Abstract
BACKGROUND:
Trastuzumab (Tz) is assumed to prime antibody-dependent cellular cytotoxicity (ADCC); however, it remains unclear whether Tz therapy can clinically induce adaptive cellular immunity.
OBJECTIVE:
Adaptive Cellular Immune Effect of Tz Therapy.
METHODS:
This study included 29 surgical invasive breast carcinomas administered neoadjuvant chemotherapy with Tz (15 cases) or without Tz (14 cases). The numbers of immunoreactive cells (CD4, CD8, CD56, and Fox-P3) in three different compartments (intratumoral, adjacent stromal, and distant stromal) were determined.
RESULTS:
The average number of adjacent stromal CD4-positive, CD8-positive, and Fox-P3-positive cells in the Tz+ group was significantly greater than that in the Tz− group (p = 0.036, 0.0049, and 0.043, respectively). However, the number of Fox-P3-positive cells was much less than that of CD4-positive cells. Moreover, distant stromal CD4-positive and CD8-positive cells in the Tz+ group was also significantly greater than that of the Tz− group (p = 0.029 and 0.032, respectively). Only a small number of CD56-positive natural killer cells, playing a main role in ADCC, accumulated at the tumor site after Tz therapy.
CONCLUSIONS:
The results suggest that Tz therapy induces adaptive cellular immunity, including infiltration of both CD4-positive helper T cells and CD8-positive cytotoxic T cells into the breast carcinoma lesion.
Introduction
Trastuzumab (Tz) therapy notably improves the prognosis of patients with HER2/neu-positive breast carcinomas, and it has been recognized as a standard therapy for patients with HER2-positive breast carcinomas [1]. Antibody-dependent cellular cytotoxicity (ADCC) is believed to mediate the effects of Tz [2,3]; however, adaptive anti-tumor cellular immunity is reported to be an important anti-tumor mechanism in Tz therapy [4–7]. Little is known about the anti-tumor immunological mechanisms of Tz therapy in carcinoma patients.
Recently, the importance of tumor-infiltrating lymphocytes (TILs) in various types of carcinomas, which may indicate immune-mediated host defences against the tumor, has been recognized. In breast carcinomas, a large number of infiltrating lymphocytes in a tumor correlate with improved overall survival and recurrence-free survival [8,9]. Moreover, TILs serve as a predictive marker of response to neoadjuvant chemotherapy (NAC) for breast carcinomas [10–14]. Denker et al. reported that intratumoral lymphocytes in pretherapeutic breast carcinoma specimens were a significant independent predictor of pathological complete response [14]. Further, high numbers of TILs in biopsy specimens has been significantly associated with a reduction in the distant recurrence rates of triple-negative breast carcinomas [10]. In addition, high levels of TILs have been significantly associated with a reduced recurrence rate in HER2-positive breast carcinoma patients treated with Tz [10].
Specific subpopulations of lymphocytes are also thought to be associated with a pathological complete response to NAC [15,16]. The presence of CD8-positive cells has been associated with a significant reduction in the relative risk of death in both ER-negative and ER-positive/HER2-positive breast carcinoma patients [15], and infiltration of CD8-positive cells and a high CD8/Fox-P3 ratio were significantly associated with pathological complete remission in triple-negative breast carcinoma cases [16]. However, it remains unclear whether Tz therapy can clinically induce adaptive cellular immunity. Here, we compare the number of cells in subpopulations of TILs in primary breast carcinomas after NAC, with or without Tz therapy.
Materials and methods
This research protocol was approved by the ethics committee of Shiga University of Medical Science (29-001).
Patients
This study consisted of 29 consecutive surgical cases of primary invasive breast carcinoma treated with NAC at the Shiga University of Medical Science Hospital between 2011 and 2014. All cases were diagnosed as invasive carcinomas by biopsy specimens before NAC, and the presence of estrogen and progesterone receptors and the HER2 status of biopsy specimens was determined. Three patients who showed complete remission with NAC were not included in this study. Tz therapy was added to the regimen in HER2-positive cases with immunohistochemical scores of 3+ or 2+ and fluorescence in situ hybridization scores of ≥2.2. Twelve HER2-positive patients were treated sequentially as NAC with docetaxel + Tz and a combination of 5-fluorouracil, epirubicin, and cyclophosphamide (FEC). Three HER2-positive patients were treated with NAC with docetaxel + Tz. All 14 HER2-negative patients were treated sequentially with NAC with the regimen of docetaxel and FEC.
Magnetic resonance imaging (MRI) scans were performed before NAC and immediately before breast cancer surgery after NAC. The tumour reduction rate was calculated with the longest diameter for the primary tumour scanned from the MRI image after NAC relative to the baseline before NAC.
Histopathological evaluation following NAC
The following Miller-Payne grading system was applied to evaluate the effects of NAC [17]: Grade 1, no change or some alterations in individual malignant cells, but no reduction in overall cellularity; grade 2, minor loss of tumor cells (up to 30%), but overall cellularity still high; grade 3, an estimated 30–90% reduction in tumor cells; grade 4, a marked disappearance (more than 90% loss) of tumor cells, such that only small clusters or widely dispersed individual cells remain; and grade 5, no malignant cells identified in tumor sections, only the vascular fibroelastic stroma (often containing macrophages) remains, and an intraductal component may be present.
Immunohistochemistry
Formalin-fixed, paraffin-embedded tissue blocks of the resected breast specimens were cut into 3-μm thick sections, deparaffinized, and rehydrated. Each section was stained with hematoxylin and eosin, and then used for immunostaining. Immunohistochemical analyses were performed using an autostainer (Benchmark XT system; Ventana Medical System, Tucson, AZ). The primary antibodies used in this study were as follows: a rabbit monoclonal anti-CD4 antibody (clone SP35, Ventana), a mouse monoclonal anti-CD8 antibody (clone 1A5, Novocastra Laboratories Ltd., Newcastle upon Tyne, UK), a mouse monoclonal anti-CD56 antibody (clone CD564, Novocastra), and a mouse monoclonal antibody anti-Fox-P3 (clone mAbcam 22510, Abcam, Cambridge, UK). Tonsil tissue was used as a positive control for CD4, CD8, CD56, and Fox-P3 lymphocytes, and primary antibodies with similarly diluted nonimmunized mouse serum were used in negative controls.
Immunostaining evaluation
The number of CD4-positive, CD8-positive, CD56-positive, and Fox-P3-positive cells in five non-overlapping high-power fields (400×) was counted. The numbers of positive cells were counted in three different compartments of each case, according to the recommendation of the International TILs Working Group [18]. Positive cells in the intratumoral compartment were defined as immunoreactive cells within carcinoma cell nests or in direct contact with tumor cells, whereas positive cells in the adjacent stromal compartment were defined as immunoreactive cells in the tumor stroma without direct tumor cell contact [14,18]. We also evaluated immunoreactive cells in the stromal compartment distant from the carcinoma. The average number of immunoreactive cells was calculated in each compartment in all cases.
Statistical analyses
Statistical analyses were performed using the 𝜒2 test, Welch’s t test, Mann-Whitney U test, and the Pearson product-moment correlation coefficient. A p-value of <0.05 was considered statistically significant.
Results
Clinicopathological features
Table 1 summarizes the clinicopathological features of the 29 cases included in this study. The patients were divided into two groups, the NAC with Tz therapy (Tz+ group, 15 cases) and the NAC without Tz therapy (Tz− group, 14 cases). All of the patients were females, and the average age in the Tz+ group was 55 years (range, 42 to 72 years) and that of the Tz− group was 50 years (range, 35 to 64 years). All patients underwent surgical axillary lymph node dissection after NAC. An average interval between the last NAC and the breast surgery was 28.2 days ± 6.1 days (range, 19 to 36 days) in Tz+ group and 26.8 days ± 5.1 days (range, 21 to 39 days) in Tz− group.
Clinicopathological features and relationships of each group
Clinicopathological features and relationships of each group
Tz, Trastuzumab.
There were no significant differences in hormone receptor status, tumor size, lymph node metastases, clinical stage, NAC regimen, or histopathological effect of NAC between the Tz+ and Tz− groups. One case in the Tz− group was classified as HER2-positive, because the operative specimen showed a HER2 score of 3+, despite a HER2 score of 0 for the biopsy specimen. Therefore, this case was classified as HER2-positive, and NAC without Tz was administered.
Figure 1 shows cells in breast carcinoma specimens that were positive for CD4 (a), CD8 (b), CD56 (c), and Fox-P3 (d) antigens.
There were no significant differences in the CD4, CD8, and FOXP3 in the tumor and the adjacent stroma of the biopsy specimen before NAC (Tz− 8 cases Tz+ 12 cases which could be scrutinized). Intratumoral CD4-positive cells averaged (three fields) from 0.2 to 8.4 cells in the Tz+ group and from 0.4 to 6.6 in the Tz− group (p = 0.069), and in the adjacent stroma, CD4-positive cells averaged between 13.8 to 186.6 in the Tz+ group and 4 to 46 in the Tz− group (p = 0.058). Intratumoral CD8-positive cells ranged from 1.8 to 49.8 in the Tz+ group and from 0 to 19.2 in the Tz− group (p = 0.139). In the adjacent stroma, CD8-positive cells averaged between 13.4 to 156.4 in the Tz+ group and 3.4 to 74.8 in the Tz− group (p = 0.129). Intratumoral Fox-P3-positive cells averaged between 0 to 2.8 in the Tz+ group and 0 to 2.8 in the Tz− group (p = 0.294). The number of adjacent stromal Fox-P3-positive cells ranged from 0.4 to 49.6 in the Tz+ group and from 1 to 34.3 in Tz− group (p = 0.411).
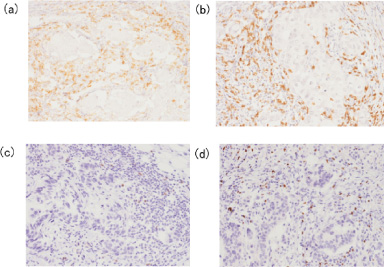
Immunohistochemical results. CD4-positive (a), CD8-positive (b), CD56-positive (c), and Fox-P3-positive (d) cells were observed in the adjacent stroma of breast carcinomas treated with neoadjuvant chemotherapy in conjunction with trastuzumab (200×).
Expression of CD4 and CD8-positive cells in each compartment
A, adjacent stromal compartment; D, distal stromal compartment; I, intratumoral compartment; Tz, Trastuzumab. The number is the average of 3 fields of view.
Expression of CD56, FOX-P3 and CD8/Fox-P3-positive cells in each compartment
A, adjacent stromal compartment; D, distal stromal compartment; I, intratumoral compartment; Tz, Trastuzumab.
Table 2 summarizes the results of the average number of immunoreactive CD4, CD8, CD56, and Fox-P3 cells in the intratumoral, adjacent stromal, and distant stromal compartments in both Tz+ and Tz− groups. Intratumoral CD4-positive cells averaged (three fields) from 0 to 4.4 cells in the Tz+ group and from 0 to 3.4 in the Tz− group, and in the adjacent stroma, CD4-positive cells averaged between 0.6 to 116.4 in the Tz+ group and 2 to 13.2 in the Tz− group; the distal stromal CD4-positive cells averaged between 0.8 to 117 in the Tz+ group and between 0 to 25.6 in the Tz− group. Intratumoral CD8-positive cells ranged from 0 to 7.2 in the Tz+ group and from 0 to 2.6 in the Tz− group. In the adjacent stroma, CD8-positive cells averaged between 1.4 to 230.6 in the Tz+ group and 0.8 to 34 in the Tz− group. CD8-positive cell counts in the distal stroma ranged from 0.6 to 63.6 in the Tz+ group and from 0.2 to 16.4 in the Tz− group. Intratumoral Fox-P3-positive cells averaged between 0 to 0.8 in the Tz+ group and 0 in the Tz− group. The number of adjacent stromal Fox-P3-positive cells ranged from 0 to 38.2 in the Tz+ group and from 0 to 0.2 in Tz− group, and distal stromal FOX-P3-positive cell counts were from 0 to 15.4 in the Tz+ group and from 0 to 1.6 in the Tz− group. CD56-positive cells were lower in general. The number of intratumoral CD8/Fox-P3-positive cells ranged from 1 to 5.5 in the Tz+ group, adjacent stromal CD8/Fox-P3-positive cells ranged from 0.4 to 29.8 in the Tz+ group and from 15 to 46 in the Tz− group, and distal stromal CD8/Fox-P3-positive cells ranged from 0.8 to 36.3 in the Tz+ group and from 1 to 18.5 in the Tz− group.
The average number of adjacent stromal CD4-positive, CD8-positive, and Fox-P3-positive cells in the Tz+ group was significantly greater than that of the Tz− group (p = 0.036, 0.0049, and 0.0043, respectively) (Fig. 2a). The average number of distant stromal CD4- and CD8-positive cells in the Tz+ group was significantly greater than that of the Tz− group (p = 0.029 and 0.032, respectively) (Fig. 2b). There were no significant differences in the numbers of intratumoral CD4-positive, CD8-positive, Fox-P3-positive cells between the Tz+ and Tz− groups (Fig. 2c). There were also no significant differences in the numbers of CD56-positive cells between the Tz+ and Tz− groups.
The average number of adjacent stromal CD4-positive and CD8-positive cells in the two groups were well correlated (Pearson product-moment correlation coefficients: r = 0.81 in both groups combined, r = 0.76 in the Tz+ group, and r = 0.66 in the Tz− group), and the number of intratumoral CD4-positive and CD8-positive cells were fairly well correlated only in the Tz− group (r = 0.64), whereas those in the combined groups (r = 0.36) and Tz+ group (r = 0.27) were not correlated. Moreover, the numbers of CD4-positive and CD8-positive cells in the distant stromal compartments were not correlated (r = 0.26 in total, r = 0.18 in the Tz+ group, and r = 0.24 in the Tz− group).
There was no significant correlation between CD4- and CD8-positive cells in each lesion and pathological effects (Grade 1, 2, 3, and 4); however, the number of infiltrated CD8-positive cells at the tumour lesion was significantly correlated with the tumour reduction ratio, and the correlation in patients without Tz therapy was stronger than in those with Tz therapy (Table 3).
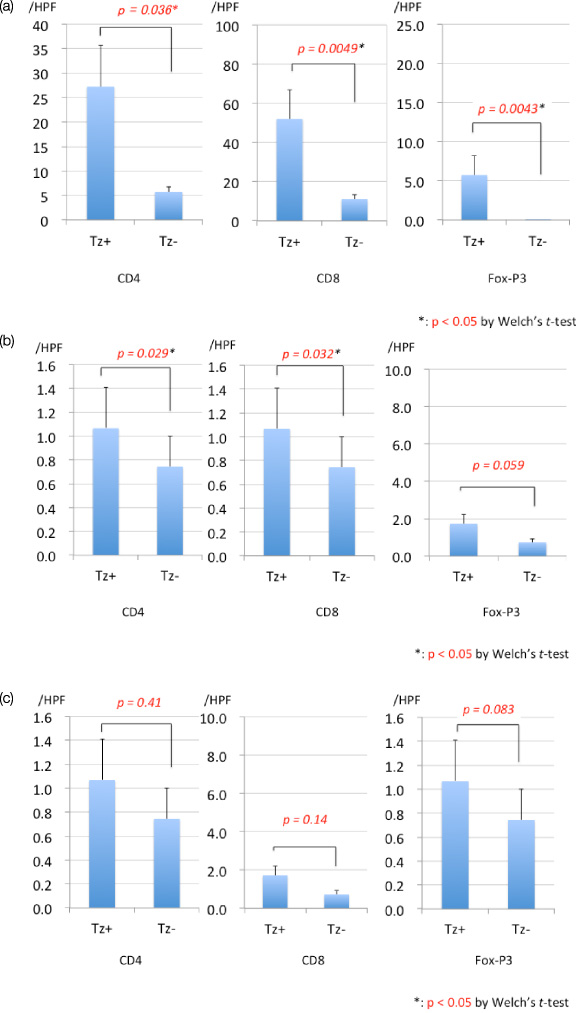
Number of CD4-, CD8-, and Fox-P3-positive cells in the three different tissue compartments, (a) adjacent lesion, (b) distant lesion, and (c) intratumor lesion, with or without trastuzumab therapy.
P values between CD8 expression and pathological effect or tumor reduction ratio
*Statistically significant, r; Pearson’s product-moment correlation coefficient, Tz; Trastuzumab.
Tz has had revolutionary effects on HER2-positive breast carcinomas in combination with chemotherapy [19]. The use for adjuvant and preoperative chemotherapies is expected to contribute even more to therapeutic efficacy in the future. Although the effects include ADCC, in addition to molecular targeting of the drug, we have shown, for the first time, that Tz can clinically induce adaptive cellular immunity at the breast carcinoma lesion. Tz is believed to have antitumor effects via ADCC, however, in the present study only a small number of CD56-positive natural killer (NK) cells, playing a main role in ADCC, accumulated at the tumor site after Tz therapy. This result would be caused by the disappearance of NK cells from the tumor site because of the long duration between the last Tz administer and the breast tumor resection (average duration was 28.2 days ± 6.1 days). In contrast, many CD8-positive cytotoxic T cells in cases with Tz therapy accumulated at the tumor site even after losing the direct effects of Tz, indicating that antitumor adaptive cellular immunity was generated after Tz therapy. Enhancement of anti-tumor CD8-positive T cell responses may be associated with a Fc-mediated activation of DCs and an increased antigen-presenting function to T cells [7].
In addition, many CD4-positive T cells accumulated at the tumor site after Tz therapy in this study. Most of these CD4-positive T cells should be helper-CD4 T cells that play a major role in generating an adaptive immunity, because in most cases with CD4-positice cell accumulation at the tumor site except case 8 (Table 2), number of CD4-positive T cells revealed much more than FoxP3-positive regulatory T cells that generally expressed CD4 on their surface.
Breast cancers with high numbers of intra-tumoural CD8+ T cells were reported to have favourable outcomes. In contrast, the presence of Fox-P3-positive regulatory T cells in breast cancers has been reported to be paradoxically associated with both reduced survival and improved survival [16]. These discrepancies may have been derived from several breast cancer subtypes with different biological characteristics and/or different therapeutic drugs [16]. In this study, increased infiltration of Fox-P3-positive cells in Tz-treated patients may be explained by the HER2-positive subtype of breast cancer and/or anti-HER2 therapy for HER2-positive breast cancers. In addition, statistically positive correlations were reported between CD8+ and Fox-P3-positive cell infiltration in each of the three TILs compartments [16]. Therefore, an induced immune reaction by Tz may accumulate both CD8+ cytotoxic T cells and Fox-P3-positive regulatory T cells in the tumour microenvironment.
Previous reports have shown that the presence of TILs is positively correlated with lymph node status and better prognosis [9], and that the presence of CD8-positive cytotoxic cells in breast carcinomas is associated with favorable outcomes [15,20], including a significant reduction in the relative risk of death. Both intratumoral and adjacent tumoral CD8-positive T cells were independently associated with a reduction in the risk of death from breast carcinomas in both HER2-positive and HER2-negative cases [15]. Taken together, these findings suggest that induced adaptive cellular immunity at the tumor site by the Tz therapy could affect antitumor effects and contribute to favorable prognosis.
There was no significant correlation between the number of infiltrated CD8+ T cells at the tumour site and pathological effect after NAC ± Tz therapy (Table 3). However, there was a positive correlation between the number of infiltrated CD8+ T cells and tumour reduction rate, particularly in patients without Tz therapy (Table 3). In the group treated with Tz, the weak correlation between the infiltration of CD8 T cells and pathological effect or tumour reduction rate may have been affected by the small number of infiltrated TILs when tumour shrinkage was large and the residual tumour was very small. Additionally, TILs could not be examined in 3 patients who received Tz therapy and achieved complete remission of the tumour.
There were several limitations in this study. First, the relationship between cellular immunity at the tumor site and anti-tumor effects by Tz therapy were not well evaluated although the number of infiltrated CD8-positive cells at the tumour lesion showed a significant and moderate correlation with the tumour reduction ratio. Breast carcinoma specimens with grade 3, 4, or 5 by the histopathological evaluation following NAC was more in Tz+ group than in Tr- group, respectively, however, 3 specimens with grade 5 (no tumor) in Tz+ group, which showed most evident anti-tumor effects, cannot be evaluated regarding the cellular immunity at the tumor site. Moreover, only Tz was available for HER2-positive NAC cases. The influence of breast carcinoma subtypes cannot be denied based on the significant high levels of adjacent stromal CD4-positive, CD8-positive, and Fox-P3-positive cells in the Tz+ group, although this study confirmed the cell-mediated immune activation effect in the Tz-administered group in three distinct tissue compartments. Assessment of pre-treatment biopsy specimens may more clearly reveal the effect of trastuzumab on cellular immunity. However, in this study, 5 (17.2%) biopsy specimens resected in other hospitals could not be examined, and insufficient amounts of the other 24 biopsy specimens were available to adequately evaluate several subsets of TILs by immunohistochemical analysis; thus, insufficient TIL data of pre-treatment biopsy were excluded from this study. Therefore, a prospective study is needed to obtain a sufficient volume of biopsy specimens to evaluate the various subsets of TILs before treatment.
In conclusion, Tz therapy as a NAC induced adaptive cellular immunity at the breast carcinoma lesion. This new immunological evidence in patients treated with Tz would contribute to developing new immunotherapy using Tz or other molecularly targeted antibodies.
Footnotes
Acknowledgements
We thank Miki Wakabayashi and Ikuko Arikawa for their help with immunohistochemical staining.
Conflict of interest
The authors declare no conflicts of interest.
