Abstract
Background:
Alzheimer’s disease (AD) is characterized by the aggregation of two pathological proteins, amyloid-β (Aβ) and tau, leading to neuronal and cognitive dysfunction. Clearance of either Aβ or tau aggregates by immunotherapy has become a potential therapy, as these aggregates are found in the brain ahead of the symptom onset. Given that Aβ and tau independently and cooperatively play critical roles in AD development, AD treatments might require therapeutic approaches to eliminate both aggregates together.
Objective:
We aimed to discover a chemical drug candidate from natural sources for direct dissociation of both insoluble Aβ and tau aggregates through in vitro assessments.
Methods:
We isolated four borrelidin chemicals from a saltern-derived halophilic actinomycete strain of rare genus Nocardiopsis and simulated their docking interactions with Aβ fibrils. Then, anti-cytotoxic, anti-Aβ, and anti-tau effects of borrelidins were examined by MTT assays with HT22 hippocampal cell line, thioflavin T assays, and gel electrophoresis.
Results:
When HT22 cells were exposed to Aβ aggregates, the treatment of borrelidins alleviates the Aβ-induced toxicity. These anti-cytotoxic effects can be derived from the inhibitory functions of borrelidins against the Aβ aggregation as shown in thioflavin T and gel electrophoretic analyses. Among them, especially borrelidin, which exhibits the highest probability of docking, not only dissociates Aβ aggregates but also directly regulates tau aggregation.
Conclusion:
Borrelidin dissociates insoluble Aβ and tau aggregates together and our findings support the view that it is possible to develop an alternative chemical approach mimicking anti-Aβ or anti-tau immunotherapy for clearance of both aggregates.
INTRODUCTION
Alzheimer’s disease (AD) is the most common type of dementia characterized by cognitive impairments associated with neurotoxicity [1, 2]. Abnormal aggregation of the extracellular amyloid-β (Aβ) and the intracellular tau is considered to be the ma-jor culprit of the AD pathology and a key target for treatments [3, 4]. Because such protein aggrega-tion induces neuronal death and interferes the syn-aptic function in the brain, several current disease-modifying treatments in phase III aim to clear the aggregates or prevent the production of them [5, 6]. However, none of these drugs have been approved for clinical use yet [6]. The possible reason of the failure is that most of drug candidates were designed to target only one among the two pathogenic proteins, Aβ and tau. It is important to remove both proteins simultaneously due to an extensive crosstalk between Aβ and tau during the development of AD pathology [7]. Synergistic interactions between Aβ and tau aggregates result in accelerated neurodegeneration and cognitive impairment compared to their individual contributions on disease progression [8]. It leads to shift attention to a new approach that simultaneously regulates two pathogenic proteins of AD. Currently, two small molecules with a dual targeting mechanism, Nilotinib and Posiphen, are undergoing phase II clinical trials; however, both drug candidates regulate Aβ and tau indirectly. Therefore, we focused on natural product borrelidins, isolated from a halophilic actinomycete living in hypersaline environments, and assessed its therapeutic activities against two pathogenic proteins of AD.
Here, we report the possibility of borrelidin as a small molecule mimicking anti-Aβ and anti-tau anti-body drug candidates to clear the toxic aggregates. Borrelidin (
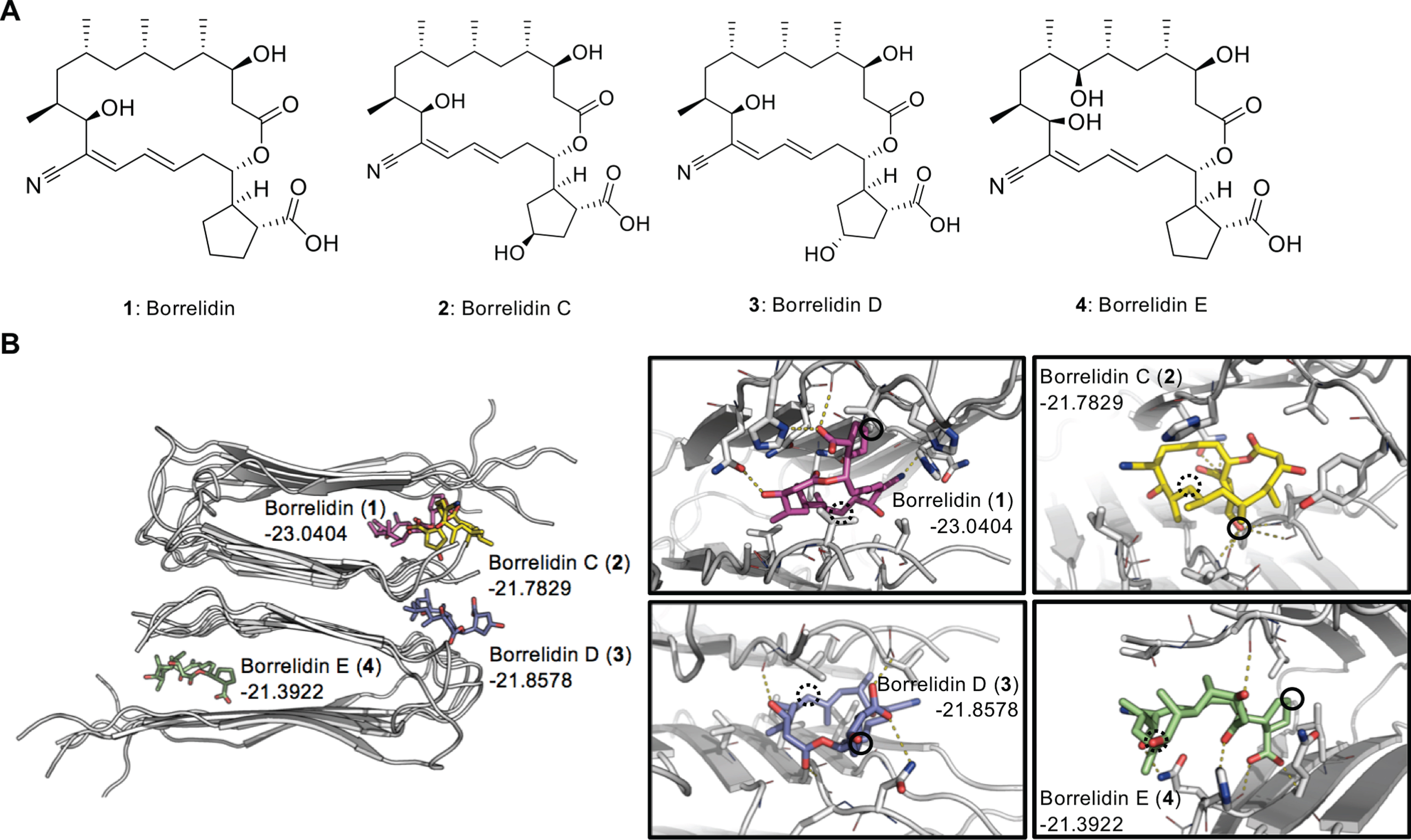
Docking simulation of borrelidins. A) Chemical structures of borrelidin (
MATERIALS AND METHODS
MTT assay
Borrelidins (1–4) were obtained as previously reported [9]. Aβ42 synthesis and MTT assay using HT22 murine hippocampal neuronal cells were performed as previously described [14–17]. Cells (2×104 cells/well) were treated with 10μM Aβ42 with 1–4 (20, 100μM) and incubated for 24 h at 37°C. After the incubation, Aβ-induced cytotoxicity was measured by absorbance as previously reported [16, 17].
Docking simulation
The docking of
Aβ inhibition and disaggregation
10 mM Aβ42 were dissolved in DMSO and diluted with distilled water to make 25μM Aβ solutions. To measure the inhibition effect of
Tau inhibition and disaggregation
Tau K18 fragment (0.5 mg/mL in PBS) was used in these assays. For tau inhibition assay, 1 mM of compound was added to tau containing 0.1 mg/mL of Heparin and 100μM of DTT, aggregation inducers. Mixed samples were incubated for five days at 37°C. For disaggregation assay, tau containing inducers was aggregated for two days at 37°C. After the pre-incubation, 1 mM of compound was added to aggregates and incubated for additional three days at 37°C. To evaluate the inhibition and disaggregation effect of compound, the fluorescence intensity was measured by ThT assay as described above.
PICUP and SDS-PAGE
To visualize inhibitory effect of compounds on Aβ aggregation, all samples were prepared under the identical conditions as used in ThT assay. For Aβ cross-linking, 10 mM Ru(Bpy) and 200 mM APS were dissolved in buffer A (0.1 M sodium phosphate, pH 7.4), and diluted with the same buffer to make 1 mM and 20 mM, respectively. Then, 1μL of Ru(Bpy) and APS were added to 10μL of each incubated sample. The mixtures were irradiated by visible light, and the reaction was quenched by adding 3μL of 5X sample buffer containing 5% reducing agent. Then, samples were boiled for 5 min at 95°C and separated by SDS-PAGE electrophoresis. After gel running, different size of Aβ peptides was visualized by silver staining according to the manufacturer’s instructions (GE Healthcare).
Statistical analysis
Graphs were obtained with GraphPad Prism 7 and statistical analysis were performed with Student’s t-tests and one-way ANOVA, followed by Bonferroni’s post hoc comparisons tests (*p < 0.05, **p < 0.01, ***p < 0.001). The error bars in all data represent the SEM.
RESULTS
Borrelidins bind to Aβ aggregates in docking model
As the abnormal aggregation of Aβ and tau exacerbate AD, the direct interaction between these pathological proteins and drug candidate is needed to dissociate the toxic aggregates. In this regard, we initially performed exhaustive low-resolution docking simulation to find the most plausible docking model of
Among the docking models,
The chemical structures of
On the other hand, the chemical structure of
Borrelidins reduce Aβ-induced cytotoxicity
Since the possibility of binding between Aβ and borrelidins was confirmed through the docking model, we performed additional biochemical assays to investigate the effects of
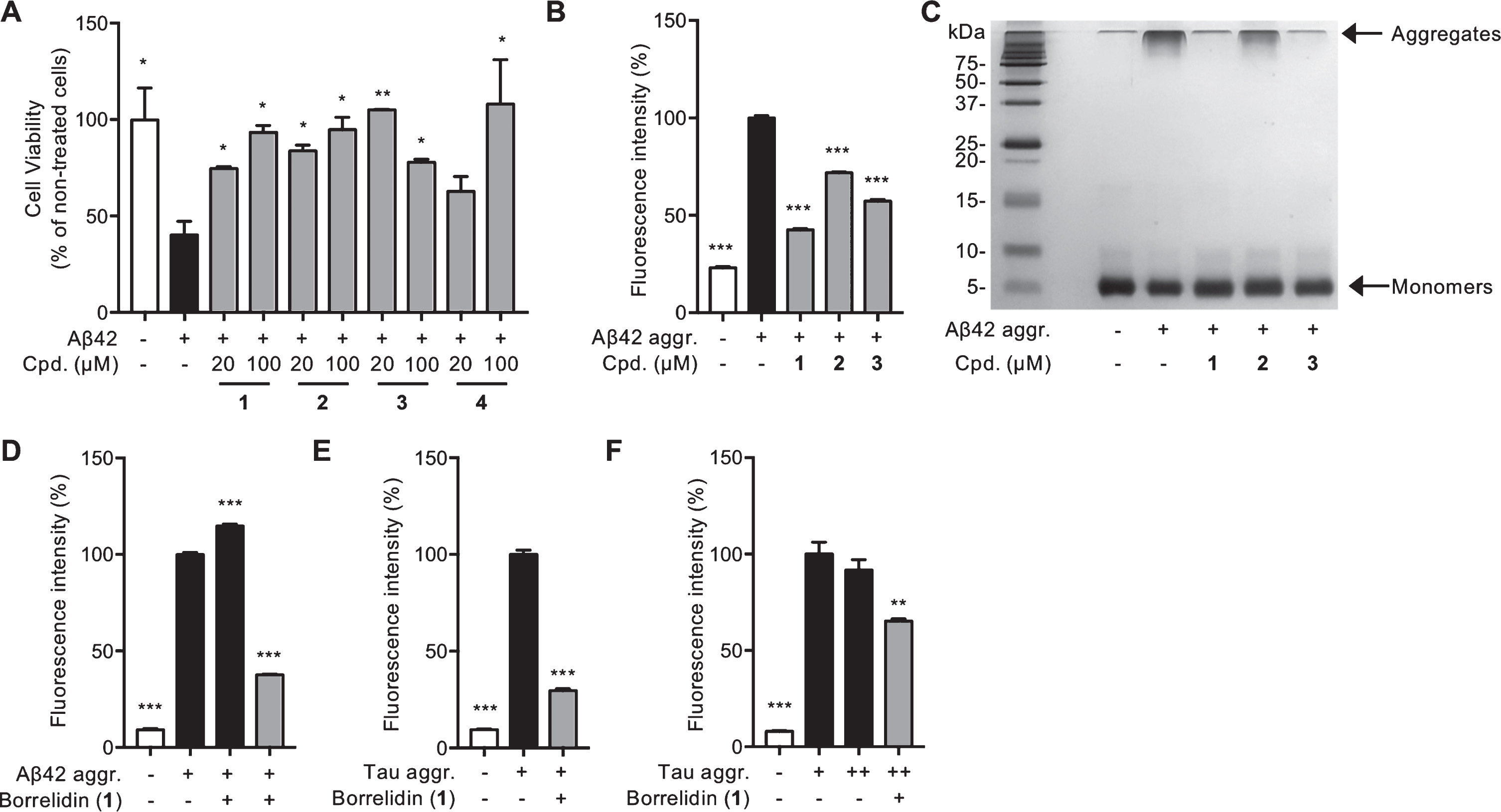
The effects of borrelidins, especially borrelidin (
Borrelidins inhibit Aβ aggregation
Next, we quantified the Aβ fibril formation through ThT fluorescence intensity [23] to examine the protective mechanisms of borrelidins against neuronal death by Aβ aggregates. All compounds significantly inhibited the Aβ aggregation, especially,
Borrelidin dissociates pre-formed Aβ aggregates and also regulates tau pathology
Overall,
As aggregation of tau proteins are also contributed to neuronal dysfunction and eventually neuronal death, we confirmed that the inhibitory effects of
DISCUSSION
Here we report that borrelidin, a natural product derived from Nocardiopsis sp. isolated from a solar saltern, can dissociate Aβ and tau fibrils in vitro, leading to attenuate neuronal cell death induced by toxic aggregates (Fig. 3). Previously, we reported that reduced Aβ aggregates and hyperphosphorylated tau together mediated by a small molecule necrostatin-1 led to inhibition of neuronal cell death, and eventually alleviation of cognitive impairments in AD model mouse [25]. Thus, our in vitro data suggests borrelidin may be potential to improve cognitive function in vivo and such possibility makes borrelidin more beneficial than necrostatin-1 in that it dissociates both Aβ and tau aggregates.
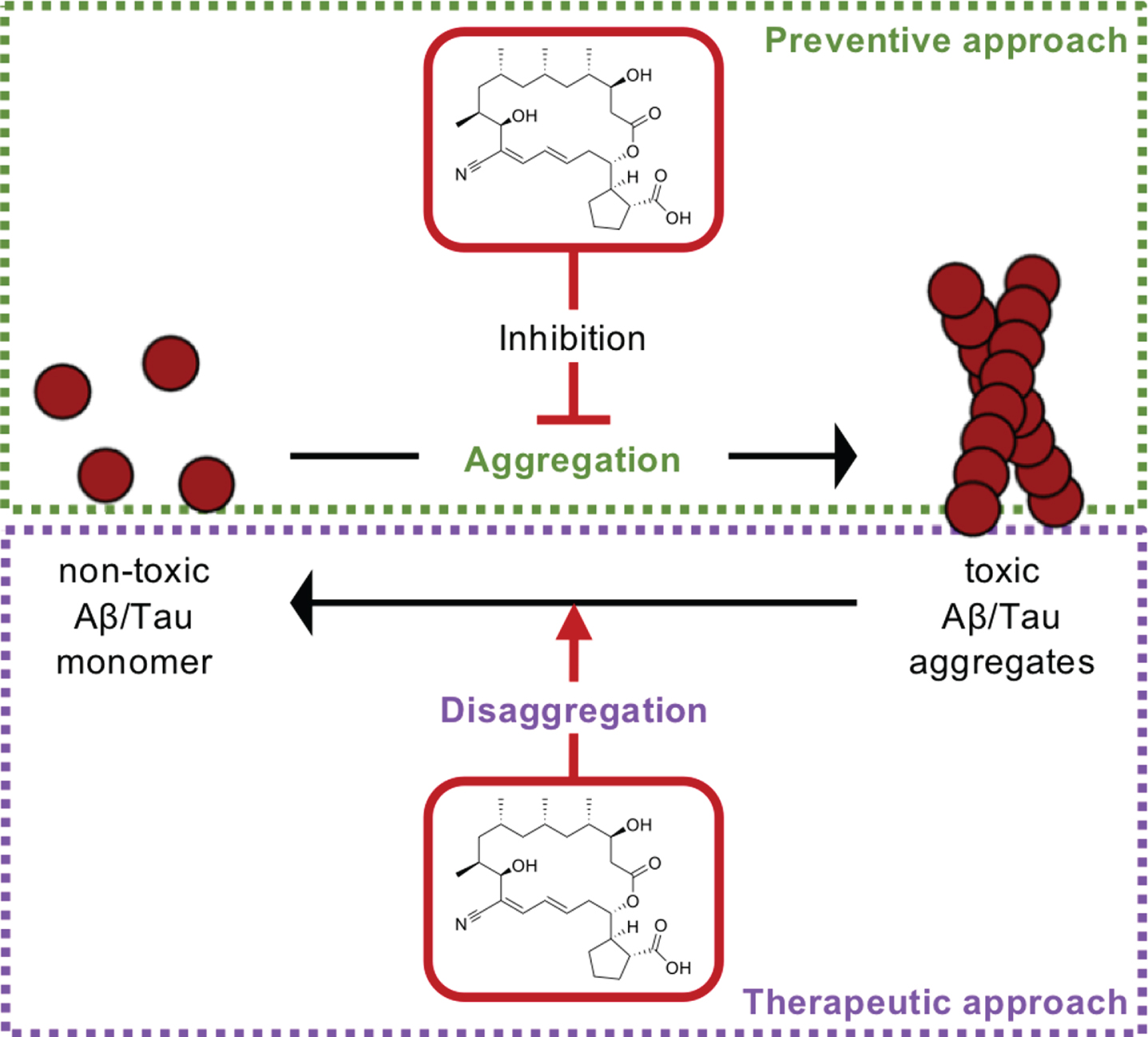
The mechanism of action of borrelidin. The inhibition of the aggregation of both protein aggregates and dissociates toxic aggregates by borrelidin.
Given that AD is a chronic disorder that may require a long-term administration of treatments, safety is one of the major issues for AD drugs. Such property can be solved by searching for drug candidates among natural products such as borrelidin. It is not common to find a single small natural product effective on both pathogenic proteins of AD, Aβ and tau, with additional anti-neurotoxic activity. Although further animal studies are required to warrant the therapeutic potency of borrelidin in vivo, the discovery of the effects of borrelidins, especially
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This work was supported by Yonsei University (2017-22-0072) and National Research Foundation of Korea (NRF-2015R1A6A3A04058568, NRF-2017M3A9E2058046, NRF-2018R1A4A1021703, NRF-2015R1A6A3A04058568, and NRF-2020R1F1A1076240) funded by the Ministry of Science and ICT. This research was also supported by the Collaborative Genome Program (20180430) of the Korea Institute of Marine Science and Technology Promotion (KIMST) funded by the Ministry of Oceans and Fisheries, Korea.
