| 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) |
Drug |
Complex I |
Inhibition of ETC, increased Ca2+ levels and reduced AKT phosphorylation, Mitochondrial complex I inhibitor |
Mouse brain concentrations of MPP+ (approximately 12–47μM) triggered approximately a 50% inhibition of Complex I. Even small (20μM) amount of MPP+ exhibited pronounced deleterious effect on both the Ca2+-induced changes in the membrane potential and the Ca2+-accumulating capacity of mitochondria [12]. |
Event: 177 Mitochondrial dysfunction, Event: 887 NADH-ubiquinone oxidoreductase (complex I), Inhibition |
AOP3: Mitochondrial dysfunction and Neurotoxicity |
Mitochondrial dysfunction |
| Acetaminophen |
Drug |
CYP46A1 |
Increased 24S-OHC levels driving increased cholesterol turn-over and hypocholesterolemia |
A CYP46A1 activator activated the enzyme in mouse brains at low concentrations (up to 30μM) but inhibited the P450 at higher concentrations. It increased by > 30% the formation of cholesterol 24-hydroxylation in isolated mouse brain microsomes [13]. |
|
|
Cholesterol dysmetabolism |
| Alcohol |
Solvent |
NMDAR, NR2B expression, NR1 expression, PSD-95 |
Blocking NMDAR, NMDAR modulation resulting in excitotoxicity and neuronal damage reflect reduction in synaptic activity |
1. After a 7-day ethanol (50 mM) exposure of human embryonic stem cell (hESC)-derived cortical neurons followed by a 24-hour ethanol withdrawal treatment, four NMDA receptor subunit genes, including GRIN1, GRIN2A, GRIN2B, GRIN2D were highly upregulated [14]. |
MIE: Event: 201 NMDA receptors, Binding of antagonist |
AOP13: Binding of antagonist to NMDARs impairs cognition, AOP12: Binding of antagonist to NMDARs can lead to neuroinflammation and neurodegeneration |
Synaptic dysfunction, Neuroinflammation, Neurodegeneration, Impaired cognition |
|
|
|
|
2. Inhibition of NMDA receptor function by ethanol (and interactions between ethanol and the noncompetitive NMDA receptor antagonist ifenprodil) in neocortical neurons from rat and human embryonic kidney (HEK) 293 cells expressing recombinant NMDA receptors [15]. |
|
|
|
|
3. A significant association between high life time drinking and high daily alcohol intake with lower DNA methylation of NR2B in alcohol-dependent patients undergoing alcohol withdrawal [16]. |
|
|
|
|
4. NR1 isoforms co-expressed in various combinations with one of the four NR2 subtypes in human embryonic kidney 293 cells, the sensitivity depended on the combination of NR1-3b/NR2C, NR1-3b/NR2D, and NR1-4b/NR2C pairs, and the NR1-2b/NR2C pair were inhibited by ethanol [17]. |
|
|
|
|
5. In rat primary hippocampal cultures, chronic exposure to ethanol induced increase in PSD-95 expression and dendritic spine size [18]. |
|
|
BDNF |
Presynaptic effects mediated by disruption of NMDAR-activity dependent BDNF signaling by inhibiting BDNF activity |
An association between BDNF serum levels and the history of alcohol consumption in humans[19]. |
| Atrazine (ATZ) |
Herbicides |
Complex I and III |
Inhibition of ETC, increased Ca2+ levels and reduced AKT phosphorylation |
Sprague-Dawley rats (n = 48) were treated for 5 months with low concentrations (30 or 300μg/ kg /day) of ATZ provided in drinking water. ATZ blocked the activities of oxidative phosphorylation complexes I and III, resulting in decreased oxygen consumption. It also suppressed the insulin-mediated phosphorylation of Akt [20]. |
|
|
Glucose dysmetabolism via mitochondrial dysfunction |
| Benzalkonium chlorides |
Antiseptics |
DHCR7 |
Impairment of cholesterol biosynthesis |
Exposure to benzalkonium chlorides exhibited high potency in inhibiting DHCR7 when tested in mouse and human neuroblastoma cells by analyzing cholesterol and its precursor using gas chromatography-mass spectrometry [21]. |
|
|
Cholesterol dysmetabolism |
| Benzo[a]pyrene (B[a]P) |
Polycyclic aromatic compounds (PACs) |
CYPA1 |
Oxidation of macromolecules before conjugation with glutathione, decreased cellular ATP production, MMP, oxidative phosphorylation and mitochondrial protein complexes I, II, and IV activity, and eventually apoptosis |
1. Intraperitoneal injection of B[a]P from embryonic day 7 at a dose of 250 mg kg-1 induced NTDs (13.3% frequency) in mice. BaP exposure significantly increased expression of genes associated with oxidative stress, Cyp1a1 [22]. |
|
|
Mitochondrial dysfunction, Oxidative stress |
|
|
|
|
2. B[a]P-induced neurotoxicity occurred through mitochondria-mediated apoptosis (rat cerebral neurons). B[a]P-induced apoptosis was accompanied by loss of mitochondrial membrane potential, release of cytochrome c from mitochondria to the cytosol [23]. |
|
|
|
|
3. Exposure of human neuroblastoma SK-N-SH cells to B[a]P at 0.5–40μM for 24 h, increased in levels of ROS, significantly decreased mitochondrial membrane potential (MMP), up-regulated pro-apoptotic genes and down-regulated antioxidative genes expression, elevated cytochrome c protein levels and lipid peroxidation (LPO) in a concentration-related manner [24]. |
| Carbofuran |
Pesticides |
PP2A, GSK3β |
Disturbed mitochondrial ETC, low ATP levels, increased oxidative stress, intracellular Ca2+ levels and tau hyperphosphorylation. Reduced dephosphorylation supporting tau hyperphosphorylation |
Carbofuran exposure (rats) led to tau hyperphosphorylation at multiple AD-related phosphorylation sites with activation of GSK-3β and inhibition of PP2A [25]. |
|
|
Mitochondrial dysfunction, Oxidative stress, tau hyperphos-phorylation |
|
|
NMDAR |
|
Carbofuran was administered respectively into the rats once a day for 28 days by gavage. Pesticide exposure induced spatial learning and memory deficits with a simultaneous decrease of NMDAR1, synaptophysin, and synapsin I, all of which are memory-related synaptic proteins [25]. |
|
|
Synaptic dysfunction |
| Cadmium (Cd) |
Heavy metals |
PI3K/Beclin/BCL2 signaling |
p-tau induction and PI3K/Beclin/BCL2 signaling followed by excessive autophagy and apoptosis |
1. Exposure of human SH-SY5Y cells to Cd increased intracellular ROS levels [26]. |
|
|
Oxidative stress, Dysfunctional autophagy |
|
|
|
|
2. Cd exposure to rat cerebral cortical neurons induced cytoprotective autophagy by activating the class III PI3K/beclin-1/Bcl-2 signaling pathway [27]. |
| Chlorophenotane |
Pesticides |
CYP51, DHCR7? |
Impairment of cholesterol biosynthesis |
Acute sublethal and chronic administration of chlorophenotane (DDT) decreased brain lipid metabolism of rhesus monkeys [28]. |
|
|
Cholesterol dysmetabolism |
| Chlorpyrifos |
Organophos-phate pesticides |
AChE |
Irreversible binding between AChE and OP pesticides due to phosphorylation of enzyme. AChE inhibitors (pesticides) bind to the enzyme and interfere with the breakdown of AChE, leading to deposition of AChE in nerve synapses, causing disrupted neurotransmission. |
1. Estuarine fish (Oreochromis mossambicus) to a 24 h LC50 concentration of chlorpyrifos, after 6 h reached > 40% AChE inhibition while after 24 h reached 90% AChE inhibition [29]. |
MIE: Event: 12 Acetylcholinesterase (AChE), Inhibition |
AOP16: Acetylcholinesterase inhibition leading to acute mortality,
AOP405
: Organo-Phosphate Chemicals induced inhibition of AChE leading to impaired cognitive function |
Impaired cognitive function |
|
|
|
|
2. An acute sublethal exposure of chlorpyrifos to Sprague-Dawley rats increased inhibition of AChE, increased levels of acetylcholine, and significantly impacted to motor activity [30]. |
|
|
PI3K-AKT |
Chronic cholinergic activity linked to defective PI3K-AKT pathway activation |
Chlorpyrifos exposure of human neural precursor cells (hNPCs) derived from human embryonic stem cells (hESCs) reduced the expression of AKT and ERK proteins involved in intracellular survival pathways [31]. |
|
|
Glucose metabolism |
|
|
NMDAR |
Glutamate excitotoxicity leading to neuronal damage |
Chlorpyrifos -induced neurotoxicity after CPF exposure with and without Ifenprodil (IFN) on 4-week differentiated human neural progenitor stem cell culture model (ReNcell CX) [32]. |
|
|
Synaptic dysfunction |
|
|
NFkB |
Release IL1β and TNFα? |
Chlorpyrifos exposure of human neural precursor cells (hNPCs) derived from human embryonic stem cells (hESCs) induced nuclear accumulation of NFκB manner via ROS generation in a concentration-dependent [31]. |
|
|
Neuroinflammation |
|
|
LC3-II expression |
Oxidative stress induced increased in numbers of autophagic vacuoles, autophagy, and apoptosis |
1. Chlorpyrifos-induced cytotoxicity in human SH-SY5Y cells, and induced autophagic cell death by upregulating the expression of LC3-II and p62 [33]. |
|
|
Oxidative stress, Dysfunctional autophagy |
|
|
|
|
2. Chlorpyrifos generated oxidative stress and lipid peroxidation in different rat cell types causing neuronal damage by elevating the production of ROS, DNA damage, and lipid peroxidation in the CNS [34, 35]. |
| Conazole |
Fungicides |
CYP51, DHCR7 |
Impairment of cholesterol biosynthesis |
Inhibits CYP51 by coordinating with the heme group, which halts substrate binding with a resulting increase of lanosterol in mouse Neuro2a cells [36]. |
|
|
Cholesterol dysmetabolism |
| Copper (Cu) |
Metals |
LC3-II expression |
Oxidative stress induced increased in numbers of autophagic vacuoles, autophagy, and apoptosis |
CuCl2 induced a dose-dependent accumulation of the autophagosome marker, LC3-II, in human neuroblastoma cell line SK-N-SH [37]. |
|
|
Oxidative stress, Dysfunctional autophagy |
| Deltamethrin |
Pesticides |
PP2A, GSK3β |
Disturbed mitochondrial ETC, low ATP levels, increased oxidative stress, intracellular Ca2+ levels and tau hyperphosphorylation. Reduced dephosphorylation supporting tau hyperphosphorylation. |
Exposure of deltamethrin (30μM), PP2A/PP2B inhibitors, induced tau aggregation in human SH-SY5Y tau-BiFC cells and in HEK293 tau-BiFC cells. |
|
|
Mitochondrial dysfunction, Oxidative stress, tau hyperphos-phorylation |
| Dibenzothiopene |
Alkyl-polycyclic aromatic compounds (PACs) |
CYP1A1, CYP1B1 |
Oxidation of macromolecules before conjugation with glutathione, decreased cellular ATP production, MMP, oxidative phosphorylation and mitochondrial protein complexes I, II, and IV activity, and eventually apoptosis |
Exposure of human neuroblastoma SK-N-SH cells to dibenzothiophene (at 0.5–40μM for 24 h) increased in levels of ROS, significantly decreased mitochondrial membrane potential, upregulated pro-apoptotic genes and down-regulated antioxidative genes expression, elevated cytochrome c protein levels and lipid peroxidation in a concentration-related manner [24]. |
|
|
Mitochondrial dysfunction, Oxidative stress |
| Dichlorvos |
Organophos-phate pesticides |
AChE |
Irreversible binding between AChE and OP pesticides is due to phosphorylation of enzyme. The AChE inhibitors (pesticides) bind to the enzyme and interfere with the breakdown of ACh, leading to the deposition of ACh in the nerve synapses and causing disrupted neurotransmission |
Rats injected with a sublethal concentration of dichlorvos found a significant decrease in AChE activity, increased ACh concentrations, and enhanced contractile responses in jejunum muscle. At sublethal concentrations (56% of the LD50), researchers found a significant (18%) increase in the amount of ACh in brain tissue of rats exposed to disulfoton for 3 days and resulted in AChE inhibition of 68% with respect to controls [38]. |
MIE: Event: 12 Acetylcholinesterase (AChE), Inhibition |
AOP405
: Organo-Phosphate Chemicals induced inhibition of AChE leading to impaired cognitive function
|
Impaired cognitive function |
| Dieldrin |
Organochloride insecticides |
iGABARs |
Directly blocks chloride conductance through the ion channel |
Long-term exposure of mouse primary cerebellar granule cell cultures to 3μM dieldrin reduced the GABAA receptor function to 55% of control, as measured by the GABA-induced 36Cl- uptake [39]. |
MIE: Event: 667 iGABAR chloride channel, Binding at picrotoxin site |
AOP10: Blocking iGABA receptor ion channel leading to seizures |
Seizures |
| Domoic acid (DomA) |
Kainic acid-type neurotoxin (ASP) |
COX2 |
|
1. DomA (4 mg/kg at 30, 60 and 240 min post-injection) promoted the expression of early inflammatory genes in mouse brain, such as COX2 and the development of neurodegeneration [40]. |
|
AOP48: Binding of agonists to ionotropic glutamate receptors in adult brain causes excitotoxicity that mediates neuronal cell death, contributing to learning and memory impairment. |
Neuroinflammation |
|
|
|
|
2. DomA treatment (2 mg/kg per day for 3 weeks) in mice significantly stimulated the expression of inflammatory mediators, including IL-1β (1.7 fold increase), TNF-α (2 fold increase), GFAP (1.4 fold increase), Cox-2 (3 fold increase), and iNOS (1.6 fold increase) compared to controls [41]. |
|
|
|
|
3. DomA (0.75 mg/kg body weight) when administered intravenously in adult rats induced neuronal degeneration followed by glial activation [42]. |
|
|
iGlu, NMDRAs, KA &ARs |
DomA induces excitotoxicity by an integrative action on ionotropic glutamate receptors at pre- and post-synaptic sides. DomA directly activates KA/AMPARs receptors followed by indirect activation of the NMDARs |
1. In rat hippocampal neurons at 5 days after DomA administration, NMDAR1 immunoreactivity increased, and glutamate receptor genes were induced in response to DomA-induced neurotoxicity [43]. |
MIE: Event: 875 Ionotropic glutamate receptors, Binding of agonist |
|
Synaptic dysfunction |
|
|
|
|
2. DomA exposure of immature and mature primary cultures of neurons and glial cells from rat cerebellum induced neurotoxicity mediated by the AMPA/KA receptors [44]. |
| Efavirenz |
Drug |
CYP46A1 |
Increased 24S-OHC levels driving increased cholesterol turn-over and hypocholesterolemia |
Efavirenz a CYP46A1 activator activated the enzyme in mouse brains at low concentrations (up to 20μM) but inhibited the P450 at higher concentrations. It increased the formation of cholesterol 24-hydroxylation in isolated mouse brain microsomes [13]. |
|
|
Cholesterol dysmetabolism |
| Endosulfan |
Organochloride insecticide |
iGABARs |
Non-competitive ion channel blocker |
Poisoning with endosulfan caused seizure, status epilepticus, or refractory status epilepticus in humans [45, 46], and eventually led to the death of a farmer [45] and a toddler [47]. |
MIE: Event: 667 iGABAR chloride channel, Binding at picrotoxin site |
AOP10: Blocking iGABA receptor ion channel leading to seizures |
Seizures |
|
|
CYP51, DHCR7 |
Impairment of cholesterol biosynthesis C |
Exposure of mouse Neuro-2a cells to endosulfan (1.1μM) induced elevation of lanosterol by inhibiting either CYP51 or DHCR7 [48]. |
|
|
Cholesterol dysmetabolism |
| Felodipine |
Drugs |
CYP27A1 |
Reduced 27-OHC production, elevated cholesterol biosynthesis and reduced steroidal acid production |
Felodipine administration to mice at a 1 mg/kg of body weight/day for 7 days induced reduction in 27-hydroxycholesterol levels in plasma, brain and liver, whereas tissue levels of total cholesterol were unchanged [49]. |
|
|
Cholesterol dysmetabolism |
| Fenobucarb, Propoxur |
Organophosphate pesticides |
AChE |
Irreversible binding between AChE and OP pesticides is due to phosphorylation of enzyme. The AChE inhibitors (pesticides) bind to the enzyme and interfere with the breakdown of ACh, leading to the deposition of ACh in the nerve synapses and causing disrupted neurotransmission. |
Female mice exposed to either fenobucarb or propoxur, reported a major increase in AChE in brain tissue 10 minutes after injection, simultaneously major elevation in AChE inhibition [50]. |
MIE: Event: 12 Acetylcholinesterase (AChE), Inhibition |
AOP405
: Organo-Phosphate Chemicals induced inhibition of AChE leading to impaired cognitive function (AOP under development)
|
Impaired cognitive function |
| Fenpropimorph |
Fungicides |
CYP51, DHCR7 |
Impairment of cholesterol biosynthesis |
Exposure of human cell lines and human neuroprogenitor model derived from hiPSCs to fenpropimorph caused hypocholesterolemia, decreased absolute cholesterol levels, inhibited CYP51 and elevated 7-DHC levels [48]. |
|
|
Cholesterol dysmetabolism |
| Fipronil |
Insecticides |
iGABARs, Cl- channels regulated by GABA receptors |
Directly blocks chloride conductance through the ion channel. |
Acute human intoxication with fipronil revealed symptoms associated with the GABA transmission within the central nervous system, including seizure, agitation, and headache. In two patients, plasma fipronil concentrations were measured at 1600 and 3744μg/L. In another patient, a peak measured plasma concentration at 1040μg/L [51]. |
MIE: Event: 667 iGABAR chloride channel, Binding at picrotoxin site), Event: 64 iGABAR chloride conductance, Reduction |
AOP10: Blocking iGABA receptor ion channel leading to seizures |
Seizures |
|
|
GSK3β |
Alteration of AKT/GSK3β phosphorylation |
In human dopaminergic SH-SY5Y cells, fipronil exposure altered the level of Akt/GSK3β phosphorylation, reduced the Akt phosphorylation on Ser473, and in parallel with the inactivation of Akt, phosphorylation of GSK3β on Ser9 which inactivated GSK3β, decreased after treatment [52]. |
|
|
Tau hyperphos-phorylation |
|
|
LC3-II &Beclin-1 expression |
Oxidative stress induced increased in numbers of autophagic vacuoles, autophagy, and apoptosis |
1. Exposure to fipronil in human neuroblastoma SH-SY5Y cells induced autophagic death by monitoring LC3-Ii and Beclin-1 expression [53]. |
|
|
Oxidative stress, Dysfunctional autophagy |
|
|
|
|
2. Fipronil exposure to human dopaminergic SH-SY5Y cells induced dopaminergic cell death involved in increase of ROS generation [52]. |
| Galantamine |
Drugs |
CYP46A1 |
Increased 24S-OHC levels driving increased cholesterol turn-over and hypocholesterolemia |
Galantine treatment in isolated bovine brain microsomes stimulated CYP46A1 activity by 6–7-fold [13]. |
|
|
Cholesterol dysmetabolism |
| Glufosinate |
Phosphorous herbicides, Fungicides |
NMDARs |
Agonist action at the NMDAR is expected to occur through direct interaction with the glutamate binding site and requires binding of the glycine co-agonist as well as release of the magnesium block from the channel pore. |
1. In one human poisoning case (267 mg/kg d,l-glufosinate, oral ingestion), the concentrations respectively of d- and l-GLF 1 h after exposure were 1050 and 1070μM in plasma and after 27 h were 7.95 and 1.93μM in plasma and 2.6 and 0.66μM in cerebrospinal fluid, respectively. d,l-Glufosinate concentrations in excess of 100μM are needed to affect the NMDAR [54]. |
MIE: Event: 875 Ionotropic glutamate receptors, Binding of agonist |
AOP48: Binding of agonists to ionotropic glutamate receptors in adult brain causes excitotoxicity that mediates neuronal cell death, contributing to learning and memory |
Synaptic dysfunction |
|
|
|
|
2. Chronic exposure to glufosinate induced structural changes in the NMDAR rich hippocampal region of the mouse brain, disrupting activation of NMDARs [55]. |
| Iron (Fe) |
Metals |
LC3-II &Beclin expression |
Oxidative stress induced increased in numbers of autophagic vacuoles, autophagy, and apoptosis |
Fe given in the neonatal period in rats (single daily oral dose of vehicle or iron carbonyl (30 mg/kg) at postnatal days 12–14) impaired inhibitory avoidance memory and induced a decrease in proteins critically involved in the autophagy pathway, Beclin-1 and LC3, in hippocampus [56]. |
|
|
Oxidative stress, Dysfunctional autophagy |
| Ketamine |
Anaesthetics |
NMDARs |
Blocking NMDA receptor channel activity |
In humans, ketamine exposure produced profound decrements in both attention and memory [57, 58] |
|
|
Synaptic dysfunction |
| Lindane |
Insecticides |
iGABARs |
Non-competitive ion channel blocker |
Toxic doses of lindane produced neuronal hyperexcitability in humans and other vertebrates by inhibiting both glycine and GABAA receptors [59]. |
|
|
Synaptic dysfunction |
| Lead (Pb) |
Heavy metals |
BDNF |
Presynaptic effects mediated by disruption of NMDAR-activity dependent BDNF signaling by inhibiting BDNF activity |
In primary rat hippocampal neurons exposed to 1μM Pb2+ for 5 days during the period of synaptogenesis (DIV7–DIV12), decreased cellular pro-BDNF protein (40% compared to control) and extracellular levels of mBDNF (25% compared to control [60]. |
Event: 389 (Release of BDNF, Reduced) |
AOP13: Binding of antagonist to NMDARs impairs cognition, AOP54: NIS inhibition and DNT effects, AOP12: Binding of antagonist to NMDARs can lead to neuroinflammation and neurodegeneration |
Synaptic dysfunction |
|
|
NMDAR |
Glutamate excitotoxicity leading to neuronal damage |
1. In rat hippocampal neurons, Pb2+ (2.5–50μM) inhibited NMDA-induced whole-cell and single-channel currents in a concentration-dependent manner, suggesting that Pb2+ can decrease the frequency of NMDA-induced channel activation. Pb2+ also affected the binding of [3H]MK-801 to the rat brain hippocampal membranes [61]. |
Event:195 NMDARs, Inhibition, Event: 201 NMDA receptors, Binding of antagonist |
AOP12: Binding of antagonist to NMDARs can lead to neuroinflammation and neurodegeneration |
Synaptic dysfunction, Neuroinflammation, Neurodegeneration |
|
|
|
|
2. Studies in animal models of developmental Pb2+ exposure exhibit altered NMDAR subunit ontogeny and disruption of NMDAR-dependent intracellular signaling [60]. |
|
|
Zn2+ regulatory site of the NMDAR |
Potent, non-competitive antagonist of the NMDAR binding at the Zn2+ regulatory site of the NMDAR in a voltage-independent manner causing inhibition of Ca2+ channels, presynaptic neurotransmission and NMDARs signaling |
Pb2+ exposure decreased Ca2+ ion concentration and increased Ca2+ efflux by a calmodulin-dependent mechanism in embryonic rat hippocampal neurons [62]. |
Event:52 Ca2+ influx, decreased |
|
Synaptic dysfunction? |
|
|
IL-1b, TNF-a |
Proinflammatory cytokines |
In vivo and in vitro models (rats), Pb exposure caused microglial activation, which upregulated the levels of pro-inflammatory cytokines IL-1β, TNF-α and of iNOS and caused neuronal injury and neuronal death in hippocampus [63]. |
Event:188 Neuroinflammation |
|
Neuroinflammation |
|
|
Tau |
Phosphorylation of tau |
1. Monkeys exposed to Pb 1.5 mg/kg/day from birth to 400 days at 23 years of age tau accumulation, Overexpression of amyloid-beta protein precursor and of amyloid-beta enhanced pathologic neurodegeneration [64]. |
|
|
Tau hyperphospho-rylation? |
|
|
|
|
2. Mice exposed toPb 0.2% in drinking water from PND 1–20 or from PND 1–20 and from 3–7 months at 700 days of age, elevated protein and mRNA for tau and aberrant site-specific tau [65]. |
|
|
|
|
3. Perinatal exposure to Pb leading to a blood concentration of 10μg/dl promoted tau phosphorylation in rat forebrain, cerebellum and hippocampus [66]. |
|
|
|
|
4. Chronic exposure of rats to Pb via drinking water induced hyperphosphorylation of tau and excessive increase in autophagy, which might induce cell-programmed death and increase neurotoxicity [67]. |
|
|
Beclin-1/ LC3-II signaling, Akt/mTOR pathway |
Beclin-1/LC3-II signaling followed by excessive autophagy |
Pb exposure induced autophagy in astrocytes, by increased LC3II and Beclin 1 protein levels in both the rat hippocampus and astrocytes through blocking the downstream Akt/mTOR pathway in astrocytes [68]. |
|
|
Oxidative stress, Dysfunctional autophagy |
|
|
PI3K-Akt signaling |
|
Pb exposure induced a decrease in rat hippocampal glucose metabolism by reducing GLUT4 levels in the cell membrane through the PI3K-Akt pathway. In vivo and in vitro GLUT4 over-expression increased the membrane translocation of GLUT4 and glucose uptake, and reversed the Pb-induced impairment to synaptic plasticity and cognition [69]. |
|
|
Glucose dysmetabolism? |
| Malathion |
Organophosphate pesticides |
AChE |
Irreversible binding between AChE and OP pesticides is due to phosphorylation of enzyme. The AChE inhibitors (pesticides) bind to the enzyme and interfere with the breakdown of AChE, leading to the deposition of AChE in the nerve synapses and causing disrupted neurotransmission |
1. In rats, administration of malathion induced reduction of plasma acetylcholinesterase activity [70]. |
MIE: Event: 12 Acetylcholinesterase (AChE), Inhibition |
AOP405
: Organo-Phosphate Chemicals induced inhibition of AChE leading to impaired cognitive function (AOP under development)
|
Impaired cognitive function |
|
|
|
|
2. In silkworm exposed to malathion for 5 days increased mortality, decreased AChE, and increased in AChE as compared to controls [71]. |
|
|
GSK3β, PP2A |
Reduced dephosphorylation supporting tau hyperphosphorylation |
In rats, administration of malathion induced spatial learning and memory deficits with a simultaneous decrease of PSD93 and tau hyperphosphorylation at multiple AD-related phosphorylation sites with activation of GSK-3β and inhibition of PP2A [70]. |
|
|
Tau hyperphospho-rylation |
|
|
SH containing proteins |
Depletion of glutathione buffer, oxidation of macromolecules, and disturbed cellular redox homeostasis |
1. Subchronic exposure of rats to malathion increased malondialdehyde and 8-OHdG levels, whereas it decreased glutathione levels, also acetylcholinesterase, superoxide dismutase, and catalase activities in the blood and brain tissues [72]. |
|
|
Mitochondrial dysfunction, Oxidative stress |
|
|
|
|
2. In rats, administration of malathion induced amelioration of reduced GSH in hippocampus [70]. |
|
|
TNF-a, IL-6 |
Proinflammatory cytokines |
In rats, administration of malathion induced elevation of TNF α and IL-6 levels in the hippocampus [70]. |
|
|
Neuroinflammation |
| Methylmercury (MeHg) |
Metals |
Beclin-1 expression |
Oxidative stress induced increased in numbers of autophagic vacuoles, autophagy, and apoptosis |
1. Exposure of human neural stem cells to MeHg upregulated autophagy by inhibiting mTOR levels and by enhancing Atg6/Beclin-1 protein levels [73]. |
|
|
Oxidative stress, Dysfunctional autophagy |
|
|
|
|
2. Oxidative stress has been suggested to enhance autophagy signaling pathway, as the underlying mechanism of MeHg-induced neurotoxicity [74]. |
|
|
m-TOR expression |
|
|
LC3-II expression |
|
In rat astrocytes, MeHg decreased cell viability in a concentration- and time-dependent manner, with induction of both apoptosis and autophagy via increasing the LC3-II–LC3-I ratio, and of Beclin-1 protein [75]. |
| Methyl parathion |
Organophosphate pesticides |
AChE |
Irreversible binding between AChE and OP pesticides is due to phosphorylation of enzyme. The AChE inhibitors (pesticides) bind to the enzyme and interfere with the breakdown of ACh, leading to the deposition of ACh in the nerve synapses and causing disrupted neurotransmission |
Sublethal exposure (12–48 h) to methyl parathion highly inhibited AChE levels in brain tissue in fish, (Oreochromis mossambicus) with inhibition increasing from 36–62% as in comparison to controls over the time elapse [76]. |
MIE: Event: 12 Acetylcholinesterase (AChE), Inhibition |
AOP405
: Organo-Phosphate Chemicals induced inhibition of AChE leading to impaired cognitive function (AOP under development)
|
Impaired cognitive function |
| Mirtazapine |
Drugs |
CYP46A1 |
Increased 24S-OHC levels driving increased cholesterol turn-over and hypocholesterolemia |
Exposure of bovine isolated brain microsomes to mirtazapine increased CYP46A1 activity [13]. |
|
|
Cholesterol dysmetabolism |
| Manganese (Mn) |
Metals |
SH containing proteins |
Depletion of glutathione buffer, oxidation of macromolecules, and disturbed cellular redox homeostasis |
1. Exposure of rat-derived mesencephalic dopaminergic neuronal (N27) cells to Mn induced ROS formation [77]. |
|
|
Mitochondrial dysfunction, Oxidative stress, tau hyperphos-phorylation |
|
|
|
|
2. Exposure of rat primary striatal neurons to Mn induced a dose-dependent decrease in MMP and complex II activity [78]. |
|
|
LC3-II &Beclin-1 expression |
Oxidative stress induced increased in numbers of autophagic vacuoles, autophagy, and apoptosis |
After a single intrastriatal injection of Mn, the short- (4–12 h) and long-term (1–28 days) effect of Mn on rat dopaminergic neurons, increased number of abnormal lysosomes, decreased protein expression of Beclin-1, and decreased ratio of LC3 II over LC3 I, concomitant with activated mTOR/p70s6k signaling [79]. |
|
|
Oxidative stress, Dysfunctional autophagy |
| Nilvadipine |
Drugs |
CYP27A1 |
Reduced 27-OHC production, elevated cholesterol biosynthesis and reduced steroidal acid production |
Administration of nilvadipine to mice at a 1 mg/kg of body weight per day, for 7 days, reduced 27-hydroxycholesterol levels in the plasma, brain, and liver, whereas tissue levels of total cholesterol were unchanged [49]. |
|
|
Cholesterol dysmetabolism |
| Organochlorine pesticides (OCPs) |
Pesticides |
CYP46A1 |
Increased 24S-OHC levels driving increased cholesterol turn-over and hypocholesterolemia |
In human studies, exposure of OCPs induced elevated levels of the total cholesterol in blood [80]. |
|
|
Cholesterol dysmetabolism |
| Okadaic acid (OKA) |
Algal toxins |
GSK3β, PP2A |
Tau hyperphosphorylation |
Administration of OKA in rats increased tau gene and protein expression, Ca2+/ CAMKII, calpain and GSK3β in the hippocampus and cerebral cortex, while decreased the PP2A gene and protein expression in these brain regions [81]. |
|
|
Tau hyperphos-phorylation |
|
|
|
Disturbed mitochondrial ETC, low ATP levels, increased oxidative stress, intracellular Ca2+ levels |
Intracerebroventricular administration of OKA increased intracellular Ca2+, impairing the mitochondrial ETC and generating intracellular ROS and RNS (reactive nitrogen species) in rat brain areas [82]. |
|
|
Mitochondrial dysfunction, Oxidative stress |
|
|
NMDARs |
|
Involvement of NMDA receptor in OKA intracerebroventricular -induced tau hyperphosphorylation in rat brain areas [81]. |
|
|
Synaptic dysfunction |
| Paraoxon |
Organophosphate pesticides |
AChE |
Irreversible binding between AChE and OP pesticides is due to phosphorylation of enzyme. The AChE inhibitors (pesticides) bind to the enzyme and interfere with the breakdown of AChE, leading to the deposition of AChE in the nerve synapses and causing disrupted neurotransmission |
Inhibition of striatal AChE activity and decreased extracellular AChE levels in rats intracerebrally perfused after exposure to paraoxon (0, 0.03, 0.1, 1, 10 or 100μM, 1.5μl/min for 45 min) [83]. |
MIE: Event: 12 Acetylcholinesterase (AChE), Inhibition |
AOP405
: Organo-Phosphate Chemicals induced inhibition of AChE leading to impaired cognitive function (AOP under development)
|
Impaired cognitive function |
| Paraquat |
Pesticides |
NFkB |
Release IL1β, IL6 and TNFα |
1. Upon paraquat exposure, HMGB1 increased, translocated into cytosol and then released to the extracellular milieu of human neuroblastoma SH-SY5Y cells, via activation of RAGE-P38-NF-κB signaling pathway and the expression of inflammatory cytokines such as TNF-α and IL-6 [84]. |
|
|
Neuroinflammation |
|
|
|
|
2. Paraquat-induced ROS inhibited human blood neutrophil apoptosis via a p38 MAPK/NF-κB–IL-6/TNF-α [85]. |
| Parathion |
Organophosphate insecticides and acaricides |
AChE |
Irreversible binding between AChE and OP pesticides is due to phosphorylation of enzyme. The AChE inhibitors (pesticides) bind to the enzyme and interfere with the breakdown of AChE, leading to the deposition of AChE in the nerve synapses and causing disrupted neurotransmission. |
An acute (48 h) sublethal exposure to methyl parathion found that AChE levels in brain tissue in fish (Oreochromis mossambicus) were significantly inhibited at all measured durations ranging from 12–48 h with inhibition increasing from 36–62% as compared to controls over the time span [76]. |
MIE: Event: 12 Acetylcholinesterase (AChE), Inhibition |
AOP16
: Acetylcholinesterase inhibition leading to acute mortality
|
| Polychlorinated Biphenyls (PCBs) |
Persistent organic pollutants (POPs) |
CYP450 |
Oxidation of macromolecules before conjugation with GSH, decreased cellular ATP production, MMP, oxidative phosphorylation and mitochondrial protein complexes I, II, and IV activity, and eventually apoptosis. |
Chronic exposure of human SH-SY5Y cells to PCBs decreased cellular ATP production, MMP, oxidative phosphorylation and the activity of mitochondrial protein complexes I, II, and IV, resulting in mitochondrial dysfunction [86]. |
|
|
Mitochondrial dysfunction, Oxidative stress |
|
|
HMG-CoA reductase |
Increased cholesterol serum levels result in impaired cholesterol biosynthesis |
In human studies, exposure of PCBs induced elevated levels of the total cholesterol in blood [80]. |
|
|
Cholesterol dysmetabolism |
| Particle matter (PM) &Sulfur dioxide (SO2) |
Air pollutants |
PSD-95, NR2B, BDNF, Synp, tau, expression |
Reduction in these synaptic markers reflect reduction in synaptic activity |
1. PM2.5 and SO2 co-exposure led to neurodegeneration at low doses, reduced synaptic structural protein PSD-95 and NMDA receptor subunits (NR2B), and elevated tau phosphorylation, in vitro (mouse primary cortical neuron culture) and in vivo (mice) [87]. |
|
|
Synaptic dysfunction, tau hyperphos-phorylation |
|
|
|
|
2. Human cocultured neurons and astrocytes with PM2.5 treatment exhibited reduction in the number of synapsin I by ≈49.6% compared to the nontreated cocultured model [88]. |
|
|
|
|
3. Humanin utero exposure to PM2.5 was inversely associated with placental BDNF expression [89]. |
|
|
IL-1β |
Proinflammatory cytokines |
Human astrocytes cocultured with neurons were activated by PM2.5 treatment produced significant levels of proinflammatory chemokines (CCL1 and CCL2) and cytokines (IL-1β and IFN-γ), which can recruit and activate microglia [88]. |
|
|
Neuroinflammation |
| Profenofos (PFF) |
Organophosphate insecticides |
AChE |
Irreversible binding between AChE and OP pesticides is due to phosphorylation of enzyme. The AChE inhibitors (pesticides) bind to the enzyme and interfere with the breakdown of AChE, leading to the deposition of AChE in the nerve synapses and causing disrupted neurotransmission. |
A time course study of earthworms (Eisenis foetida) exposed to the 4.56±0.14 and 3.55±0.10μg cm–2 for 24 and 48 h (LC50), respectively, of PPF found a significant relationship between increases in percent inhibition of AChE and increase in time of exposure from 8–48 h [90]. |
MIE: Event: 12 Acetylcholinesterase (AChE), Inhibition |
AOP16
: Acetylcholinesterase inhibition leading to acute mortality |
| Propofol |
Sedative drugs |
HMG-CoA reductase |
Increased cholesterol serum levels result in impaired cholesterol biosynthesis |
Treatment of rats with propofol induced increased levels of serum total cholesterol, possibly due to the increase in HMG-CoA reductase activity, a rate limiting step in cholesterol biosynthesis. Propofol at 2 and 4 mg/kg increased the levels of serum total cholesterol by 74% and 55%, triglycerides by 97% and 115%, and LDL-C by 45% and 73%, respectively, while HDL-C decreased by 41% and 54%, respectively [91]. |
|
|
Cholesterol dysmetabolism |
| RDX |
|
iGABARs |
Directly blocks chloride conductance through the ion channel. |
1. Human exposure to high doses of RDX causes headache, dizziness, vomiting, and confusion, followed by tonic-clonic seizures [92]. 2. RDX binds to GABAA) receptor causing reduction of GABAA receptor–mediated synaptic transmission and induction of epileptiform activity, in the amygdala, a seizure-prone structure of the limbic system, in rats [92]. |
Event: 667 iGABAR chloride channel, Binding at picrotoxin site |
AOP10: Blocking iGABA receptor ion channel leading to seizures |
Seizures |
| Retene |
Alkyl-polycyclic aromatic compounds (PACs) |
CYP1A1, CYP1B1 |
Oxidation of macromolecules before conjugation with GSH, decreased cellular ATP production, MMP, oxidative phosphorylation and mitochondrial protein complexes I, II, and IV activity |
Exposure of human neuroblastoma SK-N-SH cells to retene at 0.5–40μM for 24 h, increased in levels of ROS, significantly decreased MMP, up-regulated pro-apoptotic genes and down-regulated antioxidative genes expression, elevated cytochrome c protein levels and lipid peroxidation in a concentration-related manner [24]. |
|
|
Mitochondrial dysfunction, Oxidative stress |
| Rotenone |
Isoflavone pesticides, insecticides, piscicides |
LC3-II expression, Beclin-1 expression, mTOR |
Oxidative stress induced increased in numbers of autophagic vacuoles, autophagy, and apoptosis |
1. Robust increase in steady state expression of LC3 (LC3I and LC3II), upon rotenone treatment compared to untreated cells, and a significant decrease in phosphorylation of Akt and beclin1, in human SHSY-5Y cells [93]. 2. Exposure to rotenone at a higher dose (10μM) decreased mTORC1 activity, in human SHSY-5Y cells. Rotenone-treated cells showed ∼2 fold increase in ROS generation compared to untreated cells [93]. |
|
|
Oxidative stress, Dysfunctional autophagy |
|
|
Complex I |
Inhibition of ETC, increased Ca2+ levels and reduced AKT phosphorylation, Mitochondrial complex I inhibitor |
Mouse brain concentrations of rotenone (20–30 nM) triggered approximately a 50% inhibition of Complex I [12]. |
Event: 177 Mitochondrial dysfunction, Event: 887 NADH-ubiquinone oxidoreductase (complex I), Inhibition |
AOP48: Binding of agonists to ionotropic glutamate receptors in adult brain causes excitotoxicity that mediates neuronal cell death, contributing to learning and memory impairment. AOP3: Mitochondrial dysfunction and Neurotoxicity |
Glucose dysmetabolism via mitochondrial dysfunction |
| Silica (SiO2-NPs), Silver (AgNPs) nanoparticles |
Nanoparticles |
PSD-95, NR2B expression |
Reduction in these synaptic markers reflect reduction in synaptic activity |
1. In brains of immature rats subjected to a low dose of AgNPs, decreased expression of several NMDA receptor complex-related proteins, such as GluN1 and GluN2B subunits, scaffolding proteins PSD95 and SynGAP, as well as neuronal nitric oxide synthase (nNOS) [94]. |
|
|
Synaptic dysfunction |
|
|
|
|
2. AgNPs oral exposure significantly decreased levels of the presynaptic proteins synapsin I and synaptophysin, as well as PSD-95 protein which is an indicator of postsynaptic densities, in hippocampal region of rat brain [95]. |
|
|
GSK3β |
Induction of tau hyperphosphorylation |
Exposure of human SK-N-SH to SiO2-NPs significantly increased the intracellular levels of ROS and the hyperphosphorylation of tau at Ser262 and Ser396 accompanied by increased GSK3β activity [96] |
|
|
Tau hyperphospho-rylation |
|
|
APP |
|
Immunofluorescent staining indicated a significantly increased number of in cells containing intracellular Aβ1-42 positive deposit and upregulated APP and downregulated Aβ-degrading enzyme neprilysin. in SiNP-treated human SK-N-SH and mouse N2a cells in comparison to the control or micro-sized SiO2-treated cells [96]. |
|
|
Intracellular Aβ |
| Sodium azide (NaN3) |
Gas-forming inorganic compound |
Complex IV |
Inhibition of the mitochondrial ETC, oxidative stress |
Exposure of in rat PC12 cells to NaN3 inhibited cytochrome oxidase in the mitochondrial ETC and accumulation of mitochondrial ROS. NaN3-induced apoptosis was associated with the expression levels of Pgc-1α family proteins and Cox IV in mitochondria-mediated signaling pathway [97]. |
|
|
Mitochondrial dysfunction, Oxidative stress |
| Spiroxamine |
Fungicides |
CYP51, DHCR7 |
Impairment of cholesterol biosynthesis |
Exposure of human hiPSCs and mouse neuroblastoma Neuro-2a cells to spiroxamine induced decreased cholesterol biosynthesis either by inhibiting CYP51 or DHCR7 [48]. |
|
|
Cholesterol dysmetabolism |
| Taxol &epothilones |
Microtubule interacting drugs (MSAs) |
Tubulin (TUBB) |
MSAs bind to polymerized tubulin. Impairment in axonal transport leads to an inadequate supply of the neuronal periphery. |
1. Disruption of microtubule dynamic instability decreased transport of horseradish peroxidase in dorsal root ganglia neurons resulting in less microtubule crosslinks. Intact axonal transport regained after taxol wash-out (1 day treatment, 2 days wash-out) [98]. |
|
AOP279
: Microtubule interacting drugs lead to peripheral neuropathy
|
Impaired axonial transport |
|
|
|
|
2. Taxol inhibited anterograde fast (and retrograde) axonal transport in rat sciatic nerves [99] and anterograde transport in SK-N-SH human neuroblastoma cells and mice sciatic nerves [100]. |
|
|
|
|
3. Suppressed microtubule dynamic instability had inhibitory effects on anterograde fast axonal transport in isolated squid axoplasm [101]. |
| 2,3,7,8-tetrachloordi-benzo-p-dioxine (TCDD) |
Dioxin-like compounds |
LC3-II expression |
Oxidative stress induced increased in numbers of autophagic vacuoles, autophagy, and apoptosis |
In human neuronal cell line SHSY5Y, expression of LC3II was significantly increased in cells exposed to 200 nM TCDD, compared with control cells [102]. |
|
|
Oxidative stress, Dysfunctional autophagy |
|
|
AHR |
Potent aryl hydrocarbon receptor (AHR) ligands |
TCDD toxicity in human SHSY5Y neuroblastoma cells depended on dioxin concentration and time of incubation, with a main role of aryl hydrocarbon receptor at low nanomolar TCDD concentrations (3 nM at 24 h). Induced apoptosis by the disruption of calcium homeostasis, affecting membrane structural integrity [103]. |
MIE: Event:165 Activation, Long term AHR receptor driven direct and indirect gene expression changes, Event: 18 Activation, AhR |
AOP41
: Sustained AhR Activation leading to Rodent Liver Tumours,
AOP150
: Aryl hydrocarbon receptor activation leading to early life stage mortality, via reduced VEGF,
AOP21
: Aryl hydrocarbon receptor activation leading to early life stage mortality, via increased COX-2,
AOP131
: Aryl hydrocarbon receptor activation leading to uroporphyria |
Oxidative stress |
|
|
GSK3β |
tau hyperphosphorylation |
Acute exposure to TCDD (25μg/kg body weight) induced neuronal toxicity in the cortex of female Sprague-Dawley rats. Exposure of rat PC12 cells to TCDD induced activation of GSK3β and decreased β-catenin in neural cells [104]. |
|
|
Tau hyperphospho-rylation |
| Tri-ortho-cresyl phosphate (TOCP) |
Organophos-phorus based compound |
LC3-II &Beclin expression |
Oxidative stress induced increased in numbers of autophagic vacuoles, autophagy, and apoptosis |
Low concentrations of TOCP induced autophagy and inhibited neurite outgrowth in a dose-dependent manner in human neuroblastoma SH-SY5Y cells. After treatment with TOCP, both LC3-II and the ratio of LC3-II/LC3-I, and autophagy proteins Atg5 and Beclin 1 were increased [105]. |
|
|
Oxidative stress, Dysfunctional autophagy |
| Ultra-Fine Particles (UFP) |
Air pollutants |
SH containing proteins |
Oxidation of intracellular GSH, resulting in the formation of GSSG which alters the redox state of the cell |
Exposure to 50μg/mL UFP for 6 hours led to a decrease in GSH levels (from 17.1±1.8μM to 12.0±2.4μM) and an increase in GSSG (from 0.62±0.26μM to 1.60±0.2μM) in human aortic endothelial cells [106]. |
|
AOP149
: Oxidative Stress Leading to Hypertension
|
Oxidative stress |
| Viruses (HSV-1/2/3, HIV) |
Infectious pathogens |
BACE-1 and nicastrin |
A large proportion of Aβ plaques contain viral or bacterial DNA. Infections activate the amyloidogenic pathway of APP processing while inhibiting Aβ degradation, leading to intracellular Aβ accumulation. Infected cells also showed a disruption of Aβ autophagy, as evidenced by an accumulation of A-containing autophagic compartments that failed to fuse with lysosomes |
1. HSV-1 infection of human and rat neuronal cultures activated amyloidogenic pathway of APP processing while inhibiting Aβ degradation, leading to intracellular Aβ accumulation [107]. |
|
|
Aβ oligomers |
|
|
|
|
2. Cell culture experiments revealed that intracellular concentrations of Aβ were significantly increased as early as 24-h post-infection and that infected cells increased their expression of both BACE-1 and nicastrin, a component of γ-secretase [108]. |
|
|
|
|
3. Antiviral treatments greatly reduced Aβ accumulation in HSV-1 infected cells. Treatment with the antiviral agent Acyclovir reduced the intensity of intracellular Aβ staining to 28% of that in untreated infected cells, while also reducing the levels of BACE-1 and nicastrin [109]. |
|
|
|
|
4. In cell culture aggregates from human AD brains, treatment with the HIV factor Tat led to increased concentrations of soluble A and a reduced activity of the Aβ-degrading enzyme neprilysin [110]. |
| Voriconazole |
Antifungal drug |
CYP46A1 |
Decreased 24S-OHC levels, reduction of HMG-CoA reductase levels, downregulation of cholesterol synthesis and hypocholesterolemia |
Intraperitoneal injections of voriconazole in mice reduced the levels of 24S- hydroxycholesterol in the brain, inhibiting the CYP46A1 [111]. |
|
|
Cholesterol dysmetabolism |
| Zink (Zn) |
Metals |
Binding to Aβ oligomers, Aβ1-40-Zn2+ and Aβ1-42-Zn2+ |
Neurotoxic soluble Aβ oligomers, affecting the calcium ion channel activity in synapsis, through disrupting nerve signal transmission and damage mitochondrial causes to increase free radial lead to cell death. |
Zn2+ binding decreased the solvation energy (increase hydrophobicity) of Aβ oligomer, which enhanced the aggregation propensity, and that a higher concentration of Zn2+ could reduce aggregation kinetics. Aβ peptide can reduce Cu2+ to Cu+, and Fe3+ to Fe2+, facilitating the generation of reactive oxygen species H202 and OH• radical [112]. |
|
|
Aβ oligomers, oxidative stress |
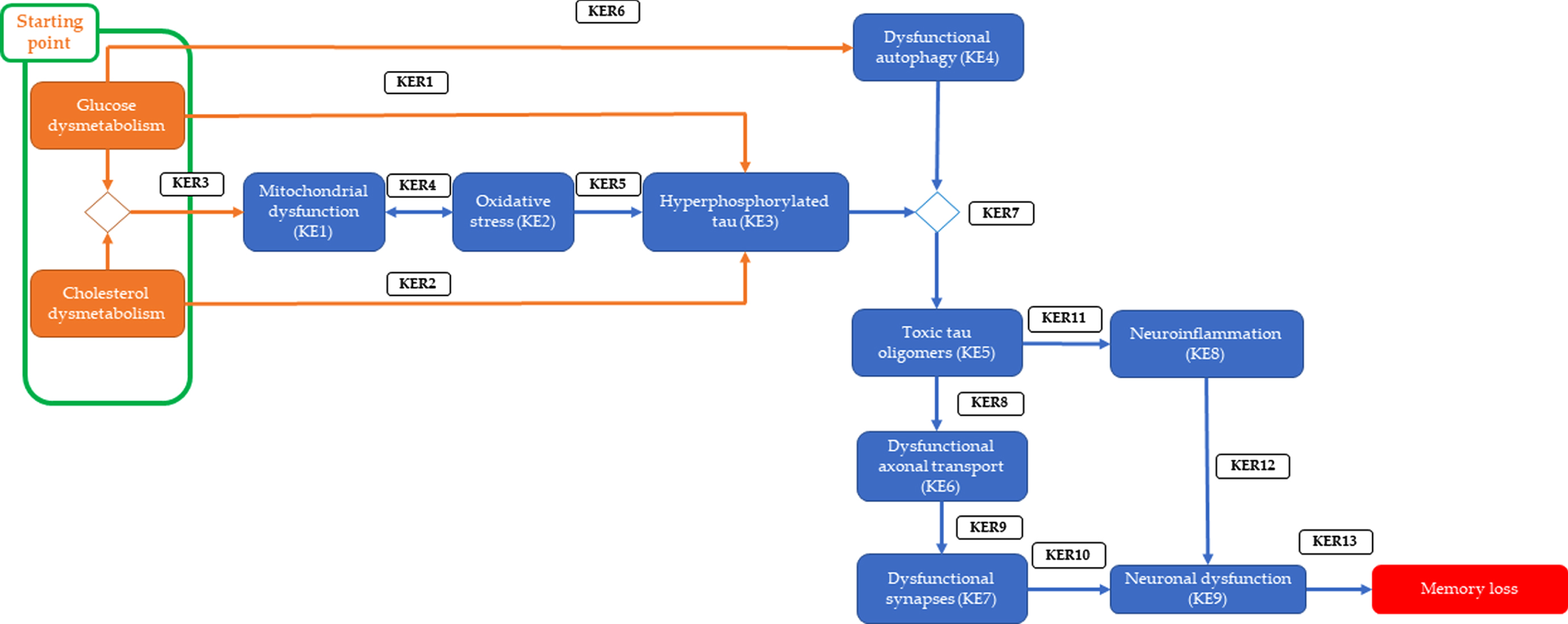
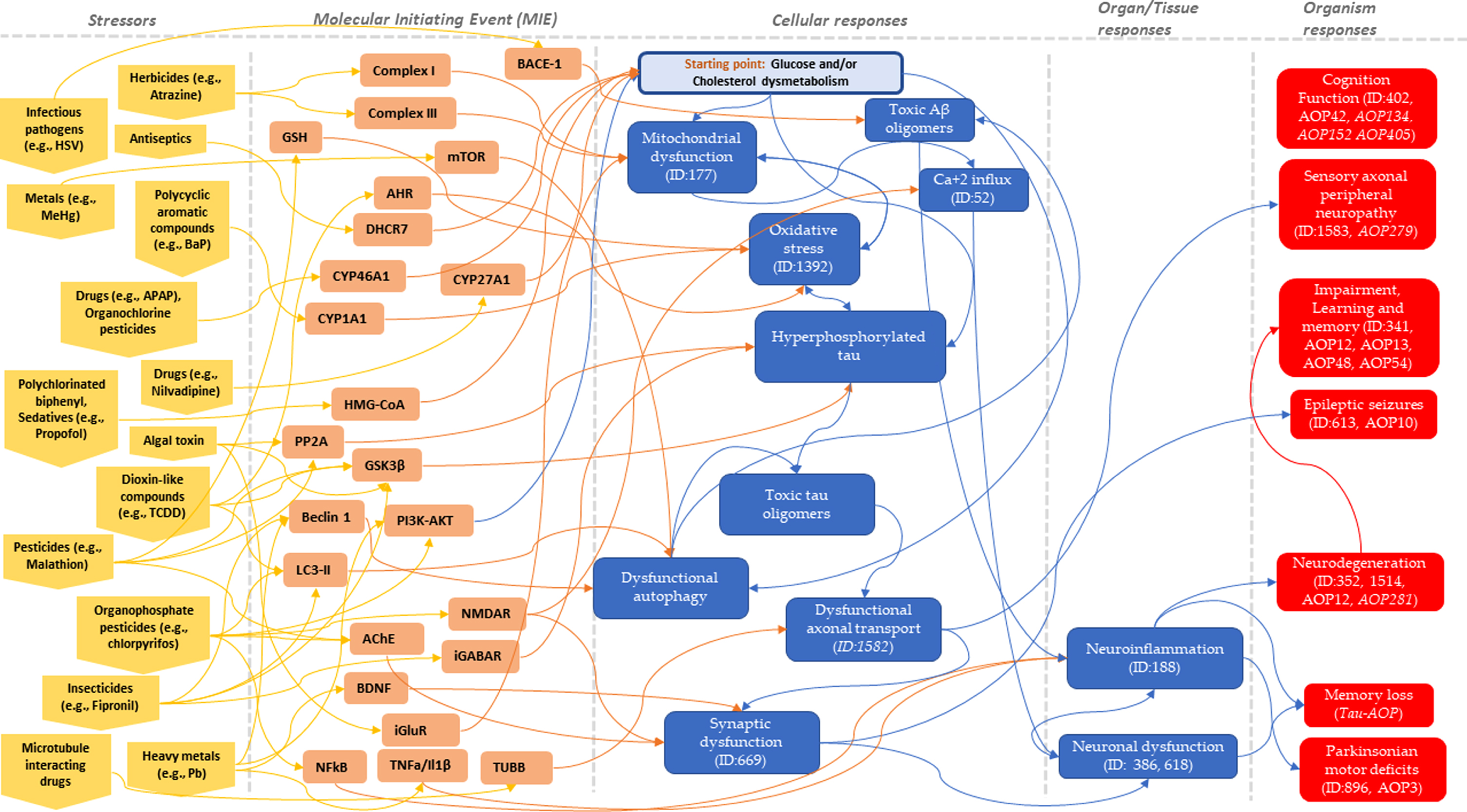
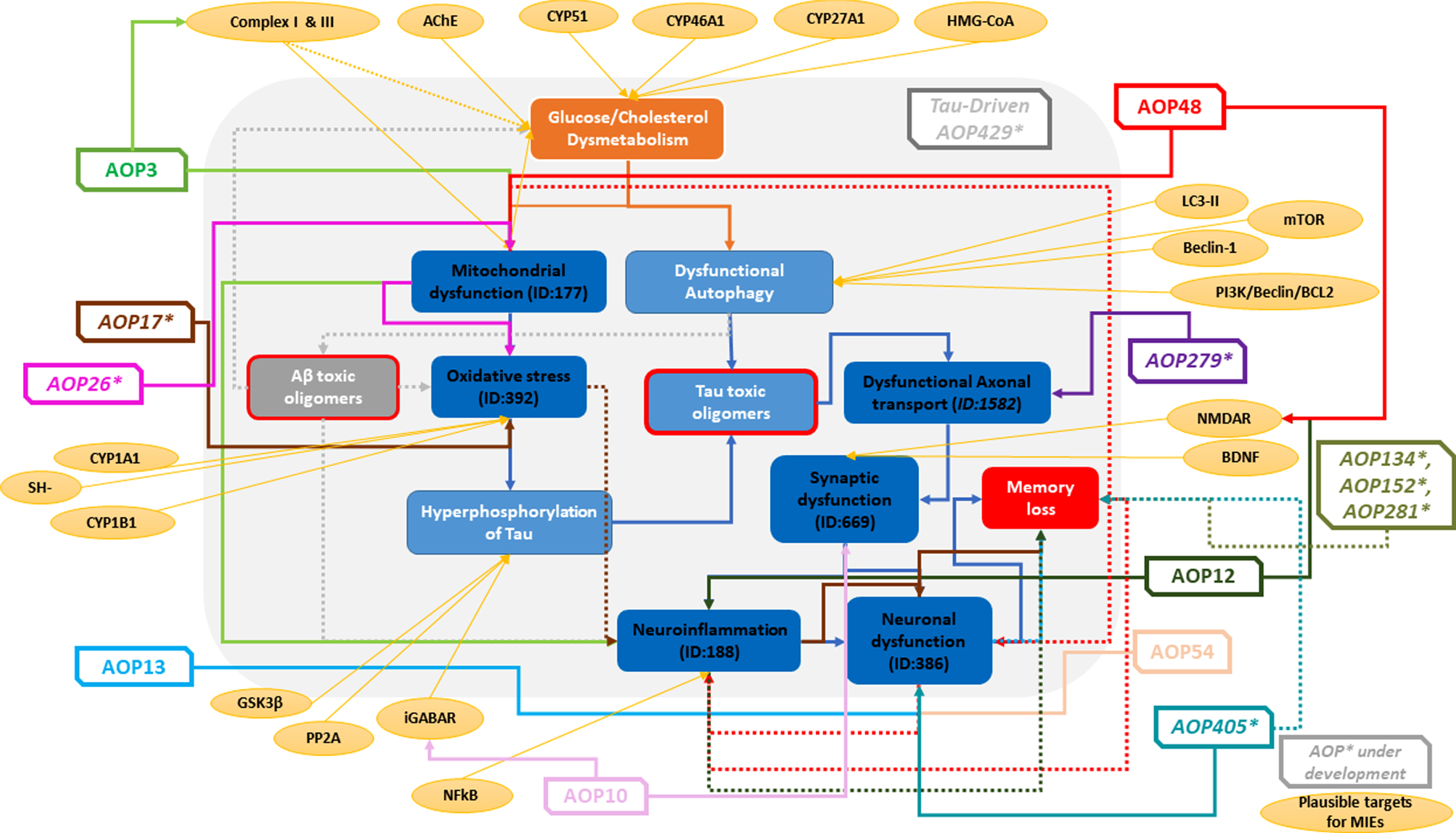
![]() ).
).