Abstract
OBJECTIVE:
To show that the expression of urokinase plasminogen activator (uPA) in the leucocytes of patients with benign and malignant breast lesions correlates with its expression in the lesions, and to explore the role of uPA as a tumour marker in breast cancer.
METHODS:
Using real time reverse transcriptase - polymerase chain reaction (RT-PCR), we examined the expression of uPA in leukocytes and tissues of three groups of women: a) with breast cancer (BC), b) with benign breast lesions and c) a control group of healthy women. An arbitrary value of 1 was assigned to the level of uPA expressed in the leucocytes of the healthy controls; all other measurements were expressed as a function of this value.
RESULTS:
The expression of uPA was significantly higher in BC samples compared to benign breast lesion (5.7 versus 3.8 times; p < 0.001). The leukocyte uPA of healthy volunteers was significantly lower than the leukocyte uPA from patients with breast lesions (p < 0.001). Leukocyte uPA level of patients with BC was higher compared to leukocyte uPA of women with benign lesions (p < 0.01). The expression of tissue uPA was the highest in triple-negative breast cancer. Methylation status was similar across tissue and leukocyte samples.
CONCLUSIONS:
Leukocyte uPA can be considered a surrogate of the tissue uPA expressed in BC samples. These results further support the use of leukocyte uPA as a biochemical marker of breast cancer.
Introduction
Breast cancer (BC) is the most common cancer in women and the leading cause of cancer death in females [32]. The growing knowledge of BC biology has led researchers to consider this disease as potentially systemic from its very early stage. Over past decades, major efforts have been made to deepen the understanding of BC metastatic mechanisms. The urokinase plasminogen activator (uPA) is a member of the serine protease family that converts plasminogen into plasmin, and therefore plays a key role in the proteolysis of the extracellular matrix, including the basal membrane [1,3]. The expression of uPA is controlled by epigenetic mechanisms and it is closely related to the degree of methylation of its promoter [21,22]. uPA’s function in cancer spreading has been shown in many types of malignancies, including BC [31]. 1988 Duffy et al. [12] showed that uPA blood levels in patients with early BC might be of prognostic value. Since then, many studies [2,25,27,30] have evaluated its role in tumor growth, angiogenesis and vascular remodelling in BC. One randomized control trial [28] is currently evaluating the role of tumor uPA and plasminogen activator inhibitor 1 (PAI-1) expression in the stratification of BC relapse risk. Data are scarce regarding its role as a potential tumor marker for BC, its use to differentiate benign from malignant breast lesions, and the correlation between uPA expression in breast tissues (both benign and malignant) and in peripheral leucocytes. We aim to analyze the expression of uPA as well as the methylation status of its promoter in tissue and blood samples of patients with benign and malignant breast lesions.
Methods
Subjects
After approval of the Ethics Committee of the Policlinico Umberto I, Rome, Italy, samples were collected at the breast unit of the University of Rome “La Sapienza’. From May 2012 to September 2014, 30 women, undergoing diagnostic incisional vacuum-assisted biopsies (Mammotome) or surgical excisional biopsies for suspicious lesions, were included. Moreover, peripheral venous blood was obtained from 10 additional healthy women. Written informed consent was obtained from all individuals included in the study.
Samples collection and preparation
A portion of the biopsied breast lesion and of adjacent healthy breast tissue as well a 10 mL sample of peripheral venous blood were collected from each participant. The breast tissue samples were frozen and stored at −80 °C within two hours of the extraction. Blood samples were stored in ice for a maximum of 6 hours and subsequently used for the extraction of leukocytes. Once extracted through a medium separation density gradient Lympholyte-H (Cedarlane®, Burlington, Canada), the leukocytes samples were frozen and stored at −80 °C. At the end of the enrolment, the following samples were collected: 14 breast carcinomas, 16 benign breast lesions (for all 30 patients, the healthy breast tissue adjacent to the lesion and peripheral leukocyte samples were collected) and 10 blood samples from healthy (without suspicious breast lesions) volunteers.
Clinical characteristics
Patient characteristics are summarized in Table 1 (BC patients) and Table 2 (patients with benign breast lesions). The median age of patients with a malignant lesion was 58 years (range: 32–81), 5 (36%) were premenopausal and 9 (64%) postmenopausal. All detected BC were invasive, 13 (93%) were ductal carcinomas, 10 (71%) were hormone receptor (estrogen (ER) and progesterone (PR)) positive and HER-2 negative. Twelve (85%) had a medium-low (≤20%) proliferation index (Ki-67). Woman who received systemic (chemotherapy) or local (radiation) therapy before enrolment were not included.
Characteristics of patients with BC (N = 14)
Characteristics of patients with BC (N = 14)
Abbreviations: ER: estrogen receptors; PR: progesterone receptors; HER-2: Human epidermal growh factor 2; IHC: immunohistochemistry. *Status of HER-2 was determined by Hercept (Dako). No tumour was classified as ++ and then analyzed with dual-colour fluorescent in situ hybridization (FISH).
The extraction of leukocytes from whole blood was performed with Lympholyte-H®, following the manufacturer’s instructions [36]. From each patient at least 3 mL of peripheral venous blood were collected in a tube with EDTA anticoagulant, preserved in ice, transported to the laboratory (within 6 hours of collection) and diluted with 3 mL of phosphate buffered saline (PBS), pH of 7.2. The 6 mL of so obtained diluted blood, were then deposited over 3 mL of Lympholyte-H® solution, using a 10–15 mL centrifuge tube and centrifuged for 20 minutes at 800 rpm, at room temperature. At the end of the centrifugation, lymphocytes and monocytes were transferred to a new centrifuge tube for a final centrifugation at 800 rpm for 10 minutes.
Characteristics of patients with benign lesions (N = 16)
Characteristics of patients with benign lesions (N = 16)
*Sclerosing adenosis (4), complex cysts (1), epithelial hyperplasias (1).
Total RNA extraction was performed with Allprep DNA/RNA/protein® mini-kit (QIAGEN Germantown, Maryland, USA) following the manufacturer’s instructions. The obtained total RNA was quantified by spectrophotometer and used as a template for real time-polymerase chain reaction (RT-PCR). cDNA synthesis was performed starting from 0.5 μg of total RNA using 50 pmol of oligo-d (T), in 50 mM Tris-HCl pH 8.3, 50 mM KCl, 4 mM DTT, 10 mM MgCl2, 20 UI of ribonuclease inhibitor (Stratagene), 0.3 mM each of dGTP, dCTP, dATP, dTTP in a final volume of 50 μL per sample containing 50 UI of reverse transcriptase Stratascript at 40 °C for 1 hour followed by heat inactivation at 95 °C for 5 minutes.
Quantification of the uPA mRNA was obtained with RT-PCR. In the amplification reaction, 0.01 μg of cDNA were used in a final 20 μL reaction mixture containing 6 pmoles of each primer and probe and 10 μL of FLUOCycle Mix specific for use in real time PCR with TaqMan probes (Euro Clone®). The primers, probes and the optimal amount of cDNA to be used in the reaction were previously developed by amplifying a scalar dilution control cDNA used to construct a standard curve. The efficiency of the primers was between 90 and 99%. The primer and the probes used for uPA, and the reference gene 18S are shown in Table 3. The amplification reaction was carried out with the MJ Research DNA Engine Opticon2® device (Bio-Rad Laboratories Inc, Hercules, California, USA), performing 10 minutes at 95 °C of initial denaturation, followed by 40 cycles thus constituted: 10 seconds of denaturation at 94 °C, 20 seconds of pairing at 62 °C, 30 seconds at 72 °C and fluorescence reading; the cycles were followed by 10 minutes of final extension at 72 °C. The signal relative to the expression of uPA, was normalized with respect to the one relative to the expression of RNA for the ribosomal subunit 18S (a housekeeping gene). This housekeeping gene was amplified in the same reaction tube of the gene of interest (reaction in duplex) thanks to the use of a TaqMan probe labelled with FAM fluorophore for the two genes of interest and a probe labelled with HEX fluorophore (which is excited and emits fluorescence at a different wavelength) for the 18S. Primer efficiencies were between 90 and 99%. We arbitrarily assigned to the expression of uPA in the leukocytes of the healthy women (control) a value of 1. We expressed the uPA values, measured in all other samples, as a function of the level expressed in the healthy control leukocytes.
Primers and probes used in Real-Time PCR
Primers and probes used in Real-Time PCR
The state of methylation on leukocytes was tested on 4 samples of leukocyte DNA extracted from patients with BC and from 4 healthy controls. Similarly, the methylation status of the tissues was tested using four samples of BC and 4 samples of normal breast tissue (healthy breast tissue adjacent to the benign breast lesion). DNA was extracted using the QIAGEN DNA Kit®, following the manufacturer’s instructions as described above. To evaluate the methylation status, the technique with sodium bisulfite was used using Epitect Bisulfite Kit® (QIAGEN Germantown, Maryland, USA) following the manufacturer’s instructions and purified with Epitect Qia Cube® (QIAGEN Germantown, Maryland, USA). The properties of sodium bisulfite to convert cytosine in uracil provided a very effective and accurate method for investigating methylation of this nucleotide. The modification reaction occurs through three successive steps in which the cytosine is first sulfonated, then deaminated by a hydrolytic, and finally desulfonated. Methylation in position 5 protects the cytosine residue from this modification so that methylated cytosines remain unchanged. At this point, DNA can be amplified using two pairs of oligonucleotides with filament-specific primers. The cytosines protected by methylation and therefore not modified will remain as such in the PCR product, while the cytosines modified in uracil will be replaced with a thymine residue during amplification. In this way, the cytosine/guanine doublet will be replaced by thymine/adenine doublet. The complete bisulfite technique to evaluate the state of methylation is better described elsewhere [20].
Statistical analysis
One-way ANOVA was used to examine the between-group differences. To avoid inflation of the alfa level due to multiple comparisons, a Bonferroni correction test was used. Two sided p-values < 0.05 were considered statistically significant. All statistical analyses were undertaken using SPSS v.24.0 (IBM Corp., Armonk, NY, USA). To draw the scatter plot of regression between tissue and leukocyte uPA levels in BC and non-BC samples and to calculate Pearson coefficient, we used Microsoft Excel® version 2003.
Results
Expression of urokinase in patients with BC and in women with benign breast lesions
The expression of urokinase was significantly higher in BC samples compared to benign breast lesions (5.7 times versus 3.8 times; p < 0.001). Additionally, in BC samples, a significant difference in uPA levels was noted between the lesion and the adjacent tissue (5.7 times versus 4.7 times; p < 0.01). No statistically significant difference was measured between uPA level in benign breast lesions and the adjacent tissue (Fig. 1).
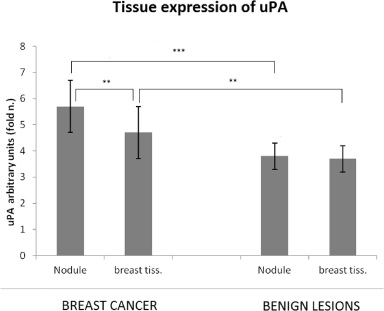
Comparison of urokinase plasminogen activator (uPA) tissue expression in BC samples, benign mammary lesions and in the tissue adjacent to both types of lesions. Histograms show the mean ± SD (standard deviation). Asterisks highlight the differences statistically significant: **p < 0.01, ***p < 0.001.
Lymphocytes from healthy volunteers showed a significantly lower level of uPA than those from women with benign and malignant breast lesions (p < 0.001). A statistically significant difference in uPA levels was also noted between lymphocytes extracted from BC patients and those belonging to women with benign breast lesions (4.32 times versus 2.98 times; p < 0.01) (Fig. 2).
uPA levels in the leukocytes and tissue samples of BC patients were similar (median ratio leukocyte/tissue of 75% ca) except in 2 patients with triple negative BC (median ratio leukocyte/tissue of 65% ca). In Fig. 3, a scatter plot represents the correlation between the expression of uPA in tissue and leukocytes both in patients with BC (section A) and in patients without BC (section B). The regression line shows an almost linear correlation between tissue and leukocyte uPA values in patients with BC (Pearson correlation coefficient 0.987). The correlation was weaker in the benign samples (Pearson correlation coefficient 0,217).
The results of the uPA gene methylation analysis showed no statistically significant differences between the groups, both in the leukocytes and in the tissue samples (Fig. 4). Of note, we found that there was a greater methylation in the BC samples compared to the benign lesions, although this difference was not statistically significant (p = 0.08).
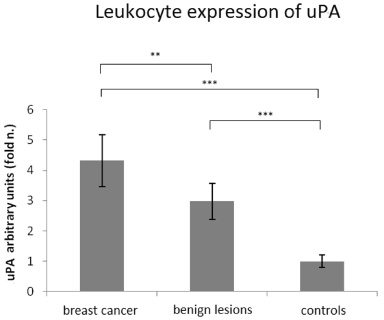
Analysis of urokinase plasminogen activator (uPA) by RT-PCR in leukocytes belonging to women with breast cancer and women with benign breast lesions; the control value is represented by uPA expressed in the leucocytes of healthy voluntary women. Asterisks highlight the differences statistically significant: **p < 0.01, ***p < 0.001.
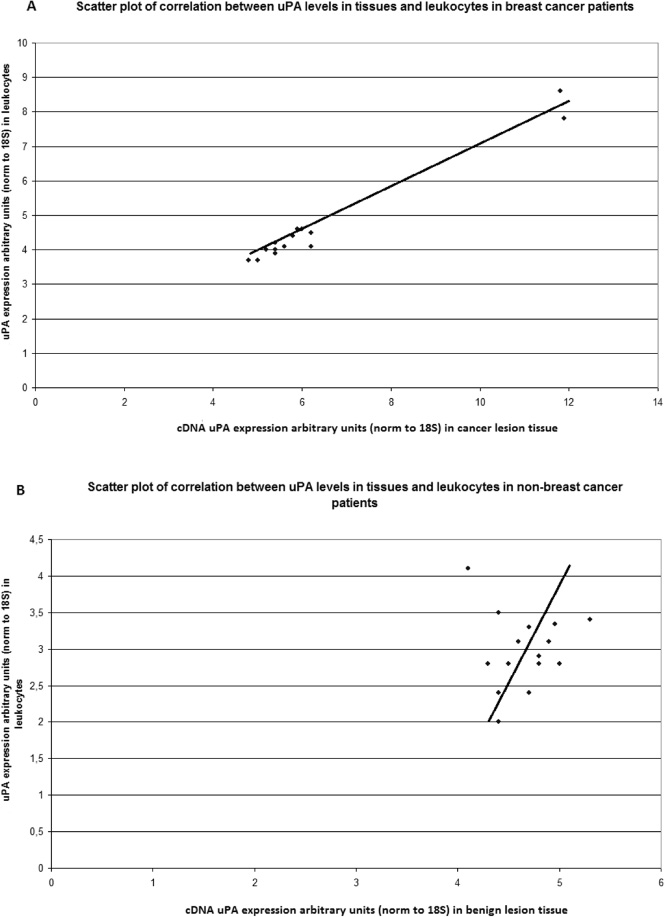
Scatter plot of the expression of tissue and leukocyte urokinase plasminogen activator (uPA) in patients with breast cancer (A) and with benign lesions (B), expressed in arbitrary units, for each sample. The bold line represents the trendline. Pearson-r = 0.987 (A). Pearson-r = 0.217 (B).
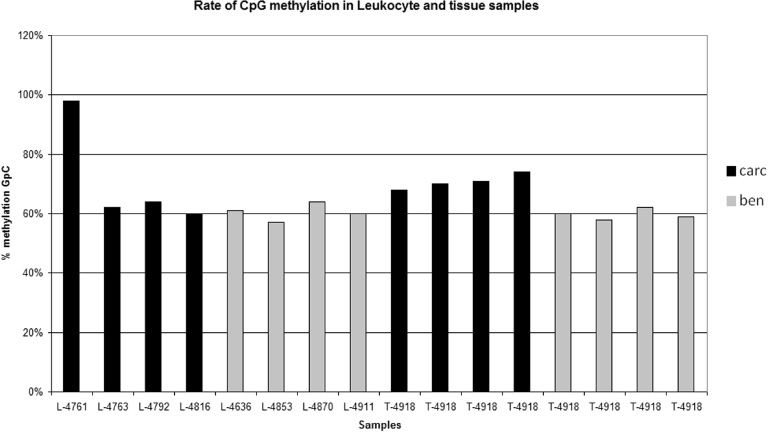
Rate of Cytosine-phosphate-Guanine (CpG) methylation of the urokinase plasminogen activator (uPA) gene in carcinomas (carc) and in benign lesions (ben) in leukocyte (L-) and tissue (T-) samples.
The expression of uPA was compared with clinical and biological parameters commonly used to define BC prognosis such as menopausal status, histological type, proliferation index (Ki-67), expression ER and PR receptors, and HER-2 over expression status. The expression levels of uPA did not significantly differ according to the aforementioned parameters, except for tumors characterized by the simultaneous negativity for ER, PR and HER2 (triple negative). In those tumors, the expression of uPA was very high (11.9 times versus 5.5 times; p < 0.001).
Discussion
The expression of uPA has been associated with the development of various types of cancer [7,14,18,35], including BC [31]. Despite its proteolytic function being strongly associated with the spreading capacity of malignant tissues, many studies have shown the presence of uPA antigens also in benign breast lesions and in healthy glandular parenchyma [9,11,13,29]. In the present study, we compared the expression of uPA in different tissue samples (BC, benign breast lesions and healthy glandular parenchyma) and leukocytes. To our knowledge, only one previous study compared tissue and leukocyte uPA levels in patients with breast lesions [9]. Compared to the study from Ceccarelli et al. [9], our results are derived from a higher number of patients, lymphocytes samples were available for each breast lesion (benign or malignant), and methylation status was studied. The leukocytes of the healthy volunteers showed a weak expression of uPA, which is consistent with the previous literature [5]. Moreover, we found that BC lesions had an increased expression of uPA. This can be explained by the role of proteases in the process of neoplastic invasion [10,16]. In order to have a better understanding of the uPA expression, we measured it in three types of tissues including breast lesions, normal parenchyma adjacent to the lesions, and in leukocytes extracted from peripheral blood. In our sample of 14 BC patients (Fig. 1), we found that uPA was over expressed also in the tissue adjacent to the neoplastic process. A possible explanation for this finding is that uPA is expressed by leukocytes and other cells (i.e. macrophages, myofibroblasts and endothelial cells), that are found on the tumour microenvironment [23,26]. Additionally, we found a good correlation between the level of uPA in BC samples and the peripheral leukocytes. This finding is potentially clinically relevant and should be further explored. If confirmed, the expression of leukocyte uPA could be used as a surrogate for the expression of uPA in malignant breast tissue. Additionally, if ongoing studies [28] confirm the prognostic role of uPA, its measurement in BC patients could become standard in clinical practice. We also found a positive correlation with the expression of uPA in benign breast lesion and the adjacent parenchyma (data not shown). This finding has not been reported previously. Further studies should investigate this correlation in deeper detail in order to allow a correct interpretation.
Leukocytes have been shown to be good markers of the methylation status of an organism. Leukocytes obtained from healthy volunteers subjected to a diet low in folate showed a reduced degree of global DNA methylation [17,24]. Although we found a significant difference in the expression of uPA according to the type of breast lesion, we did not find a significant difference in the methylation level either between different type of lesions (benign versus malignant) or between healthy breast parenchyma and breast lesions. We can therefore postulate that the over expression of uPA observed in our study was not due to over methylation.
Although uPA is considered to be an independent prognostic factor in BC, second, for power, only to the status of axillary lymph nodes [15,19,35], it is currently not used for risk stratification in clinical practice. In our study, the expression of uPA was not correlated to hormone receptor and HER-2 status, nor to the Ki-67 rate or menopausal status, with the exception of 2 patients with triple negative BC (TNBC) whose uPA mRNA levels were significantly higher than the rest of the malignant tissues analysed.
Conclusions
Our results, although derived from a small sample size, confirm that uPA is over expressed in BC tissue. We found a strong positive correlation between uPA expression in the lesions and in the peripheral leukocytes, thus indicating the possibility of using leukocyte uPA as a surrogate for tissue uPA. In our opinion, the homogeneous over expression of uPA found in all BC patient tissues (malignant breast lumps, normal parenchyma adjacent to the lesion and leukocytes) should be seen as a trait of the whole organism affected by cancer as previously suggested [9]. In addition, our results show that the expression of uPA is regulated by its promoter methylation. Finally, further studies with larger sample sizes, should be carried out to confirm our preliminary results. Efforts should be made to characterise the mechanisms involved in the over expression of uPA in BC patients.
