Abstract
Background
Long-term prescription patterns of polypharmacy and potentially inappropriate medication (PIMs) in dementia patients were limited in the Chinese population.
Objective
Examine the prevalence trends of polypharmacy and PIMs in people living with dementia.
Methods
Using electronic healthcare data from Hong Kong(2006–2021), we evaluated trends of polypharmacy (≥5 medications) and hyperpolypharmacy (≥10 medications) prescribed within 60 days per year in dementia patients aged ≥65 years old. PIMs were identified using the 2019 Beers criteria and the Screening Tool of Older Person's Prescription version 2, with at least one prescription per year. The COVID-19 pandemic period was defined as 2020–2021. The prevalence of polypharmacy, hyperpolypharmacy, and PIMs was calculated as the affected people divided by the total number of dementia patients alive each year. Joinpoint regression models was used to analyze trends and estimate average annual percent change (AAPC).
Results
The prevalence of polypharmacy increased from 2006 (36.4%) to 2021 (66.4%) (AAPC: 4.0). Hyperpolypharmacy also increased from 3.3% to 21.0% (AAPC:12.7). During pandemic, stable trends were observed in polypharmacy (p = 0.08) and hyperpolypharmacy (p = 0.20). The prevalence trend of PIMs from 2006 to 2021 was stable (35.8%–35.2%, p = 0.17), with a slight increase observed after 2015 (33.5%–35.2%, p < 0.0001). Between 2006 and 2013, and after the release of the new guideline (2019–2021), the prescription rates of antipsychotics and anticholinergics decreased (both p < 0.001), while prescriptions for non-benzodiazepine (AAPC: 4.4) and benzodiazepine (AAPC: 3.6) increased.
Conclusions
Reviewing polypharmacy and hyperpolypharmacy among people living with dementia is needed to ensure the appropriate use of medications.
Keywords
Introduction
Dementia represents a significant global public health concern, requiring effective management strategies. 1 While there are currently no cures for dementia, pharmacological interventions are crucial in managing disease progression and alleviating symptoms.1,2 Recently, a novel medicine called Lecanemab has shown a moderate attenuation of cognitive and functional decline compared to a placebo over 18 months. However, it is worth noting that the use of this medicine has been associated with adverse events, including infusion-related reactions. 3
People with dementia often have multiple comorbidities and related complications that require multiple medication management. 4 This may result in polypharmacy or hyperpolypharamcy. 5 Generally, polypharmacy is defined as using ≥ 5 medications in a period, and hyperpolypharmacy is defined as using ≥ 10 medications in a period. 6 On average, people with dementia concurrently take 5–10 medications, of which 1–2 are prescribed for dementia and the remainder for other conditions. 7 Polypharmacy and hyperpolypharmacy are independent risk factors for adverse drug events and hospitalization in older people, possibly due to drug-drug interaction and drug-disease contraindication. 8 However, polypharmacy can be appropriate if the complete medication list is reviewed and the patient's risk/benefit ratio is assessed. 9
Polypharmacy may contribute to potentially inappropriate medication (PIMs), where the risks of adverse drug reactions outweigh the benefits in older people.9,10 Beers and Screening Tool of Older Person's Prescription (STOPP) criteria are guidelines that help identify PIMs in older people and recommend them for people with chronic conditions, such as dementia.11,12 These guidelines are updated regularly according to panel composition and literature review. Despite these recommendations, which advise against the use of certain medications (e.g., anticholinergics, antipsychotics, antidepressants, benzodiazepines, and nonbenzodiazepines) in people with dementia, a significant portion of this population is still prescribed these medications to manage their symptoms. People with dementia are especially at high risk for adverse drug reactions due to the decline of cognitive function, highlighting the burden associated with the inappropriate use of medications in this population.
The optimized management of polypharmacy, hyperpolypharmacy, and PIMs in dementia remains unclear regarding which medications and combinations are used and how these patterns change. By examining the temporal changes in polypharmacy, hyperpolypharmacy, and PIMs, researchers and healthcare providers may identify the drawbacks in current prescribing practices and provide a timely improvement. Previous studies have investigated the changes in polypharmacy and PIMs in people with dementia.13–17 However, it is worth noting that the population and the patterns are from the Western population, and the change in prescriptions has not been fully understood in the Chinese population. While one study is working on the patterns of PIMs in Eastern China, they only focused on a limited study period from January to December 2020. 18 The long-term trends in PIMs prescriptions for this population have yet to be thoroughly explored. Understanding the patterns in the Chinese population will provide insight into evaluating the issue of polypharmacy and PIMs from a global perspective. Furthermore, these studies have not included the periods of the updated Beers criteria (2019), which included additional medications for the anticholinergics group (diphenhydramine, oxybutynin, and amitriptyline) in people with dementia, compared to the 2015 version. The COVID-19 pandemic began in late 2019 and has significantly changed healthcare service delivery, including medication prescribing. There is also a concern that people with dementia may be affected by social distancing, leading to an increase in psychiatric symptoms. However, very limited studies examined the trends of polypharmacy, hyperpolypharmacy, and PIMs in this population during the pandemic.
Therefore, our study aims to examine the temporal trends of polypharmacy and PIMs in people with dementia in Hong Kong (HK) using a population-based cohort from 2006 to 2021, covering the updated guideline (2019 for Beers criteria) and pandemic period. The objective is to gather evidence to determine the trends of polypharmacy, hyperpolypharmacy, and PIMs in people living with dementia, covering the guidelines changes and the COVID pandemic. Our results are crucial for the optimal allocation of healthcare resources in managing individuals with dementia in the future.
Materials and methods
Data source
We retrieved data from the Clinical Data Analysis and Reporting System (CDARS), a computerized clinical management system developed by the HK Hospital Authority that contains electronic patient records. The system includes demographics, diagnoses, procedures, prescription records, laboratory tests, and hospitalization details from primary, secondary, and tertiary sectors. CDARS captures more than 70% of hospitalizations in HK, and patients’ records are deidentified to ensure confidentiality. This study did not involve any human subjects or animals directly. A unique reference key was generated and assigned to each patient to facilitate data retrieval and further analysis. CDARS has been used in previous high-quality epidemiological studies.19–24
Study population
The study population included those (1) aged ≥ 65 or older at the first diagnosis of dementia and (2) diagnosed with dementia from 1 January 2006 to 31 December 2021. As polypharmacy was examined in each calendar year, the study population should follow the inclusive criteria before the start of the calendar year. The International Classification of Disease-9 version (ICD-9) identifying dementia is listed in Supplementary Table 1.
Definition of polypharmacy and PIMs
We used the simultaneous polypharmacy method for this study, 6 which is defined by the number of concurrent medications used during a period to ensure we consider medications prescribed at different times. In this study, polypharmacy is the use of ≥ 5 different medication types concomitantly for ≥ 60 days per calendar year from 2006 to 2021 after the first diagnosis of dementia. Hyperpolypharmacy was defined as using ≥10 different medication types concomitantly for ≥ 60 days per calendar year after the diagnosis of dementia. The definition was selected after reviewing previous literature on polypharmacy definitions applied in research and practice. 25 We considered the medications to be continuous if the duration between consecutive prescriptions of the same drug was within 5 times the half-life of the corresponding drugs. One calendar day was added if the duration of 5 half-lives was less than one day (or 24 h). The concomitant period was defined as the days that covered different medication types for more than 60 days within a year (Supplementary Figure 1). We excluded the medications of topical, intra-articular, OTC, and ophthalmic routes because these medications have limited systemic synergetic effects. We also excluded the prescriptions not intended for regular use, including those specified to be used, when necessary, prescriptions for vaccines, wound cleaning, surgical or other procedures.
PIMs were defined as using at least one script of locally prescribed medications identified from Beers criteria (2019) and STOPP v2 for older people with dementia.11,12 PIMs included in this study were the classes of anticholinergics, tricyclic antidepressants (TCA), antipsychotics, antiparkinsonian antimuscarinics, benzodiazepines (BZD), nonbenzodiazepine (non-BZD) hypnotics, and urinary antimuscarinics. Detailed medications for these medication classes are listed in Supplementary Table 2.
In HK, the first case of COVID-19 was detected in January 2020, and the region experienced four waves of the epidemic up to 2021. The localized nature of the epidemic resulted in restricted access to the healthcare system and the implementation of social distancing measures. These factors may have influenced the prescription practices for people with chronic diseases, including dementia. Therefore, our study defined the pandemic period from 2020 to 2021 and examined the patterns of polypharmacy, hyperpolypharmacy, and PIMs during this specific timeframe.
Statistical analysis
The prevalence of polypharmacy, hyperpolypharmacy, and PIMs was calculated as the number of people experiencing each condition divided by the total number of people alive with dementia each year. People who died halfway through the corresponding year were excluded. All annual prevalence was expressed as a percentage of patients, separated by sex. The age-standardized rate was calculated using the total population of 2006 from HK as the reference population.
We used the joinpoint regression analysis to test the polypharmacy, hyperpolypharmacy, and PIMs trends from 2006 to 2021. The joinpoint models provide a flexible and powerful tool for analyzing trends and identifying significant changes in data over time, allowing for the detection of joinpoints where the direction or magnitude of a trend shifts, enabling researchers to understand the prescription patterns better and make informed decisions. We fitted a series of connected lines to the prevalence of polypharmacy, hyperpolypharmacy, and PIMs, respectively. We joined straight-line segments at joinpoints, where the change in trend was statistically significant. 26 We presented the joinpoint as missing if the trends were a continuing straight-line without joinpoint. We calculated the annual percent change (APC) and average annual percent change (AAPC) and their 95% confidence interval (CI) using the prevalence of polypharmacy, hyperpolypharmacy, and PIMs to assess the temporal trends. The APC characterized trends in prevalence over time and was obtained by a regression in a logarithmic scale model. The joinpoint model used statistical criteria to determine when and how often the APC changes. 26 The best-fitted joinpoint model allows us to determine how long the APC remained constant and when it changed. The AAPC summary measures over a fixed pre-specified interval, making it possible to use a single number to describe the average APCs over the study period. 27 All the statistical tests were two-sided, and p-values < 0.05 were considered significant. All the statistical analyses were conducted using SAS (version 9.4) and R 4.1.2 (R Development Core Team, 2008). Joinpoint regression analysis, APC, and AAPC used Joinpoint Statistical Software (version 4.7.0.0) provided by the National Cancer Institute, US.
Results
We observed 168,446 patients with dementia from 2006 to 2021, with 21,641,060 prescriptions for regular systemic use. 60% of people were female, with a mean age of 82.4 (Standard deviation: 9.0) at the first dementia diagnosis (Table 1). Characteristics of patients for every 5 years during the follow-up was shown in Supplementary Table 4.
Baseline characteristics of study population.

As shown in Table 2, 36.4% (95% CI: 35.8%, 37.0%), 3.3% (95% CI: 3.1%, 3.5%), and 35.8% (95% CI: 35.2%, 36.4%) of people with dementia had polypharmacy, hyperpolypharmacy, and PIMs in 2006, respectively. These are compared to the prevalence of polypharmacy, hyperpolypharmacy, and PIMs of 66.4% (95% CI: 66.0%, 66.8%), 21.0% (95% CI: 20.7%, 21.3%), and 35.2% (95% CI: 34.8%, 35.6%) of people in 2021. Figure 1A shows the trends of age-standardized polypharmacy from 2006 to 2021. There was a dramatic increase from 2006 to 2011, with an APC of 6.9 (95% CI:6.4 −7.5, Supplementary Table 3). After 2011, the trend of polypharmacy steadily increased, with a prevalence of 65.5% (95% CI: 65.1%, 65.9%) in 2019, when the updated guideline was released, and stabilizing at 66.4% (95% CI: 66.0%, 66.8%) during the pandemic. The AAPC for overall polypharmacy was 4.0 (95% CI: 3.7, 4.3). Similar patterns were found in the trends of hyperpolypharmacy (Figure 1B), with an AAPC of 12.7 (95% CI: 11.6, 13.8). Subgroup analyses were similar to the overall patterns of polypharmacy and hyperpolypharmacy (Supplementary Figure 2).
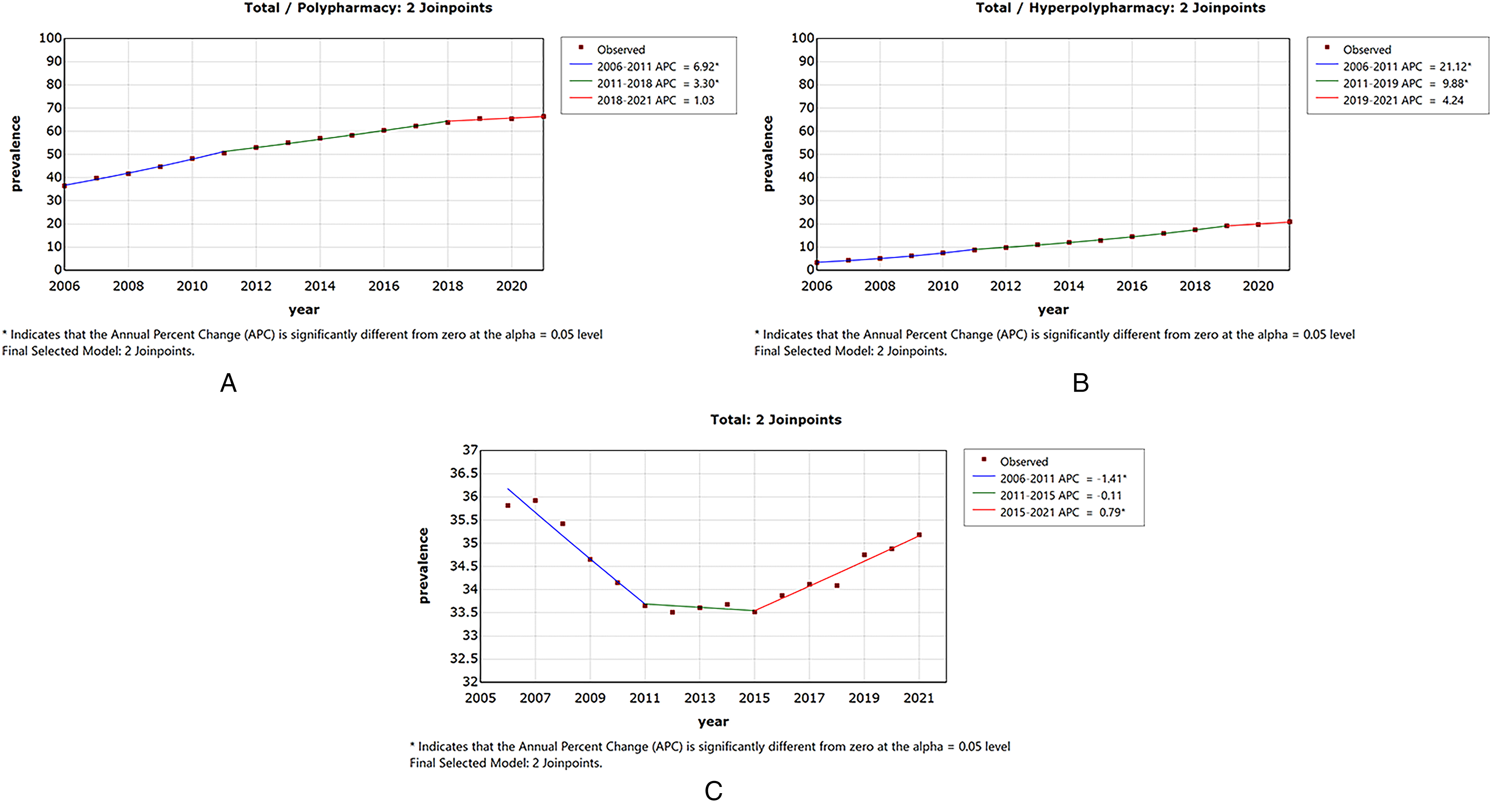
Trends of the age-standardised prevalence of (A) polypharmacy (B) hyperpolypharmacy, and (C) PIMs.
Prevalence of polypharmacy, hyperpolypharmacy, and any PIMs across the years and AAPC (95% CI).
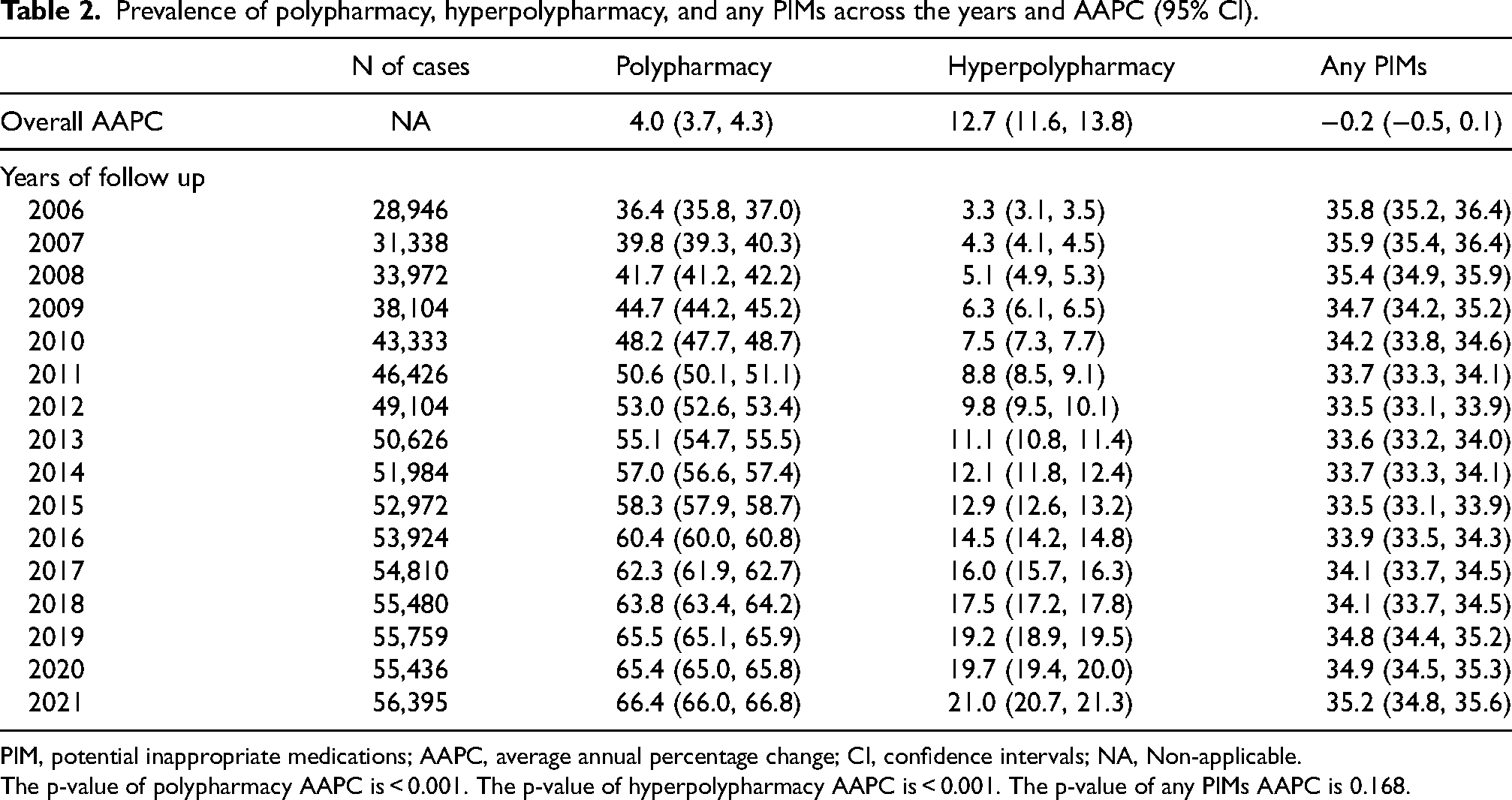
PIM, potential inappropriate medications; AAPC, average annual percentage change; CI, confidence intervals; NA, Non-applicable.
The p-value of polypharmacy AAPC is < 0.001. The p-value of hyperpolypharmacy AAPC is < 0.001. The p-value of any PIMs AAPC is 0.168.
Figure 1C shows a stable age-standardized trend for any PIMs from 2006 to 2021, with an AAPC of −0.2 (95% CI: −0.5, 1.0). In detail, the prevalence of people using any PIMs decreased from 2006 to 2011 (APC: −1.4, 95% CI: −1.8, −1.0 Supplementary Table 3), then experienced a plateau till 2015 before increasing significantly up to the pandemic (APC: 0.8, 95% CI: 0.5, 1.1, Supplementary Table 3) (Table 2). The prescription of PIMs increased after the release of the updated Beers guideline in 2019 and during the pandemic. Although the most prescribed PIMs were anticholinergic and antipsychotics, the overall trends were decreasing for these two medications during the study period, with the AAPC of −0.9 (95% CI: −1.5, −0.3) and −0.6 (95% CI: −0.8, −0.5), respectively (Table 3 and Figure 2). Anticholinergic prescriptions experienced a significant decrease after 2018 and continued after the release of the updated guideline until 2021. In contrast, antipsychotic prescriptions were stable after the release of the updated guideline. During the study period, the number of people prescribed TCA and antiparkinsonian was low, and the trends were declining (Table 3 and Figure 2). Although the number of people being prescribed BZD, non-BZD hypnotics, and urinary antimuscarinics was low, the trends were increasing during the study period, with the AAPC of 3.6 (95% CI: 2.9, 4.3), 4.4 (95% CI: 4.0, 4.9), and 5.3 (95% CI: 4.6, 6.0), respectively (Table 3 and Figure 2). For the prescriptions of BZD and non-BZD, the prescription continued to increase after the updated guideline was released and during the pandemic. The subgroup analysis for overall PIMs (Supplementary Figure 3) and varied medication classes (Supplementary Figure 4A-G) was consistent with the main analysis. Supplementary Table 5 presents the APC before and during the COVID-19 pandemic. Except for an increase in the prescription of PIMs during the pandemic (0.8, 0.5–1.1), the prescription rates of polypharmacy and PIMs remained stable, with APC values of 1.0 (−0.2, 2.2) and 4.2 (−2.6, 11.6), respectively.
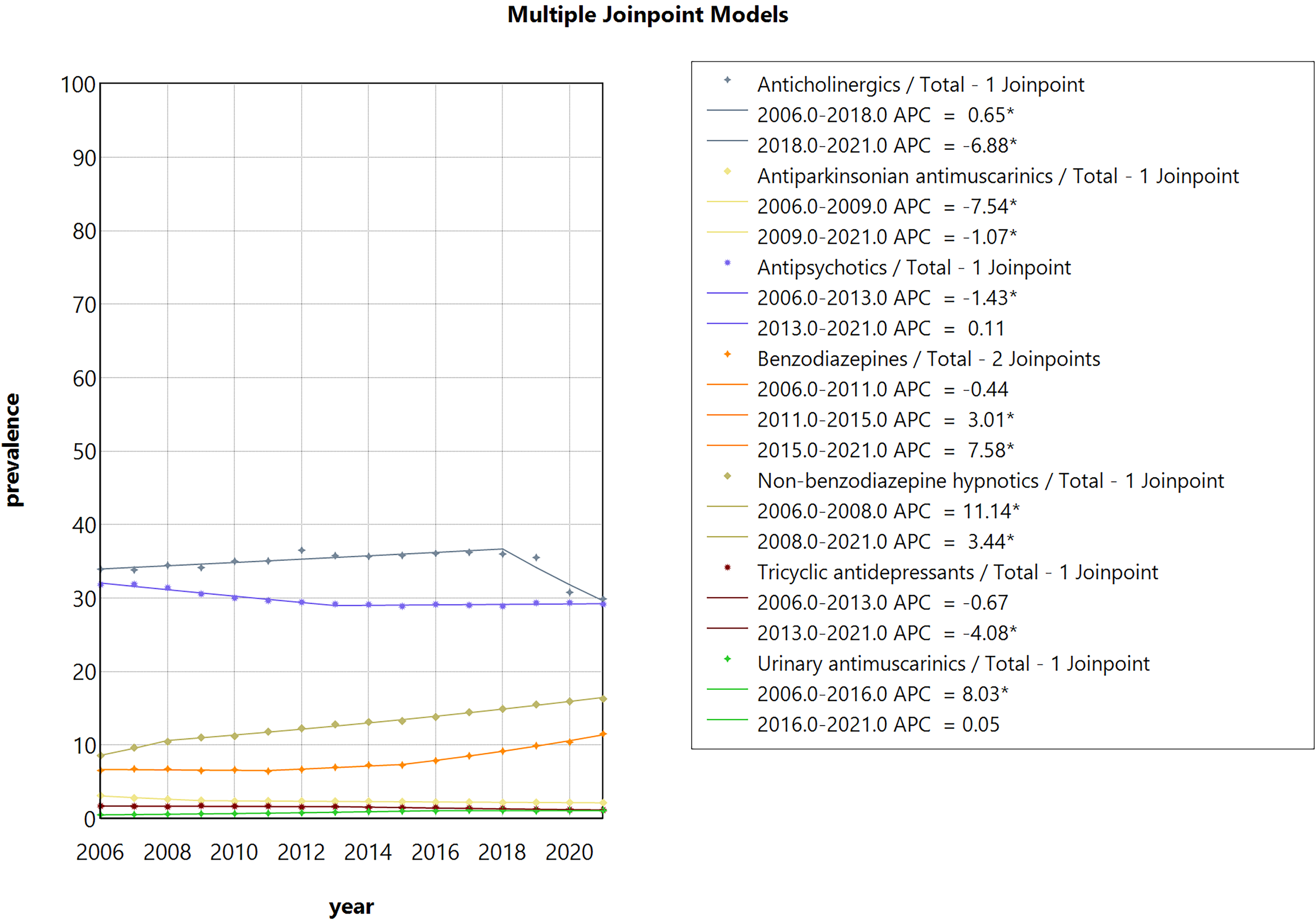
Trends of age-standardized prevalence of different PIMs.
Prevalence of each class of PIMs and AAPC (95% CI).
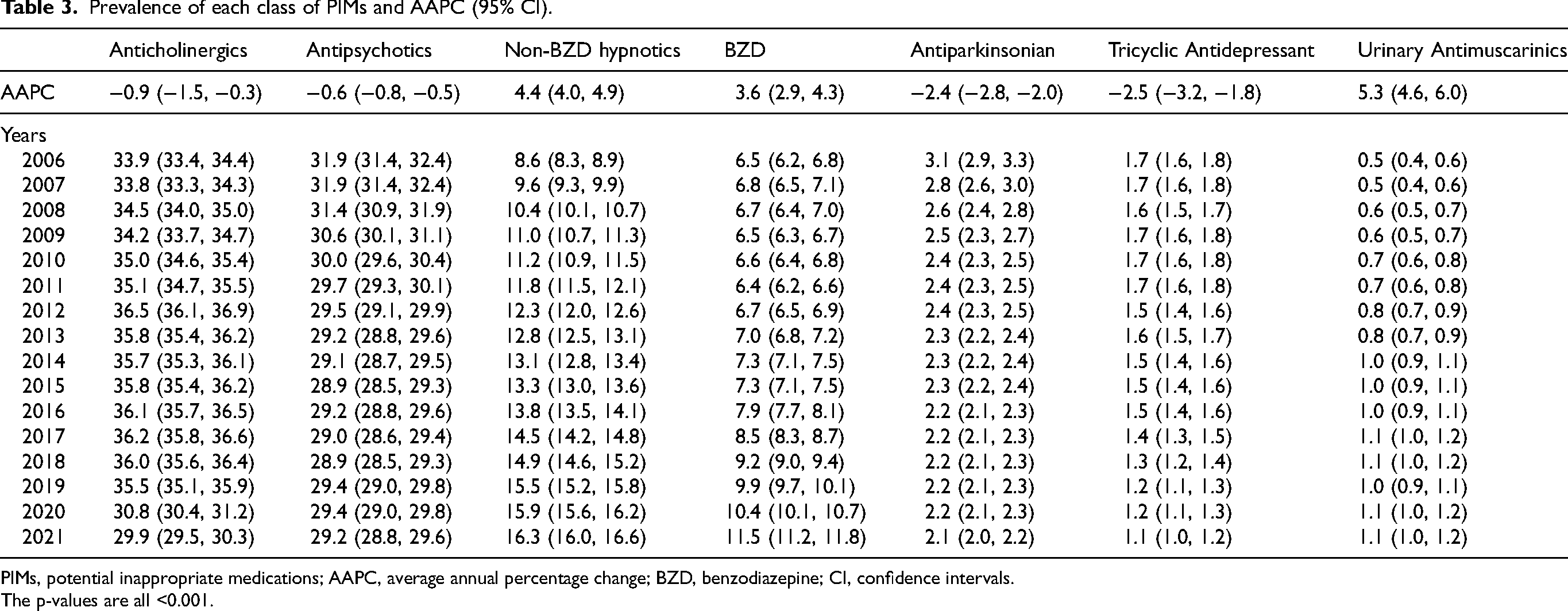
PIMs, potential inappropriate medications; AAPC, average annual percentage change; BZD, benzodiazepine; CI, confidence intervals.
The p-values are all <0.001.
Discussion
Our study showed notable trends in polypharmacy, hyperpolypharmacy, and PIMs over 16 years, covering the updated guidelines and the pandemic period. Both polypharmacy and hyperpolypharmacy showed an increasing trend from 2006 to 2021. The change in prescription guidelines nor the pandemic did not change the prescription patterns of polypharmacy and hyperpolypharmacy among people with dementia. In contrast, using PIMs was stable from 2006 to 2021, albeit slightly increased from 2005 to 2021, covering the period the updated guideline was released and the pandemic. Over the study period, most people were prescribed anticholinergic medications and antipsychotics, despite the overall trends for these medications declining. The use of non-BZD and BZD increased over the study period. There was no increased prescription of anticholinergics and antipsychotics after the updated guideline was released and during the pandemic, while the use of Non-BZD and BZD was on the rise.
The prevalence of polypharmacy and hyperpolypharmacy is lower than in previous studies in people with dementia, where the prevalence ranged from 43% to 82%. 28 However, our findings may be influenced by the varied definitions of polypharmacy used in different studies. Unlike previous studies that focused solely on the number of medications used, 28 we also considered the continuing duration of multiple medications used to highlight the medication burden. Therefore, our results should be interpreted in the context of chronic polypharmacy. 25
While numerous studies have examined the prevalence of polypharmacy in elderly populations, 28 only one study conducted in Denmark has investigated the trends of polypharmacy and hyperpolypharmacy in people with dementia. 15 This study found that the prevalence of polypharmacy increased from 47.3% to 69.4% between 2000 and 2011, and hyperpolypharmacy increased from 7.4% to 20.9%. Our study confirms these increasing trends in polypharmacy and hyperpolypharmacy during similar periods. The increased prevalence of polypharmacy and hyperpolypharmacy in people with dementia may reflect the increasing prevalence of multimorbidity which often requires multiple medications to control symptoms. 28
We observed over half of the people with dementia remained polypharmacy after 2011. These findings are consistent with a recently published multinational study testing the trends from 2013 to 2016, 25 which reported a 45% prevalence of polypharmacy in Hong Kong's elderly population aged ≥ 65 years, higher than that of Taiwan (38.8%), South Korea (32.0%), the United Kingdom (23.5%), and Australia (20.1%). This high prevalence of polypharmacy may be partially attributed to the lack of integrated care for those with multimorbidity. The fragmentation of care is characterized by patients with multimorbidity having follow-up visits in multiple specialized clinics in HK.29,30 Therefore, integrated care models centered on polypharmacy are needed to improve the optimal prescribing in people with dementia. 31
Our findings indicated the prevalence of polypharmacy and hyperpolypharmacy remained stable during the pandemic. This finding contrasts a study conducted in the US, 32 which reported an increased use of polypharmacy in older people during the pandemic. Our results suggest that continuity of care may have played a significant role in this stability. Healthcare professionals familiar with the medication regimens of individuals with dementia may have been cautious about making unnecessary changes during the pandemic. Additionally, limited access to healthcare facilities, reduced face-to-face consultations, and postponed routine care have hindered conducting comprehensive medication reviews or adjusting medication.
Our study demonstrated a stable prescription of any PIMs in people with dementia from 2006 to 2021, in contrast to current studies from Denmark, 14 where the prevalence decreased from 55% to 44% between 2000 and 2015. In addition, in Australia, 13 the prevalence of prescribing psychotropic medications, which largely contribute to PIMs, decreased from 21.7% to 18.1% in people with dementia between 2011 and 2020, showing a different prescription pattern to this study. The stable trends of PIMs could be attributed to the rising use of non-BZD hypnotics and BZD prescriptions, despite their low prescription rates, during the study period. Additionally, the stable use of antipsychotics from 2006 to 2013, before increasing until the pandemic, may also contribute to the stable trends of PIMs.
The increased use of non-BZD hypnotics during the study period is consistent with the recently published global trends in Z-drug consumption. 33 This increase in non-BZD hypnotics may be attributed to the rising prevalence of insomnia, which tends to become more common and severe as dementia progresses and can last for long periods in people with dementia. 34 We also found the use of non-BZD rose during the pandemic; this may result from the heightened stress, anxiety, and disruption of daily routines caused by the pandemic, which likely contributed to increased sleep difficulties and anxiety-related symptoms. Additionally, there were slightly increased trends in the use of BZD and urinary antimuscarinics, which are of particular concern as they are positively associated with self-harm, suicide, cognitive dysfunction, Parkinson's disease, cancer, cardiovascular diseases, and diabetes.33,35–37 Given the increased use of these medications, further exploration is needed to focus on their safety profile and management of exposure in people with dementia.
The updated Beers and STOPP criteria advised against using anticholinergics and avoided using antipsychotics as the initial treatment option for managing behavioral and psychological symptoms.11,12 in individuals with dementia. Our study demonstrates a decreasing trend in the overall prescribing of antipsychotics and anticholinergics from 2006 to 2021. Notably, we observed a significant decline in the use of anticholinergics starting in 2018, which persisted after the release of the 2019 Beers guideline and during the pandemic. These changes in prescribing are probably influenced by the findings of previous large population-based studies that identified the adverse events associated with anticholinergic use in people with dementia.38,39 The sustained decrease following the updated guideline release is encouraged because the HK prescribers followed the guidelines recommendations to reduce the use of anticholinergics. However, we found a stable prescribing pattern of antipsychotics after 2019, which contrasts with the UK and Canada,38,39 showing a rise in the prescription rates of antipsychotics and cognitive medications after 2019 (during the pandemic). This is probably an effect of the HK Hospital Authority providing comprehensive services for patients in residential care homes for the elderly, including regular outreach services and telehealth consultations with doctors and receiving medicines from dispensaries during the pandemic. 40
Our study found that about 30% of people with dementia used antipsychotics in 2021. This finding is consistent with the prevalence of prescribing antipsychotics during the pandemic in Canada. 41 Although no studies report the prevalence of prescribing anticholinergics using the most recent data, the prevalence of anticholinergics (35.0%) in our study is similar to the review, 42 including the studies before 2019, with one-third of people prescribing anticholinergics. This high utilization of these medications is concerning. Strategies such as deprescribing should be incorporated into routine care, involving clinicians, pharmacists, and other healthcare professionals in people with dementia.
Although we observed an overall decline in using PIMs from 2006 to 2021, our included PIMs are those specific for people with dementia listed in Beers and STOPP criteria. Those guidelines have not included medications including anticholinergic properties, such as anticonvulsants, cardiovascular medications (metoprolol and propranolol), and opioids. However, using anticholinergic medications is common and associated with adverse events in people with dementia. With the high prevalence of polypharmacy in HK, the risk of prescribing multiple anticholinergic medications, the anticholinergic burden, is high. Thus, we cannot rule out the possibility of adverse drug events induced by the anticholinergic burden.
Strengths and limitations
A major strength of our study is the availability of long-term prescription data, which allowed us to investigate trends in polypharmacy and PIMs in people with dementia, covering the guidelines change and the COVID-19 pandemic. This study helps track the impact of guideline changes on prescribing practices over time and identifies areas for further improvements. Furthermore, understanding the prescription patterns during the pandemic helps to identify shifts in prescribing practices, such as the use of certain medications or changes in medication regimens. As people with dementia may be more likely to experience psychiatric symptoms during the pandemic due to social isolation, our study helps inform strategies to manage the prescription of PIMs in the future. However, there are some limitations to our study. First, although the study was based on valid information on all filled prescriptions, we cannot know whether the medication was consumed. Furthermore, we have no information on the use of over-the-counter medications. Our study defined polypharmacy as concurrently using at least five different medications over 60 days. This has largely included the chronic medications used and excluded the as-needed and non-regular used medications. Thus, the underestimation of the trends may be marginal. Second, our database does not have the disease severity. The hyperpolypharmacy may be a proxy for severe dementia patients since they may take more medications than those with mild symptoms. Third, since our study concluded at the end of 2021, it does not encompass the fifth wave of the pandemic that began in early 2022, during which the city experienced its highest COVID-related mortality rate. Consequently, the current estimates may not fully capture prescription patterns during the pandemic. Nevertheless, our findings still offer insights into the prescription patterns at the onset of the pandemic. They can inform the development of interventions to enhance medication management during future pandemics or other healthcare crises.
Conclusion
Our study reveals significant upward trends in polypharmacy and hyperpolypharmacy from 2006 until before the pandemic, which subsequently stabilized during the pandemic. Additionally, we observed a steady prescription trend of PIMs from 2006 to 2021. However, there was a notable increase in the prevalence of BZDs and non-BZDs. Notably, anticholinergics and antipsychotics emerged as the most frequently prescribed PIMs in individuals with dementia, although their prescription trends decreased from 2006 to 2021. Reviewing polypharmacy and hyperpolypharmacy among people living with dementia is needed to ensure the appropriate use of medications. 43
Supplemental Material
sj-docx-1-alr-10.3233_ADR-240036 - Supplemental material for Trends of the prevalence of polypharmacy and potentially inappropriate medications in people with dementia from 2006 to 2021 in Hong Kong
Supplemental material, sj-docx-1-alr-10.3233_ADR-240036 for Trends of the prevalence of polypharmacy and potentially inappropriate medications in people with dementia from 2006 to 2021 in Hong Kong by Xiwen Simon Qin, Yang Wang, Miriam Tim Yin Leung, TianTian Ma, Kailin Chen, Hao Luo, Jacqueline Kwan Yuk Yuen, Kui Kai Lau, Doris Sau Fung Yu and Celine Sze Ling Chui in Journal of Alzheimer's Disease Reports
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
Author contributions
Simon Qin (Conceptualization; Data curation; Formal analysis; Methodology; Software; Writing – original draft; Writing – review & editing); Yang Wang (Formal analysis; Methodology; Writing – review & editing); Miriam Tim Yiin Leung (Formal analysis; Writing – review & editing); TianTian Ma (Writing – review & editing); Kailin Chen (Writing – review & editing); Hao Luo (Writing – review & editing); Jacqueline Kwan Yuk Yuen (Writing – review & editing); Kui Kai Lau (Writing – review & editing); Doris Sau Fung Yu (Writing – review & editing); Celine Sze Ling Chui (Conceptualization; Methodology; Writing – review & editing).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
