Abstract
Background:
Dementia is a spectrum of neurological diseases characterized by memory impairment and cognitive decline with the pathogenesis and effective management remaining elusive. Several studies have identified a correlation between anemia and Alzheimer’s disease and related dementias (ADRD); however, anemia subtypes and association with ADRD have yet to be studied conclusively.
Objective:
To study an association between ADRD and anemia of chronic inflammation.
Methods:
We conducted a retrospective case-control study of the patients, diagnosed with ADRD at Brookdale Hospital. Pair-wise comparisons between means of controls and cases in terms of iron studies and laboratory results were performed using a Mann–Whitney U test. Pair-wise comparisons between anemia subgroups (moderate and severe) were performed using a Two Sample proportion Z-Test, where for each couple of normally distributed population.
Results:
There was a total of 4,517 (1,274 ADRD group; 3,243 Control group) patients. There was significant difference in hemoglobin 10.15 versus 11.04 [
Conclusion:
We demonstrated an association between ADRD and anemia of chronic inflammation independent of age, renal function, and HgbA1C levels.
Keywords
INTRODUCTION
Dementia consists of a broad spectrum of neurological diseases characterized by memory impairment and cognitive decline which disproportionately affects those in later stages of life. The pathologies under the umbrella of dementia include, but are not limited to, Lewy body dementia (LBD), frontotemporal degeneration (FTD), vascular dementia, and mixed dementias with the most common being Alzheimer’s disease (AD), accounting for up to two-thirds of the cases of dementia [1]. AD currently affects 5.7 million Americans, and with an ever-growing aging population it is expected to affect as many as 13.8 million by 2050 [1, 2]. Death rates from Alzheimer’s disease and related dementia (ADRD) have more than doubled from 30.5 deaths per 100,000 in 2000 to 66.7 in 2017, with a precise pathogenesis and effective treatment remaining unclear [3].
Current evidence suggests a significant overlap between the phenotypes of dementia and their underlying pathoetiologies. For example, there exists a vascular component present on histology in patients with AD, LBD, and FTD, and amyloid-β neuritic plaques (Aβ), neurofibrillary tangles, and Lewy bodies may be found in any type of dementia. Due to a similar clinical picture and pathophysiology AD, vascular dementia, LBD, FTD, and mixed dementia are included under the umbrella of ADRD [4, 5]. Current pharmacotherapies have been unsuccessful in the reduction of morbidity and mortality associated with ADRD, and at best, only temporarily improve symptoms without successfully targeting the degenerative process [2]. Other treatment modalities, such as dietary/nutritional supplements and lifestyle modifications, have also been investigated, but there still remains insufficient evidence to support efficacy [2].
Investigation into other etiologic factors has revealed a possible correlation between anemia and ADRD [7–9]. Despite the wide-ranging severities of anemia and its multiple subtypes, there is minimal literature to define this association. We proposed that there exists an association between ADRD and anemia of chronic inflammation (ACI), and decided to investigate the association between ADRD and different subtypes of anemia, including ACI, iron deficiency anemia (IDA), and vitamin B12 and folic acid deficiency anemia.
MATERIAL AND METHODS
Patient population
To define the relationship between anemia and dementia, we conducted a retrospective case-control observational study. A review of chart data was performed on the electronic medical record at Brookdale University Hospital Medical Center (Epic© EMR Wisconsin, USA) on all patients that were seen within the healthcare system as an outpatient at one point in their clinical history from January 1, 2016 through January 1, 2019. Inclusion criteria were defined as patients who had a diagnosis that fell under the umbrella of ADRDs [Alzheimer’s disease (G30.0), vascular dementia (F01), dementia with Lewy bodies (G31.83), frontotemporal dementia (G31.0), or unspecified dementia (F03.0)]. Patients with any other causes of symptomatology similar to that of dementia due to trauma, infection, and anoxic/metabolic causes which included patients with a diagnosis of diffuse traumatic brain injury (S06.2), anoxic brain damage, not elsewhere classified (G93.1), Other and unspecified encephalopathy (G93.4), metabolic encephalopathy (G93.41), Parkinson’s disease (G20), Creutzfeldt-Jakob disease, unspecified (A81.00), and Huntington’s disease (G10) were excluded from this study.
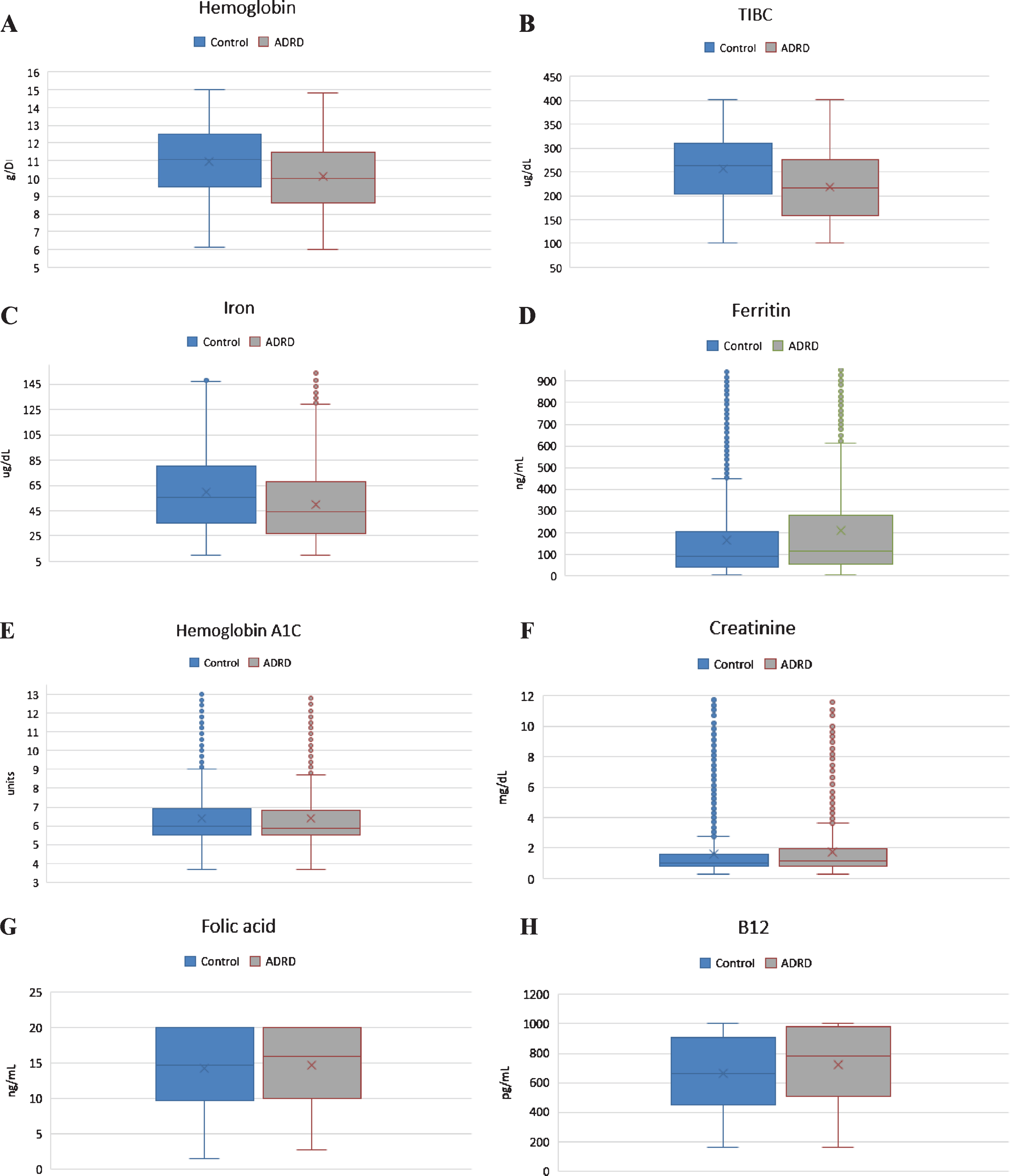
Charts showing graphical representation of pair-wise comparison of laboratory data between clinical groups with respective
In efforts to ensure patients with anemia secondary to acute blood loss anemia were not considered anyone with a diagnosis of upper and/or lower gastrointestinal bleeding, genitourinary and/or obstetrical bleeding, and traumatic bleeding were not considered for the study. Patients with a diagnosis of malignancy were also not considered for the study due to a high prevalence of malignancy and/or chemotherapy related anemia in this patient population [10]. Patients with hematological disorders such as aplastic anemia, myelodysplastic syndrome, thalassemia, or sickle cell anemia were also not included in the study as presence of these pathologies may act as a cofounder. Patients with a diagnosis of HIV were also not considered for the study due to higher prevalence of the anemia and dementia in this population due to a different pathomechanism [11, 12].
In efforts to ensure that we conducted an age appropriate study, only patients with an age >60 were included in the study as dementia in the younger population often carried a diagnosis/history that would exclude the patient from the study. Controls were defined as patients that were seen during the above-mentioned specified time period that did not carry a diagnosis of exclusion.
Definitions
Anemia was defined in accordance with the adult World Health Organization (WHO) definition of anemia, in terms of hemoglobin (Hb) which included mild, moderate, and severe. For males greater than 15 years of age, categories were as follows: mild 11.0–12.9 g/dL; moderate 8.0–10.9 g/dL; severe <8.0 g/dL. For females, non-pregnant, greater than 15 years of age, categories were as follows: mild 11.0–11.9 g/dL, moderate 8.0–10.9 g/dL severe <8.0 g/dL [13]. Additional information was obtained from chart review to help specify category of anemia, i.e., iron deficiency, vitamin B12, folate deficiency, and anemia of ACI. For all patients with available data, serum iron, total iron binding capacity (TIBC), ferritin, vitamin B12, and folic acid was obtained. To mitigate any confounding factors, serum creatinine (Cr) level and hemoglobin A1C (HgbA1C) were obtained to rule out chronic kidney disease (CKD) and diabetes mellitus (DM) as a possible cause of ACI.
Statistical analysis
All the data are reported as sample means with standard deviations. Pair-wise comparisons between means of controls and cases in terms of Hb, iron, TIBC, folic acid, vitamin B12, Cr, and HgbA1C were performed using a Mann–Whitney U test where the null hypothesis presuming means are equal was tested. Statistical significance was defined as
Pair-wise comparisons between anemia subgroups (moderate and severe) were performed using a Two Sample proportion Z-Test, where for each couple of normally distributed population, the null hypothesis that the probability for event within each group is identical was verified. Statistical significance in Two Sample proportion Z-Test was defined as a
RESULTS
A total of 4,517 patients (43% males; 57% females) with a mean age of 75.6±9.9 years were enrolled in the study. Of those, 1,274 patients (42% males; 58% females) with mean age of 80.8±9.4 years with diagnosis of ADRD and 3,243 patients (44% males; 56% females) with mean age of 73.5±9.4 years without a diagnosis of ADRD were included in the study based on exclusion/inclusion criteria.
In our analysis, we examined nine clinical tests relevant to the etiology of the anemia which have been shown in Table 1. Pair-wise comparison of means were analyzed using the Mann–Whitney U test, which revealed that although both groups had low Hb level, there was significant difference in Hb between the ADRD and control group (10.15 g/dL versus 11.06 g/dL,
Laboratory data mean values in the clinical groups
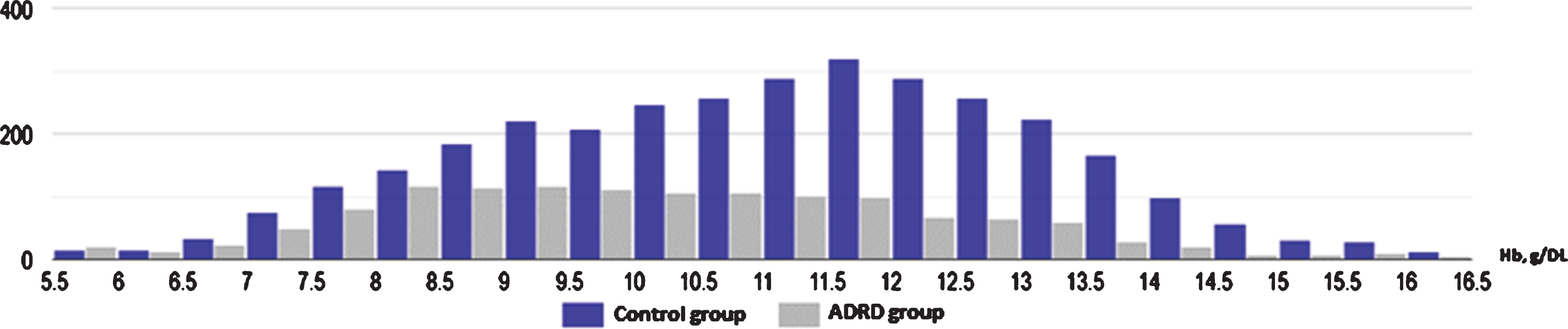
Histogram illustrating significantly higher proportion of moderate and severe anemia in ADRD group (gray) in comparison to control (blue) (95% CI: 15.8–22.1).
Mean differences between groups without and with controlling for age and test of the differences between groups controlling for age
Means of the dementia group and the control group adjusting for age
DISCUSSION
Our data suggests a clear statistically significant association between ADRD and ACI, given low serum iron and TIBC levels, elevated serum ferritin with normal vitamin B12 and folic acid level in ADRD group. We did not find statistical association between HgbA1C and anemia in the ADRD group, which excluded DM as the cause of ACI. Serum creatinine also was not found to be clinically substantial, which precludes CKD as contributor of ACI. These findings suggest that there may be an independent association between ADRD and ACI.
Anemia and ADRD
Preliminary reports of a possible association between anemia and ADRD emerged in late 20th century, and later has been strongly supported by large prospective and retrospective studies [7, 14–17]. In a prospective study from Hong et al., presence of anemia was associated with developing of ADRD (HR = 1.64; 95% CI: 1.30–2.07) [8]. This association was confirmed with another prospective study by Atti et al., demonstrating a two-fold increase in incidence of dementia in patients with anemia within 3 years after adjusting for confounders like inflammation, malnutrition and chronic disease (HR 1.6, 95% CI: 1.1–2.4) [15]. A large retrospective study by Jeong et al. enrolled 37,900 persons and found significant association between anemia and ADRD (HR 1.24; 95% CI: 1.02–1.51), and interestingly, hazard ratio (HR) increased to 5.7 (95% CI: 1.84–17.81) for patients with severe anemia [7]. Despite the strong evidence of the general association, the relationship between anemia subtypes and ADRD has remained largely undefined, which could limit further research for potential treatment options.
Smaller studies examining single subtypes of anemia have also been presented. A case-control study from Chung at el. found an association between IDA and ADRD in females (OR 2.02, CI: 1.43–2.84), but interestingly, no association was found in the male population [18]. The study did not investigate an association between other types of anemia aside of IDA. Weuve et al. investigated the association between ADRD and anemia with an elevated RDW, which is usually associated with both IDA and ACI; however, no statistically significant difference was found (OR, 0.99; 95% CI: 0.86–1.18) [19]. In a retrospective study by Faux et al. an Australian Imaging, Biomarkers and Lifestyle (AIBL) cohort was used to investigate an association between anemia and dementia in an Alzheimer’s population. The study included 211 patients with AD, 133 patients with mild cognitive impairment and 768 healthy adults [9, 20]. This study again showed a strong association between anemia and ADRD (OR 2.43, CI: 1.31–4.54), but more importantly, it was the first study which investigated a possible association between ADRD and certain types of anemia. No statistically significant differences were found between serum vitamin B12, folic acid, serum Cr, blood urea nitrogen, and haptoglobin, which did not support an association ADRD with some subtypes of anemia such as vitamin B12 and folic acid deficiency anemia, hemolytic anemia, and anemia of CKD. Serum iron level was normal in both groups, and ferritin was elevated in ADRD group (
Similarly, we found no association between ADRD and IDA (in both the male and female population), anemia of CKD, and vitamin B12 and folic acid deficiency anemia. In contrast to Faux at el., we found a statistically significant difference in serum iron, ferritin and TIBC between the control and study groups. Although normal in the control group, we found significant evidence of iron dyshomeostasis and ACI in ADRD group (low iron and TIBC levels, and high ferritin level) (Table 1).
Though the association between anemia and dementia has been well described in literature, the pathophysiology of this association remains not well understood. It has been hypothesized that anemia may lead to memory loss due to chronic brain hypoxia [7]. Alternatively, it has also been hypothesized that it could be a sign of underlying ADRD and a reflection of a pathologic process that is yet understood. We found a strong association with ADRD and ACI, which implies that a combination of anemia and iron dyshomeostasis play a significant role in pathogenesis of dementia.
It is also important to mention that there have been no long-term studies to assess the effect of untreated anemia on the brain and memory function. We hypothesize that the brain, the most sensitive organ to hypoperfusion and hypoxia, may require a higher level of hemoglobin to maintain cortical functions as we age. We found a significant difference of almost 1 g/dL of Hb between the ADRD and control group (10.15 and 11.06,
If an acute reduction in Hb can cause such a change in cognition, it is within reason to postulate that long standing anemia may cause chronic damage to the brain, contributing to memory loss and development of dementia. Further prospective studies are needed to determine, if blood transfusion targeting a Hb level of more than 11 g/dL and/or correction of moderate-to-severe anemia reduces the incidence or progression of ADRD.
Anemia of chronic inflammation
Anemia of chronic inflammation (ACI), formerly termed anemia of chronic disease, is defined as an impaired production of erythrocytes due to inflammatory conditions such as chronic infection, cancer, autoimmune diseases or milder but persistent inflammatory states such as DM, obesity, aging, and CKD [26, 27]. A diagnosis of ACI requires normal iron stores with low circulation iron less than 60
Prevalence of anemia increases rapidly with age, and in general 11.0% of men and 10.2% of women 65 years and older, and 20% of those greater than 85 years of age are found to be anemic [29]. As was defined previously, ACI is frequently multifactorial and commonly associated with chronic diseases, including DM and CKD, and aging itself. Joosten et al. observed 191 hospitalized elderly patients with anemia, and showed that 70% of the patients had ACI. 16% of the patients with ACI had concomitant chronic renal failure, 71% of them had an acute infection, 12% had cancer, and 16% had a chronic infection, such as pressure ulcer, or a chronic autoimmune inflammatory disease [30]. In addition, a multi-center study with 4,892 ICU patients in United States revealed that ACI is particularly common in critically ill patients [31].
We investigated the association between ACI in ADRD and CKD and did not find a clinical difference in serum creatinine (1.8 mg/dL versus 1.7 mg/dL,
Blood transfusion, intravenous iron, and erythropoietin are established standards of care in treatment of ACI [26]. Administration of erythropoietin in anemia has shown to be effective by elevating concentrations of reticulocytes and serum transferrin receptors [33]. There is also novel experimental therapy targeting IL-6 and hepcidin-ferroportin axis [26]. Tocilizumab, an anti-IL6 receptor antibody, has demonstrated efficacy in reduction of hepcidin level and improving ACI in patients with rheumatoid arthritis. In addition, therapies targeting the hepcidine-ferroportine axis include LDN-193189 and NOX-H94, these agents have shown to be effective in improvement of ACI in animal models.
Iron dyshomeostasis and ADRD
The phenomenon of iron dyshomeostasis in patients with ADRD has been reported in multiple studies, however the studies are of relatively smaller sample size [34–43]. The role iron elements play in AD could be theoretically established by trying to quantify tissue concentrations as compared to controls. In a postmortem case-control study of 16 patients by Grundke-Iqbal et al. ferritin was observed in Aβ plaques [35]. In addition, two
Evidence of iron dyshomeostasis in peripheral blood associated with ADRD has been reported previously. In a systematic review by Lopes da Silva et al. in 2014 with a total of 153 ADRD patients and 544 controls across five studies, only two studies showed a statistically significant decrease in plasma iron levels though the parameters of TIBC and ferritin levels were not investigated [45]. Faux et al. used the AIBL cohort to investigate the association between anemia and dementia, and as was discussed earlier, was the first study to investigate the association of all major types of anemia with ADRD [9, 20]. No statistically significant association was observed between specific types of anemia and ADRD, but the authors, based on elevated ESR and ferritin in ADRD group, proposed an association with ACI. In 2017, a prospective study by Goozee et al. showed statistically significant difference in serum ferritin in patients with preclinical dementia with high neocortical Aβ load, but serum iron, TIBC, or Hb were not investigated [46]. In our study, we were able to show a statistically significant association between ACI and ADRD, which we theorize is due to the higher power of our study compared to previous reports.
While dysregulation of iron homeostasis in patients with ADRD is evident, the exact origin of excessive iron and ferritin stores as well as its precise pathogenesis of its accumulation remain unclear. Iron induced oxidative stress has been proposed and is well supported in literature. Excess iron could promote the conversion of hydrogen peroxide (H2O2) to hydroxyl radical (–OH) with high toxic properties and trigger Aβ production, and thus, Aβ plaques [34, 47].
Another intriguing hypothesis is that excessive iron interferes with protein transcription and translation in neurons and glial cells [38]. The cerebral cortex has the largest iron storage in the body and iron-regulation gene expression only second to the liver as iron serves an essential role in myelin formation, neurotransmitter production and metabolic activity of the neurons [43]. There exists several proteins involved in brain iron metabolism, including transferrin, transferrin receptor 1, ferritin, iron-regulatory proteins (IRPs), ferroportin 1 (FPN1), lactoferrin, divalent metal ion transporter, and brain-specific ceruloplasmin [38, 49]. Iron and IRPs controls transcription of these proteins in the brain via iron regulatory element (IRE) located on mRNAs [50]. Interestingly, IRE was also found on amyloid-β protein precursor (AβPP) mRNA, the direct progenitor of Aβ, and it has been postulated than AβPP itself can further regulate iron metabolism, inducing influx of the iron into the cell via FPN1 [50, 51]. Further excess of iron accumulation leads to increase of aggregation of AβPP,
Blocking IRE on AβPP,
Inflammation, infection, and ADRD
Neuroinflammation plays a major role in the pathogenesis of ADRD with involvement of microglia, cytokines and complement complex [54]. One hypothesis is that Aβ peptides along with CD40 co-stimulation trigger the inflammatory response [55]. Others have suggested that acute and/or chronic inflammation can be transmitted to the brain via cytokines acting on endothelial cells of the blood-brain barrier. Subsequently a pro-inflammatory state triggers Aβ-dependent or Aβ-independent inflammation inside the CNS, eventually leading to cognitive impairment [56, 57]. Our study revealed changes in the peripheral blood affecting complete blood count and iron panel, suggesting chronic inflammation in the patients affected by ADRD.
Association between inflammation and ADRD is also well-supported in prior studies. In 2002, Schmidt et al. published a large prospective study which showed an association between elevated high-sensitive CRP) and ADRD, and proposed that inflammation precedes manifestation of dementia [58]. In 2004, Engelhart et al. discovered that other inflammatory proteins such as
Summary of the studies of association of the inflammation and ADRD
hsCRP, high-sensitivity C-reactive protein; ADRD, Alzheimer’s disease and related dementia; TNF, tumor necrosis factor; CI, confidence intervals; RR, relative risk.
Aβ deposition has been considered the major force behind ADRD pathophysiology, and, specifically, AD [64]. However, there is a growing body of literature that suggests that role of Aβ in neuronal damage might be misunderstood as new evidence is suggestive of a protective role [57, 65]. Recent
Evidence of inflammation and antimicrobial properties Aβ have led to further research in this area. Emerging data strongly point to the evidence of infection etiologies of the ADRD, including viral, bacterial, and fungal origin [65, 70]. Recent
Summary of the studies of association of the infection and ADRD
AD, Alzheimer’s disease; HSV, herpes simplex virus; OR, odds ratio; HR, hazard ratio; CI, confidence intervals.
Limitations of the study
There were certain limitations of this study. The retrospective nature of the study allowed us to show an association between ACI and ADRD but not causation. Elevations in ESR and CRP are laboratory markers of the ACI in addition to Hb and iron profile, but these markers were not available for investigation. Autoimmune disease, such as rheumatoid arthritis and systemic lupus erythematosus can lead to ACI; however, association with ADRD, ACI, and rheumatologic disorders were not investigated in this study due to relatively low prevalence in the population (prevalence of rheumatoid arthritis in the US is 0.55% [75]; prevalence of systemic lupus erythematosus is 0.006% [76]).
Conclusion
There exists a fair amount of literature examining the role of anemia in ADRD though not one study encompasses a global view of anemias in their analysis. We chose to investigate the subtypes of anemia and its role in dementia. We found a statistically and clinically significant decrease in the Hb level between ADRD group and control groups (
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
