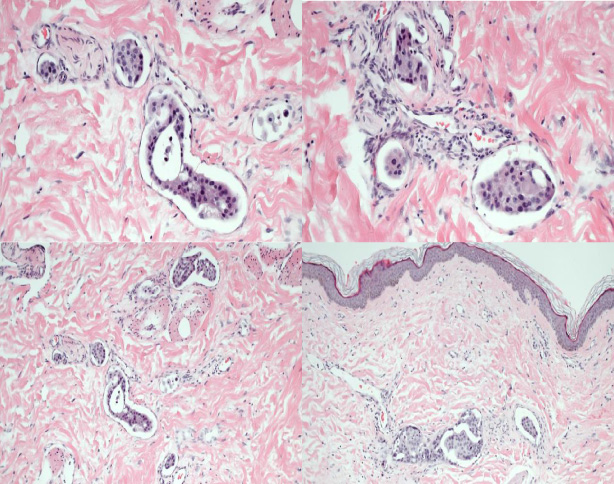
Tumoremboli in lymphatic vessels in the dermis.
In IBC the clinical findings are important, but the diagnosis also requires imaging and biopsy. Imaging techniques include mammography, ultrasonography, magnetic resonance imaging (MRI), and positron emission tomography-computed tomography (PET-CT) [5]. Mammography and breast ultrasound are the most frequently used investigations, and especially ultrasound plays an important role in the localization of a mass prior to biopsy.
In mammography the most frequent inflammatory changes seen are extensive skin and trabecular thickening of the parenchyma and/or augmented breast density.
Ultrasound shows commonly high echogenicity of the breast tissue, the presence of tumor or mass and lymphatic vessels dilatation. These findings are sometimes associated with pathological lymph nodes of the axilla and the neck.
Often IBC does not present with a distinct lump, but with changes to the skin. Therefore a skin punch biopsy is often used to assist the diagnosis.
In the last decade breast MRI has become the most reliable test for gathering more information about IBC. This imaging technique together with mammography and ultrasound is also useful for the evaluation of the treatment response [5].
The MRI images often visualize a mass or multiple masses, small and confluent with irregular margins, and internal diffuse, heterogeneous enhancement pattern. MRI also shows skin thickening and oedema in most cases.
Once IBC is diagnosed, additional tests are used to determine the staging. Tests that may be used include chest X-ray, CT scan of the chest, abdomen, and pelvis, bone scan, and liver function tests.
The aim of this retrospective single institutional study at Aarhus University Hospital in Denmark was to obtain more knowledge of diagnosis, treatment, and outcome of IBC.
Patients diagnosed with IBC at Arhus University Hospital from 2000–2014 were included. Patients were identified from the population-based registry of the Danish Breast Cancer Group (DBCG). The records of all identified patients were reviewed to include only those having IBC criteria fitting the definitions of the American Joint Committee on Cancer AJCC (2010): “IBC is a clinical-pathologic entity characterized by diffuse erythema and edema (peau d’orange), often without an underlying palpable mass, involving one-third or more of the skin of the breast. The diagnosis is based upon the clinical presentation”. The histological verification of carcinoma was made by a core and punch biopsy.
Information on tumor size, lymph node involvement, estrogen receptor (ER) status HER2 status, histological subtype, and the date of biopsy was obtained from the DBCG database and the pathology registry If information was missing the patient records were reviewed to ensure completeness of data.
Data regarding age, survival, metastases at diagnosis, recurrence status, systemic therapy, surgical treatment, and radiotherapy (RT) was acquired from patient records.
According to Danish and international guidelines, IBC is included in the group classified as locally advanced breast cancer (LABC), which are not suitable for surgery up front. Standard treatment recommendation is neoadjuvant systemic treatment preferably with chemotherapy. Patients who were considered operable after systemic therapy underwent surgical treatment consistent of mastectomy and axillary lymph node dissection.
In Denmark three different chemotherapy regimens has been used since 2000:
From 2000 to the spring of 2007: CEF (Cyclophosphamide 600 mg/m2, Epirubicin 90 mg/m2, 5-Fluorouracil 600 mg/m2) × 4 cycles - mastectomy and axillary dissection - followed by CEF × 4 cycles and RT and endocrine therapy if indicated. From spring 2007 to October 2010: CE (Cyclophosphamide 600 mg/m2, Epirubicin 90 mg/m2) × 4 cycles - mastectomy and axillary dissection - followed by Docetaxel, 100 mg/m2 × 4 cycles and RT and endocrine therapy if indicated. From October 2010 to 2015: CE (Cyclophosphamide 600 mg/m2, Epirubicin 90 mg/m2) × 4 cycles - followed by Docetaxel (100 mg/m2) × 4 cycles, mastectomy and axillary dissection – followed by RT and endocrine therapy if indicated.
The targeted HER2 treatment was introduced in 2007 and consists of trastuzumab (4 mg/kg loading dose, followed by 2 mg/kg every three weeks for 17 cycles) initiated at the same time as docetaxel treatment is started.
Target for RT was the chest wall and axillary lymph nodes level I–IV and internal mammary lymph nodes in most patients. The dose was 50 Gy in 25 fractions.
Local recurrence (LR) was defined as recurrence at the chest wall. Regional recurrence (RR) was defined as recurrence in the regional axillary lymph nodes (level I–IV) or internal mammary lymph nodes.
Recurrence outside the loco-regional ipsilateral areal or in the contralateral breast was defined as distant metastases (DM).
Statistical analysis
The Kaplan–Meier method and the log-rank test was used to evaluate overall survival. All statistical tests were two-tailed and p-values <0.05 were considered significant. Overall survival (OS) rates were calculated on the basis of the period from the time from diagnosis to the time of death or to 31st December 2014. Disease free survival (DFS) was defined as survival without breast cancer recurrence, death, or, if the patient was still alive, to last follow up. Statistical analyses were performed using Stata version 12 [10].
The study was approved by the Danish Data Protection Agency.
Results
A total of 89 women were eligible for the study. Clinical and pathological characteristics are presented in Table 1. The mean follow-up was 3.6 years (range 0.1–12.3). The mean age at diagnosis was 60.9 years (range 28.1–91.5).
All 89 patients had a core biopsy at diagnosis. The majority was invasive ductal carcinoma (79%), four patients had invasive lobular carcinoma (5%), and the remainder (17%) had poorly differentiated tumors and could not be assigned a histological subtype (Table 1).
Clinical & Pathological Characteristics (N = 89)
Clinical & Pathological Characteristics (N = 89)
Forty patients (45%) were HER2 positive and 56 patients (63%) were ER positive. Eleven patients (12%) were both ER and HER2 negative (Table 2). Among those who underwent surgery 61% were ER positive.
Subtyping (N = 89)
Thirty patients presented with both inflammatory cancer and verified DM (Fig. 2), of these did 76% receive chemotherapy, 70% RT, 36% trastuzumab, and 50% endocrine therapy.
DM = Distant metastases.
(a) Disease Free Survival (DFS) among patients receiving radical surgery.
Fifty-nine patients had purely IBC without DM at time of diagnosis and were eligible for surgery after neo-adjuvant therapy (85% chemotherapy, 98% RT, 41% trastuzumab, 58% endocrine therapy).
Four patients refused surgery, one did not finish neo-adjuvant therapy prior to last follow-up, and three patients had progression during neoadjuvant chemotherapy (NACT) and were never able to undergo surgery. Hence, 51 patients out of 59 (86%) with pure IBC at time of diagnosis had surgery after neoadjuvant chemotherapy.
Among the 51 patients with pure IBC operated following NACT, we found a pathological complete response (pCR) in 10 patients. The remainder had no response or only discreet response, among these 31 had radical surgery.
Thirteen of the 30 patients with DM at time of diagnosis responded well to neoadjuvant chemotherapy and were offered surgery as a palliative procedure.
Thus, in total 64 patients out of 89 patients (72%) underwent surgery with concomitant axillary lymph node dissection after NACT. Among patients older than 70 years, 50% underwent surgery. In patients younger than 70 years 81% underwent surgery.
Among the 64 patients receiving surgery and axillary dissection, 56 patients (88%) were lymph node positive (Table 3). Most patients (n = 41) had more than three positive nodes with 15 (23%) having more than 10 positive nodes.
Lymph node status (N = 64)
Lymph node status (N = 64)
Further systemic treatment and radiotherapy was given to most patients (Table 4). RT was given to 72 out of 89 patients (81%). Mainly patients receiving surgery had RT (95% compared to 44% among those not having surgical treatment).
Inflammatory breast cancer. Post-surgical treatment in patients having surgery after primary systemic treatment with chemotherapy
Percentages from within collums.
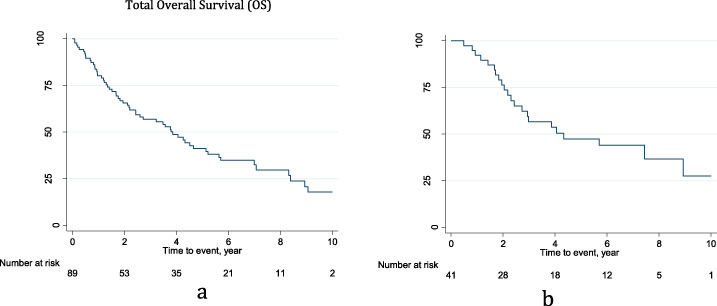
For the total cohort of patients presenting with inflammatory breast cancer the OS rates at 2-, 5-, and 10-year were 66%, 41%, and 18%, respectively (Fig. 3a,b).
ER positive patients had a significantly better OS than ER negative patients: at 2 years 74% versus 51% and at 5 years 49% versus 27% (p = 0.01) in the total cohort (Fig. 4). Likewise, DFS was higher among ER positive patients: at 2 years 87% versus 52% and at 5 years 61% versus 40%, although not significant (p = 0.14).
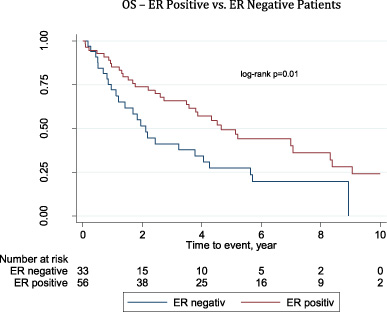
Overall survival - ER positive vs. ER negative patients.
OS among patients with pure IBC without DM who underwent surgery (N = 51) at 5-years and 10-years was 57% and 29%, respectively, which is significantly better than in patients presenting with DM (p < 0,001). The ER positive patients in this group had a significantly better OS at both 5 and 10 years compared to ER negative patients: at 5-year OS 73% and 30%, respectively, and a 10-year OS at 40% and 0%, respectively (p = 0.001 - Fig. 5). We observed a 100% OS, within the first 5 years, in the group of 10 patients with pCR as opposed to 48% in the group without pCR (p = 0.02).
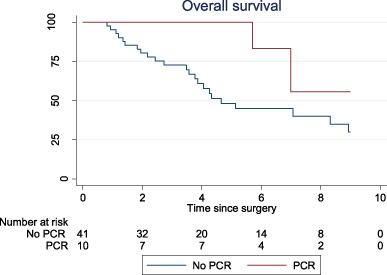
Overall survival - No pCR vs. pCR.
Nine out of the 41 radically operated patients developed recurrence, of these 33% LR, 44% DM, and 22% contralateral breast. DFS among these patients was 76%, 47%, and 27% at 2-, 5- and 10 years, respectively (Fig. 3a).
No significant difference in OS was observed based on age (p = 0.11), tumor size (p = 0.36), lymph node involvement (p = 0.19), or HER2-status (p = 0.86).
This study confirms the aggressive nature and poor prognosis of IBC The tumor characteristics were unfavorable in a large proportion of patients with 45% being HER2 positive, 33% presenting with DM, and only 57% of the patients being were able to undergo radical surgery. The total 5-year OS was 41% and for patients who received radical surgery after neoadjuvant treatment the 5-year OS was 57%.
Patients with IBC have been described as somewhat younger than patients with other types of breast cancer. Schairer et al. [11] found a mean age at diagnosis of 57.5 for IBC vs. 64.6 for non-IBC and Schlichting et al. [12], estimated a mean age of 57.1 for IBC vs. 57.8 for non-IBC at stage III. However, Goldner et al. [4] found that IBC affects older women, and that the incidence of IBC rises sharply with age and reaches a plateau after the age of 65. Our study estimates a mean age of 60.9 years, very close to what is seen in early breast cancer in Denmark (http://dbcg.dk/PDF%20Filer/aarsrapport_2017_final.pdf).
As Bates et al. [13] previously reported, the present study shows no significant age dependency of survival.
The present study estimated a total 5-year and 10-year OS at 41% and 18%, respectively and among patients with radical surgery and no DM at time of diagnosis, a DFS at 47% and 27%. One other study estimated survival rates in similar ranges [14]. However, several other studies reported better OS and DFS [13,15] (Table 5).
Overview of studies reporting survival
Overview of studies reporting survival
Dawood et al. [16] found a clear improvement in breast cancer specific survival over a period of two decades. This suggests an improvement in the management of IBC. The small number of patients in our study makes it difficult to predict and comment on long-term survival estimates. However, according to Jensen et al. [17] the 5-year OS for local advanced breast cancer in Denmark was 41% in 2009 compared to 28% in 1999.
Before the introduction of systemic neoadjuvant chemotherapy, the prognosis of IBC was considerably worse than today. In a study from 1981 Bozzetti et al. [19] found that less than 5% of the patients survived for more than 5 years after treatment with surgery and/or radiotherapy. Doxorubicin-based therapy was the first chemotherapy introduced in the neoadjuvant treatment of IBC and it contributed to an improvement of OS [18–20]. One study has described OS rates of 44% at 5-years and 32% at 10-years in patients treated with either 3 cycles of neoadjuvant CAF (cyclophosphamide, doxorubicin, and 5-fluorouracil) or CEF (cyclophosphamide, epirubicin, and 5-fluorouracil) followed by surgery and/or adjuvant RT [21]. Further improvement was observed by Christofanilli et al. [22,23] after introduction of taxane-based therapy. Thus, anthracycline-based therapy combined with a taxane increased the response rate to primary systemic chemotherapy and improved prognosis (median OS: group 1, 32 months; group 2, 54 months; p = 0.03; median PFS (Progression free survival): group 1, 18 months; group 2, 27 months; p = 0.04). Thus, the prevailing evidence tells that chemotherapy is an important component in initial treatment of IBC. In the present study, 72% of patients received primary systemic chemotherapy.
Today the recommended surgical procedure for patients who respond to neoadjuvant chemotherapy is mastectomy with axillary lymph node dissection. Breast conserving surgery is not recommended in patients with IBC [15,24,26].
Anderson et al. [27] found that 55–85% of the patients with IBC had lymph node involvement and Matro et al. [28] found that more than 80% had N1-N3 disease at diagnosis. The present study found in line with these studies that 88% of the patients undergoing surgery after chemotherapy had lymph node involvement at diagnosis and that 41% had more than six positive lymph nodes. This underlines that complete axillary lymph node dissection is an important standard care for IBC patients.
Despite the small number of patients included in our study, we observed a 100% OS within the first five years in the pCR group compared to 48% in the group not having a pCR.
Among patients without DM at time of diagnosis, there were a significant better OS in the group of patients having surgery compared to the patients without surgery (Fig. 6). Two other studies [13,29] found no evidence of a survival benefit with surgery. However, Aborus-Anane et al. [29] found a significant improvement in loco-regional control, but no significant difference in OS or DFS rates.
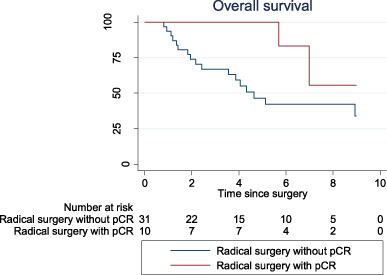
Overall survival - Radical surgery without pCR vs. Radical surgery with pCR.
RT seems to play an important role in the loco-regional management of IBC. Adjuvant RT is recommended for all women with IBC who undergo mastectomy [7]. RT schedules and doses vary among institutions but cumulative doses are usually between 55–66 Gy [7,14]. In the present study, all the patients who underwent surgery received loco-regional RT.
Several studies [30,32] show that HER2 is overexpressed or amplified in 35–60% of patients with IBC. In this study 45% of patients were HER2 positive. Trastuzumab is proved to have significantly effect on the survival outcomes in patients with HER2 positive LABC [33,35]. A prospective study that randomized women with LABC, including IBC, to an anthracycline-based chemotherapy with or without one year of trastuzumab shows that patients receiving trastuzumab had a significantly better pCR (38% versus 19%, p = 0,001) and event-free survival (3 year event-free survival 56% versus 7%, p = 0,013) [36].
Previous reports found that tumors in patients with IBC predominantly were ER hormone-receptor negative compared to non-IBC.
Turpin et al. found that 40–50% of the patients had ER positive tumors. In this study 63% of the patients were ER receptor positive, but the fraction of ER-negative tumors was much higher than seen in early breast cancer [17]. Our study showed no significant improvement in DSF for patients who were ER positive compared with patients who were ER negative, and only a slightly better OS.
IBC have a highly metastatic potential presumably because of the lymphatic involvement. It is well known that 20–50% of women have metastasis at the time of diagnosis, which emphasizes that IBC is an aggressive cancer type. In this study 34% had metastatic disease at time of diagnosis.
Harris et al. [15] found a 5-years DFS-rate at 59% in patients with a pathological complete response compared to 45% in patients with either gross or microscopic residual disease in the breast and/or regional nodes at the time of mastectomy.
The registration methods changed over the years, which may have constituted selection bias. In order to minimize this patient records have been investigated carefully to ensure a correct diagnosis. We are aware that the number of patients is quite limited
We are not able to make relevant conclusions regarding the treatment and follow-up of IBC in Denmark due to small number of patients. The diversity of treatment management in IBC makes comparison of different clinical trials and studies challenging. It is important to define a set of international guidelines regarding diagnostic criteria as well as a treatment regime. This will make it more expedient to compare different studies worldwide and obtain more knowledge of the nature of IBC.
This study shows that IBC is an aggressive type of breast cancer of which one third have distant metastasis at diagnosis. In the two thirds of patients with pure locally advanced disease who are eligible for treatment with curative intend the prognoses is significantly worse than in “early breast cancer”.
