Abstract
To date, dysregulation of the insulin signaling pathway in the brain has not been demonstrated unequivocally in Alzheimer’s disease (AD). The purpose of the study was to examine the possible dysregulation of insulin signaling pathway in an AD rat model. Furthermore, the present study investigated the effect of Donepezil and Curcumin on insulin signaling, insulin, and glucose levels in AD rat brain. The rats were induced to develop AD by intraperitoneal administration of Scopolamine. We found that glucose levels in plasma and brain were decreased in AD rats, whereas the insulin levels was increased in plasma but decreased in brain in AD rats. In addition, insulin signaling proteins IR-β, IGF-1, IRS-1, IRS-2 p-Akt (Ser473), and Akt were markedly reduced in the AD rats. Furthermore, GLUT3 and GLUT4 levels in the brain were markedly reduced in AD rats. All these data were compared to Saline-treated control rats. Curcumin significantly increased glucose levels in plasma and in brain. However, insulin levels was decreased in plasma and was increased in AD rats’ brain. Moreover, GLUT3 and GLUT4 levels were significantly increased in Curcumin-treated AD rats. All these data were compared to Scopolamine– induced AD rats. Thus amelioration of impaired insulin signaling and improved glucose regulation in AD rats by Curcumin may be beneficial in the management of AD.
INTRODUCTION
Alzheimer’s disease (AD) is the most devastating neurodegenerative disease among older people (≥65 years) that ultimately causes death of the affected individuals. AD is a major global health problem that results in significant burden on the patients due to the high cost of managing AD [1]. Neuropathologically, AD is characterized by brain atropy, loss of neurons, synaptic dysfunction, extracellular accumulation of amyloid-β (Aβ), and intracellular neurofibrillary tangles (NFT) composed of hyperphosphorylated tau [2, 3]. Despite indistinguishable clinical dementia symptoms, less than 1% familial early-onset AD is caused by missense mutations in three genes of presenilin-1, presenilin-2, and amyloid precursor protein [4]. However, most AD cases are sporadic and are believed to result from multiple etiologic factors including genetic susceptibility (such as ApoE4 allele), aging, type 2 diabetes, and metabolic factors [5, 6].
Defective glucose utilization [7–9] or hypometabolism of glucose in the brain may precede the onset of cognitive deficits [10–12]. Furthermore, hyperinsulinemia is associated with higher risk of AD and decline in memory [7, 11]. Peripheral glucose metabolism is primarily regulated by insulin. Therefore, hyperinsulinemia associated with insulin resistance can affect insulin signaling that controls brain glucose metabolism and facilitates the neuronal development and functions in learning and memory [13]. It is hypothesized that possible dysfunction of insulin signaling is linked with AD pathogenesis [14–17]. Although, how the impaired insulin signaling contributes to the AD pathogenesis is not fully understood.
Numerous research groups [18–21] have provided robust evidence regarding the supporting evidences the use of Scopolamine in AD animal model. Scopolamine, a muscarinic cholinergic receptor antagonist, has been commonly used for the study of cognitive deficiency in experimental animals. Intraperitoneal injection (i.p.) of Scopolamine into animals causes blockage of the cholinergic neurotransmission that leads to cholinergic dysfunction and impaired cognition in animals [18–21]. Bihaqi et al. found that the expression of tau and the amyloid-β protein precursor (AβPP) were increased in rat brain [20]. Scopolamine-induced dementia model is the most appropriate dementia model to reveal AD-related cognitive impairment profile [21]. Donepezil, a well-known pharmacological drug used for the treatment of AD, acts as an acetylcholinesterase inhibitor, reduces the hydrolysis of acetylcholine, and improves AD pathogenesis. Throughout this study, Donepezil was used as a reference drug. Curcumin, a natural compound derived from plant, is also known for its protective effect on AD pathogenesis. It is known that Curcumin has various properties such as anti-inflammatory, anti-oxidant, and scavenger of free radicals [20]. Importantly, curcumin has also been shown to have beneficial effects in various neurodegenerative disease models [23–26]. However, to date, there is very little information about any effects of Donepezil and Curcumin on insulin signaling, glucose, and insulin levels in AD rats.
Thus, in this study, we investigated the effects of Donepezil and Curcumin on the mediators of insulin signaling such as IR-β, IGF-I, IRS-1/2, PI3K, p-Akt (Ser473), and Akt and also on the levels of key glucose transporters such as GLUT3 and GLUT4 in a rat model of sporadic AD, which was generated by i.p injection of Scopolamine. In addition, the effects of Donepezil and Curcumin on glucose and insulin levels in both plasma and in the brain of AD rats were evaluated. We found that glucose levels in plasma and brain were decreased in AD rats. Whereas, the insulin levels was increased in plasma but decreased in brain in AD rats. In addition, insulin signaling proteins IR-β, IGF-1, IRS-1, IRS-2 p-Akt (Ser473), and Akt were markedly reduced in the AD rats. Furthermore, GLUT3 and GLUT4 levels in the brain were markedly reduced in AD rats. All these data were compared to Saline-treated control rats. Curcumin significantly increased glucose levels in plasma and in brain. However, insulin levels was decreased in plasma and was increased in AD rat brain. Moreover, GLUT3 and GLUT4 levels were significantly increased in Curcumin-treated AD rats. All these data were compared to Scopolamine– induced AD rats.
MATERIAL AND METHODS
Materials
Scopolamine, Donepezil, and Curcumin were purchased from Sigma Aldrich (St. Louis, MO, USA). Protease inhibitor tablets were purchased from Roche Applied Science (Mannheim, Germany). Amplex Red Glucose/glucose Oxidase Assay Kit were purchased from Invitrogen (Carlsbad, CA, USA) and ELISA kit for Insulin were purchased from Cloud-Clone Corp (Houston, TX, USA). Antibodies against IR-β, IGF-I, IRS-1, IRS-2, Akt (pan), p-Akt (Ser 473), and GLUT4 were purchased from Cell Signaling Technology (Danvers, MA, USA). p-PI3K (p85) was purchased from Santa Cruz Biotechnology (Dallas, TX, USA). Antibodies against GLUT3 and β-Actin were purchased from Upstate Biotechnology (Lake Placid, NY, USA) and Sigma Aldrich (St. Louis, MO, USA) respectively. Immunohistochemistry kit (R.T.U. Vectastain Universal Elite ABC Kit and DAB kit) was purchased from Vector Laboratories (Burlingame, CA, USA). PierceR BCA Protein Assay Kit and ECL advanced reagent kit were purchased from Thermo Fisher Scientific (Waltham, MA, USA) and Bio-Rad (Hercules, CA, USA) respectively. Other analytical grade reagents were purchased from Merck (Kenilworth, NJ, USA).
Animals
24 male Sprague Dawley rats aged 60–70 days and weighted 300±20 g provided by PAPRSB Institute of Health Sciences, UBD were utilized. The rats were provided standardized rat food (Specialty Feeds, Western Australia) and drinking water, and were housed in temperature-controlled cages (12-h light/dark cycle, room temperature 26°C±2°C). The experiments were performed according to standard guideline approved by PAPRSB, Institutes of Health Sciences Research Ethic Committee and Universiti Brunei Darussalam Research Ethic Committee.
Study design
The rats were randomly divided into four groups as follows; (a) Control (saline-treated), (b) Scopolamine-induced, (c) Scopolamine+Donepezil-treated, and (d) Scopolamine+Curcumin-treated. Control group was administered with 0.9% saline (w/v) for 28 consecutive days. Scopolamine (2.5 mg/kg), as a disease inducer, was administered to all the groups through i.p route. The treatment groups were given Donepezil (5 mg/kg) and Curcumin (8 0 mg/kg) by oral gavage for 28 consecutive days (Table 1). Although oral gavage was as invasive procedure, however we did not observe any signs of suffering in animal in the study.
Experimental design and drug doses in different groups of rats
Preparation of blood plasma
After 28 days of either Scopolamine or Saline treatment, rats were anesthetized using carbon dioxide in a closed chamber and then sacrificed by decapitation using guillotine. 5 ml blood was collected in anticoagulant heparin tube and it was fractionated by centrifugation at 3000×g for 15 min. Supernatants (plasma) were collected and stored frozen at – 80°C until further use.
Brain tissue preparation and processing
After 28 days of either Scopolamine or Saline treatment, rats were anesthetized using carbon dioxide in a closed chamber and then sacrificed by decapitation using guillotine, dissected and the brains were quickly collected and washed three times in chilled 1x phosphate buffered saline (PBS) (pH 7.4) (Bio-Rad). Each group consists of six brains in which two brains were used for immunohistochemistry (IHC) and the remaining four brains were used for immunoblotting.
Immunohistochemistry
Brains from each group were washed in 1X PBS, and were subsequently used in tissue processing. Each brain from rat was cut into smaller pieces using a tissue slicer and then processed using an automated tissue processor (Leica ASP 300 S Tissue Processor, Leica Microsystem, USA), embedded in paraffin wax and then sectioned using a microtome (Medite, Medizintechnik, Germany) into 4-μm thickness. Brain sections from different groups of rats (
Brain homogenate preparation
The remaining four brains from each group were used for immunoblotting. Each brain with known weight was snapped frozen in liquid nitrogen, homogenized in cell lysis buffer (1X) (Cell Signalling Technology) containing protease inhibitor tablet (Roche) at 3 pulses for 10 s each, with a 5 s interval on ice. Tissue lysates were diluted 10-fold in 1X cell lysis buffer and were sonicated to ensure complete lysis. The crude brain homogenates were stored at – 80°C and the protein concentrations were determined by using PierceR BCA Protein Assay Kit (Thermo Fisher Scientific).
Measurements of glucose and insulin levels
Glucose levels in plasma and in brain were measured using Amplex Red Glucose/glucose Oxidase Assay Kit (Invitrogen) and the insulin concentrations were measured in plasma and in brain using ELISA (Cloud-Clone Corp) following the instructions provided by the manufacturer. Absorbance was measured at 560 nm and 450 nm (BioTek Instruments, USA) respectively.
Immunoblotting
50 μg proteins were separated on 10–12.5% SDS-polyacrylamide gel and transferred to nitrocellulose membranes (Amersham Bioscience, Piscataway, NJ, USA), blocked for 1 h at room temperature with 4-5% BSA in T-TBS, incubated at 4°C overnight with respective primary antibodies, followed by incubation in horseradish peroxidase (HRP) conjugated secondary antibodies. Finally, the reactive protein bands were visualized using chemiluminescence detection reagent (Clarity™ Western ECL Substrate, Bio-Rad) and signals were captured and visualized using VersaDoc Imaging system (Bio-Rad). Pre-stained Precision PlusTM protein standards (Bio-Rad) was used to estimate the apparent molecular weight of the protein bands. β-actin was used as a control to ensure equal amount of protein loading.
Statistical analysis
Data were expressed as mean±S.E. and analyzed using SPSS 20.0 statistical software (SPSS Inc., Chicago, IL, USA). The
RESULTS
Lower levels of plasma glucose and brain glucose in AD rats and curcumin treatment significantly increased glucose levels
Glucose contents in the plasma were measured in all groups of rats and showed in Fig. 1. The glucose concentration in the plasma (
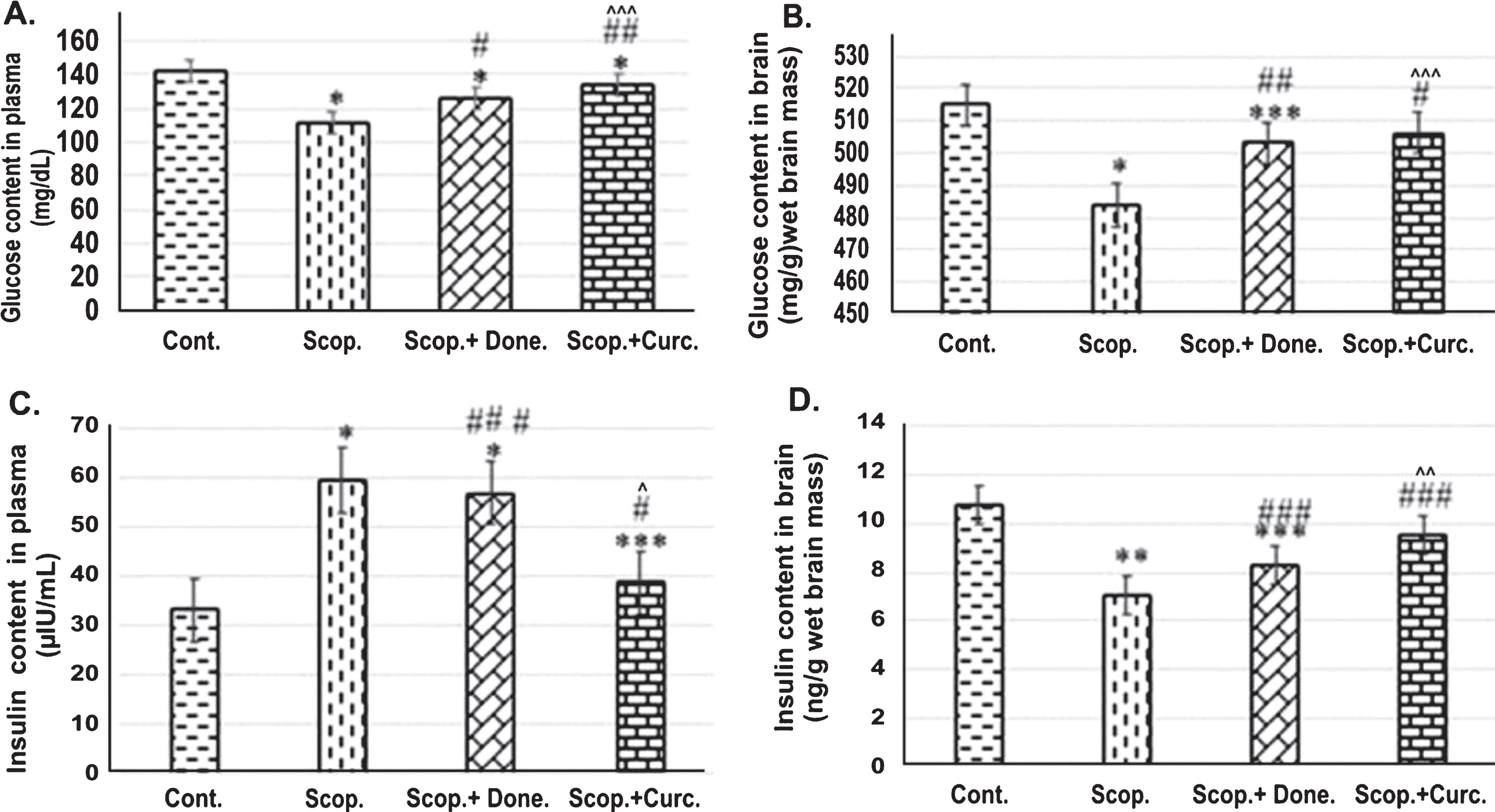
ELISA analysis of (I) glucose and (II) insulin content in (A) the plasma (
Furthermore, glucose concentration in the brain was measured in all groups of rats. Glucose concentration in the brain lysate was 515±4.22 mg/g wet brain mass in the Saline-treated control group and 483.76±9.69 mg/g wet brain mass in Scopolamine-induced AD group respectively. In Donepezil-treated and Curcumin-treated AD rats, glucose concentration was found to be 503.02±5.10 mg/g wet brain mass and 506.32±2.26 mg/g wet brain weight (Fig. 1B) respectively. Taken together, it was found that glucose level was decreased 1.52-fold (
Higher levels of plasma insulin and lower levels of brain insulin in AD rat, curcumin treatment significantly reversed the insulin levels
Plasma insulin levels were measured in all groups of rats. Insulin concentration was found to be 33.11±1.16 μIU/mL in Saline-treated control group in comparison to 59.38±3.56 μIU/mL in Scopolamine-induced AD rats. In Donepezil-treated AD rats, the insulin concentration was found to be 56.94±0.44 μIU/mL. Interestingly, Curcumin treatment in AD rats markedly lowered insulin levels to 38.74±0.44 μIU/mL compared to Scopolamine-treated AD rats. Thus, plasma insulin level was 1.79-fold higher (
Furthermore, insulin levels in the brain were measured in all groups of rats. The insulin level in the brain was 10.78±1.64 ng/g wet brain mass and 7.08±1.25 ng/g wet brain mass in Saline-treated and Scopolamine-induced AD rat group respectively. In Donepezil-treated AD rats, the insulin level was at 8.3±1.86 ng/g wet brain mass. In addition, Curcumin- treatment in AD rats increased insulin levels to 9.53±1.25 ng/g (Fig. 1D) compared to Scopolamine-induced AD rats. Therefore, it was found that insulin level was 1.52-fold (
Reduced number of immunostained cells positive for insulin signaling proteins and glucose transporter in AD rat brain and curcumin reversed the effects
Immunohistochemistry was used to detect the presence of insulin signaling proteins and glucose transporter in the brain cortex of the rats. There was a reduced number of the immunostained cells for (i) IR-β, (ii) Akt, (iii) Phospho-Akt (Ser473), and (iv) GLUT4 in AD rats compared to Saline-treated control rats. Treatment with either Donepezil or Curcumin increased numbers of positive insulin signaling proteins such as IR-β, Akt, Phospho-Akt (Ser473), and GLUT4 immunopositive cells in AD rats (Fig. 2).
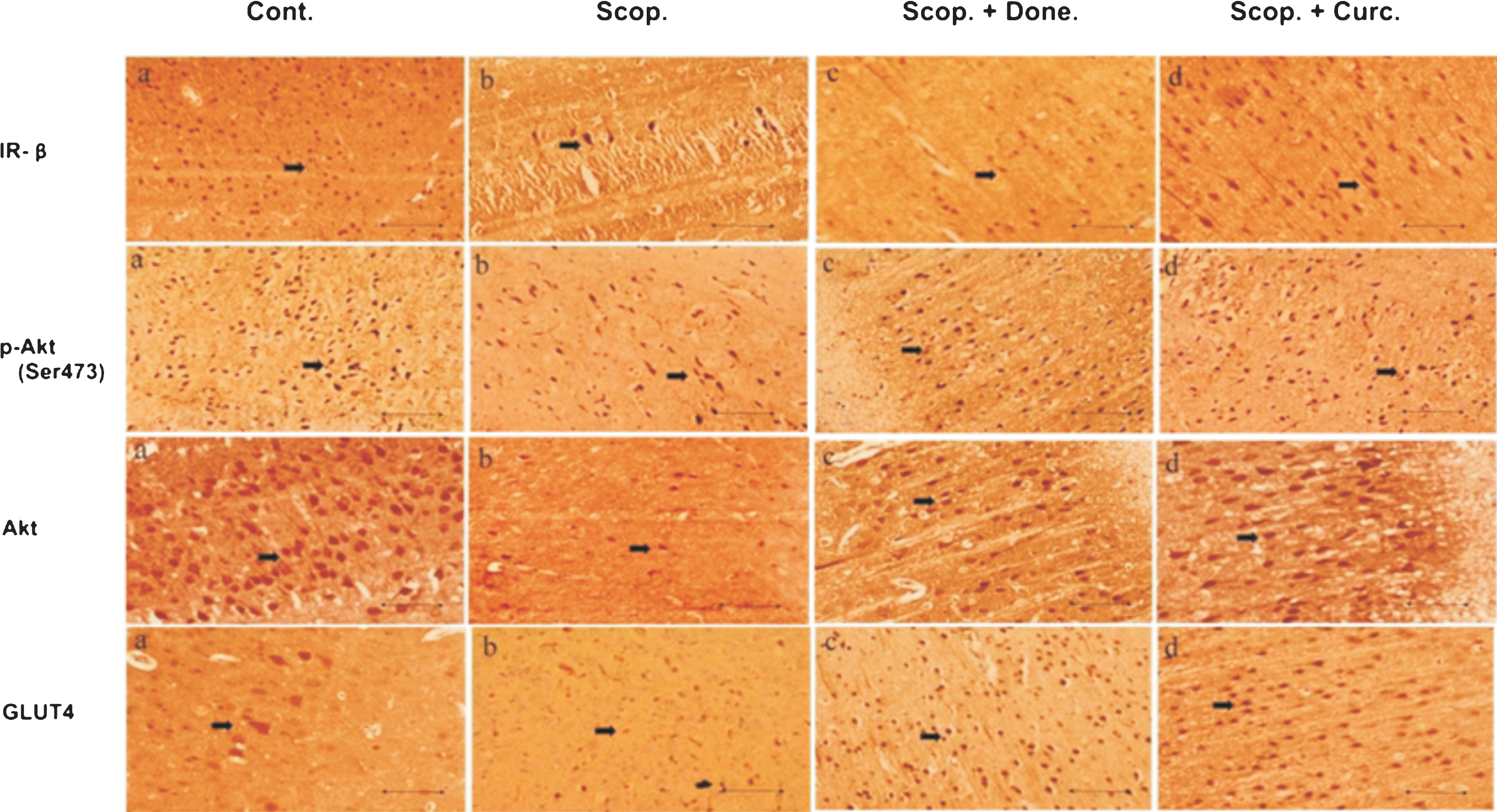
Immunohistochemistry of (i) IR-β, (ii) Akt, (iii) p-Akt (Ser 473), and (iv) GLUT4 in the brain sections (cortex) of several groups of rats. The numbers of immunopositive cells stained with IR-β, Akt, p-Akt (Ser 473) and GLUT4 antibody were decreased in Scopolamine-induced AD rat brain and were increased in Donepezil- and Curcumin-treated AD rats. The assay was performed in triplicate. Lens objective: 40x. Scale bar represents 1 μ. Cont., control; Scop., Scopolamine; Done., Donepezil; Curc., Curcumin.
Lowered levels of upstream insulin signaling proteins in AD rat brain and curcumin significantly reversed it
To examine the expression of major insulin receptor in the brain involved in insulin signaling pathway, we investigated the protein levels of insulin receptor β-subunit (IR-β) (Fig. 3a, a’) and IGF-1 receptor (IGF-1) (Fig. 3b, b’) in AD-rat brain homogenates by immunoblotting. We found a significant decrease in the levels of IR-β (0.72-fold,
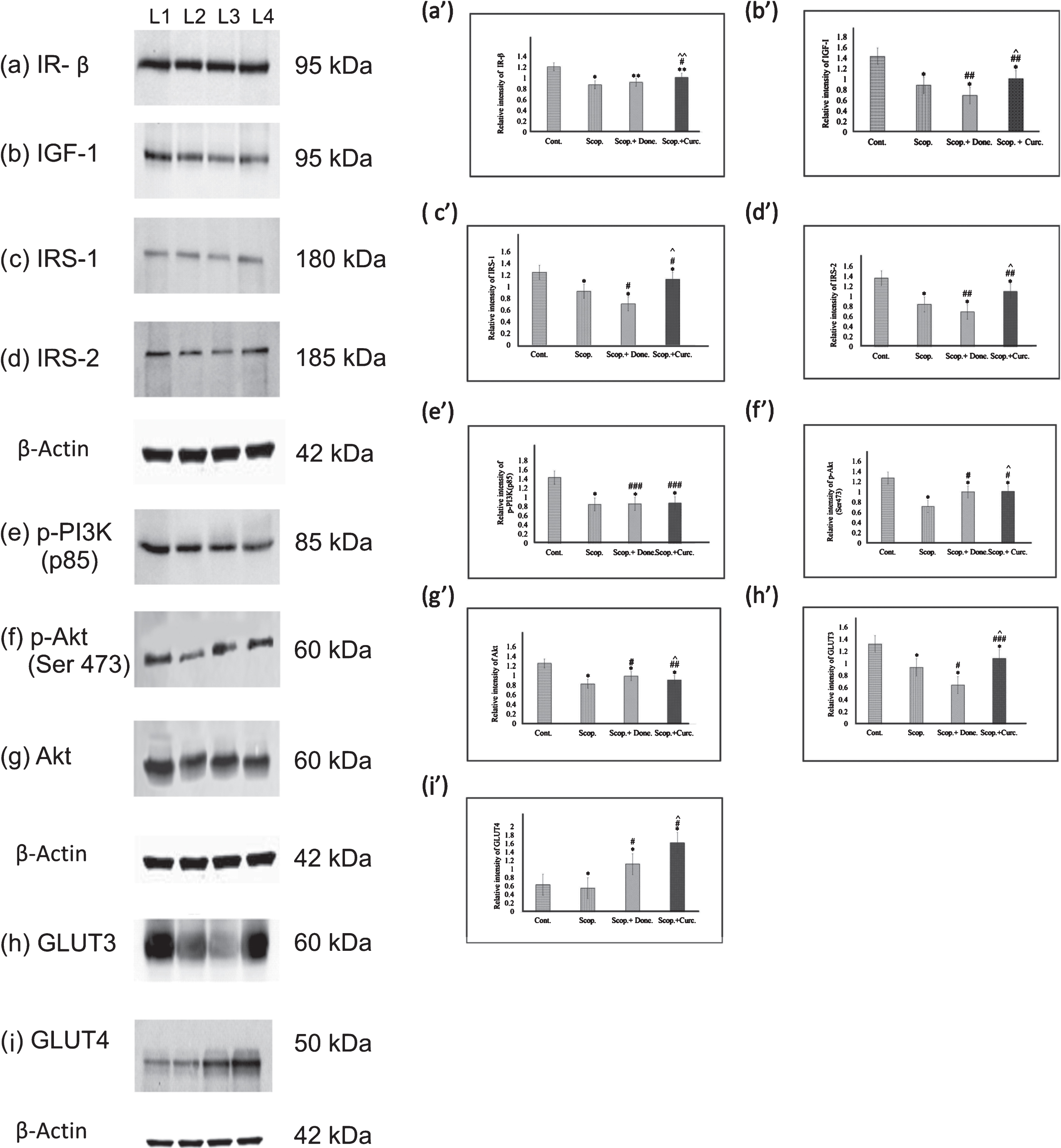
Immunoblotting and densitometry analysis of (a) IR-β, (b) IGF-I, (c) IRS-1, (d) IRS-2, (e) p-PI3K(p85), (f) p-Akt (Ser473), (g)Akt, (h) GLUT3, and (i) GLUT4 in brain lysates. Analysis was performed using NIH ImageJ software and the values obtained for the individual protein target for each brain sample were normalized against β-actin from the same brain sample. The assay was performed in triplicate and ANOVA was used to analyze the differences among groups. Each value represents mean±S.E. for individual rat brain sample (
We next investigated the expressions of insulin receptor substrate 1(IRS-1) (Fig. 3c, c’) and IRS-2 (Fig. 3d, d’) in rat brain. It was found that the levels of IRS-1 (0.74-fold,
Reduced levels of downstream insulin signaling proteins in AD rat brain, curcumin significantly reversed it
We examined quantitatively the expression of p-PI3K (p85) (Fig. 3e, e’) in the rat brains. In AD rats, the levels of p-PI3K (p85) was significantly decreased (0.59-fold,
Lowered levels of glucose transporters in AD rat brain, curcumin significantly reversed it
To investigate if there was any alteration in the levels of glucose transporters among various rat groups, we examined expression of the two key glucose transporters (GLUT3 and GLUT4) in the rat brains quantitatively using immunoblotting (Fig. 3). We observed that the levels of both GLUT3 (0.71-fold,
DISCUSSION
Hypometabolism of glucose is one of the most important key factors in the AD brain and glucose metabolism is linked to insulin signaling pathway. Therefore, impaired insulin signaling may have a pivotal role on impaired cognition such as in AD. Insulin signaling plays important roles in regulation of metabolism of the brain glucose, neuronal development and activities which affect learning and memory [27]. In our studies, we found that the glucose (Fig. 1) and insulin (Fig. 1) levels were decreased in scopolamine-induced brain through the impaired insulin signaling which causes the insulin resistance. These observations indicated that hypometabolism of glucose; hyperinsulinemia and impairment of insulin signaling play roles in the development of AD [28, 29].
In brain, insulin/insulin growth factor (IGF-1) binds to the extracellular
From previous studies, it was found that lower expression of IRs and IGF-1Rs and IRS expressions progressed with AD [23–31]. In addition, insulin/ IGF-1 signaling defects predominately decreased PI3K/Akt pathway-mediated GLUTs activation, and their reduced expression in AD brain may result in brain glucose hypometabolism [23]. The decrease of IGF-I levels indicated that the insulin transport into the brain was impaired [32] or deregulation of blood-brain barrier function was induced by prolonged peripheral hyperinsulinemia [31, 33]. In this study, we found that the levels of upstream insulin signaling molecules such as IR-β (0.72-fold,
Akt regulates phosphatidylinositol-3-kinase (PI3K) survival signaling for insulin-like growth factor I (IGF-I) [36]. Akt also has multiple phosphorylation sites and phosphorylation of Serine 473 (Ser473) in Akt allows full activation of Akt [37]. In general, phosphorylation at Ser473 requires the activation of Akt and the levels of phosphorylation at Ser473 denotes the degree of Akt phosphorylation [38]. The activation of Akt plays a beneficial role in neurodegenerative disease such as AD [39]. In addition, a previous studies [40, 41] had suggested that the activation of Akt prevented neuronal loss in AD and it was reported that there was a reduced localization of cells immunostained for pAkt (Ser473) in AD brains. In our studies, we observed a reduction in the localization of cells immunostained for Akt and pAkt (Ser473) in AD rats compared to Saline-treated control (Fig. 1). Previous study [37] also showed disruption in Akt signaling is an important contributor in the development of AD. We observed a significant reduction in expression of Akt (
GLUT3 is a key neuronal glucose transporter in the brain and it has a higher affinity and more potential [44, 45] than GLUT1 for glucose transport [45, 46]. Therefore it is believed that a higher GLUT3 expression might improve glucose uptake in the neuron [47]. Previous studies showed that GLUT3 levels were decreased [48–50] in AD-affected brain, suggesting a lowered GLUT3 levels may contribute to AD neurodegeneration. Insulin-sensitive glucose transporter GLUT4 [51–53] has been found in the pituitary and the hypothalamus [54]. Furthermore in cells, there was a slow recycling of GLUT4 between the plasma membrane and the vesicular compartments in the absence of insulin [55]. It was found that insulin stimulates glucose transport by translocation of GLUT4 from the intracellular sites to the plasma membrane [56], although the exact mechanisms are unclear [30, 57]. Furthermore, a study using IHC found that, compared to other GLUTs such as GLUT3 and GLUT4 were predominantly expressed in the brain [58]. Another study reported the association of IRSs/PI3K/Akt pathway with high expression of GLUT3, and GLUT4 translocation as well as their attachment to the plasma membrane [59]. In this study, we observed a reduced localization of cells immunostained with GLUT4 in AD rats compared to Saline-treated control (Fig. 2). A study showed that GLUT4 knockout mice had impaired insulin signaling and developed insulin resistance, however the exact mechanism was unclear [60]. It was also suggested that insulin signaling was impaired due to decreased expression of GLUT3 and GLUT4 [61]. In our study, we also observed that in conjunction with impaired insulin signaling, GLUT3 and GLUT4 levels were reduced in AD rats. Interestingly, Curcumin treatment in AD rat brain significantly improved insulin signaling and rescued the reduced expression of GLUT3 and GLUT4 (Fig. 4). In our study, we observed increased insulin levels in AD rats compared to control rats and Curcumin markedly lowered the insulin levels both in the plasma and in the brain. Hyperinsulinemia was shown to be associated with AD pathogenesis in humans [62]. In addition, lower levels of glucose were found in AD patients with significant cognition decline [63]. In our study, glucose levels were decreased in AD rats compared to control rats and Curcumin significantly rescued the lowered glucose levels in AD rats that may lead to improved cognitive function.
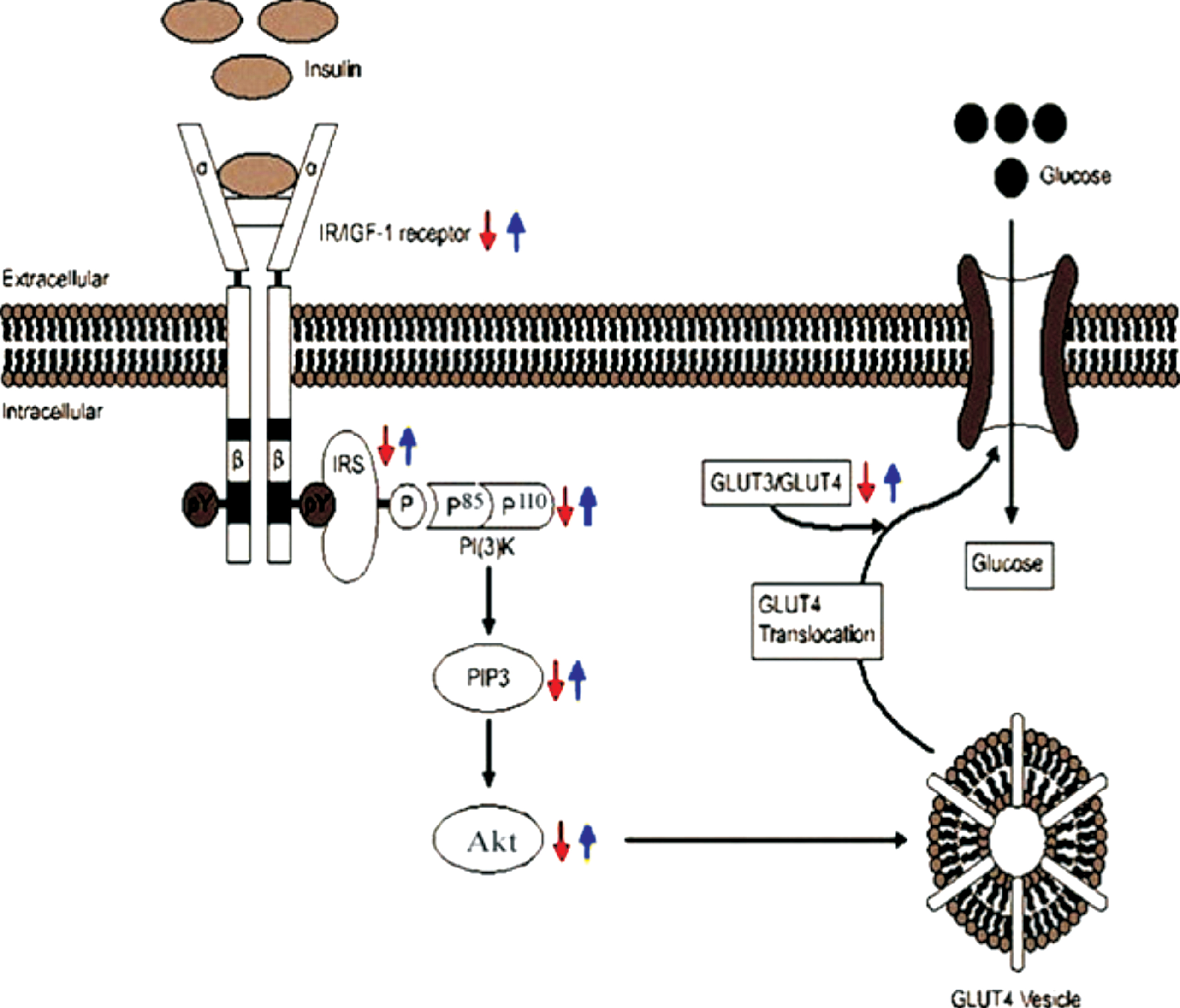
Schematic representation of insulin signaling and the regulation of glucose uptake in the AD brain (red arrow). In our study, due to the reduced levels of IR/IGF, IRS, PI3K, Akt, and pAkt (Ser473) proteins, insulin signaling was impaired in induced AD rats. In addition, GLUT3 and GLUT4 levels were also decreased in AD rats resulting reduced translocation of glucose transporters into the membrane. Curcumin (blue arrow) rescued lowering of the levels of these signaling proteins and glucose transporters in AD rats.
Conclusion
Taken together in the present study we found downregulation of insulin signaling proteins and major glucose receptors in AD rats compared to the Saline-treated control rats. Thus our
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
We thank to Estinnorell Yong, PAPRSB Institute of Health Sciences, Universiti Brunei Darussalam; for her support and contributions to this study. This study was funded by the Universiti Brunei Darussalam/ Brunei Research Council-2(UBD / BRC-2) (Ref: JPKE/DG/83) and Graduate Research Scholarship from UBD (Ref: UBD/GS/1)’2015-16.
