Abstract
Background:
Two large studies suggest that risk is not increased. But other studies have found increased risk of Alzheimer’s disease and impaired cognition.
Objective:
To determine whether androgen deprivation therapy increases the risk of impaired cognition or Alzheimer’s disease in men with prostate cancer.
Methods:
We used data from MedWatch, the Food and Drug Administration (FDA) Safety Information and Adverse Event Reporting Program. Machine-readable data from MedWatch, including adverse drug reaction reports from manufacturers, are part of a public database. We used the online tool OpenVigil 2.1 to query the database. OpenVigil calculates proportional reporting ratios (PRRs) from adverse drug reaction reports to determine whether the combination of drug and adverse event are related. For example, PRR = 2 indicates that the adverse reaction is two times more frequent in users of the drug than in the general population.
Results:
We analyzed adverse event reporting data for these androgen-deprivation drugs: The luteinizing hormone releasing hormone (LHRH) agonists leuprolide, goserelin triptorelin, histrelin; the anti-androgens flutamide, nilutamide, enzalutamide, and bicalutamide; the LHRH antagonist degarelix; the CYP17 inhibitor abiraterone; the anti-fungal ketoconazole, which is also an anti-androgen administered to men with advanced prostate cancer.
Conclusion:
Our analysis of FDA MedWatch adverse event data reports does not support the idea that androgen deprivation therapy
INTRODUCTION
Does androgen deprivation therapy (ADT) increase the risk of impaired cognition or Alzheimer’s disease in men with prostate cancer? Three studies suggest that risk is not increased [1–3]. But other studies have found increased risk of Alzheimer’s disease and impaired cognition [4, 5].
In the present study, we analyzed FDA MedWatch adverse event reporting data for these androgen-deprivation drugs: The luteinizing hormone releasing hormone (LHRH) agonists leuprolide, goserelin triptorelin, histrelin; The anti-androgens flutamide, nilutamide, enzalutamide, and bicalutamide The LHRH antagonist degarelix; The CYP17 inhibitor abiraterone; The anti-fungal ketoconazole, which is also an anti-androgen administered to men with advanced prostate cancer.
We determined the frequency of Alzheimer’s disease and cognitive disorder as adverse events reported after use of these medications, and whether Alzheimer’s disease or cognitive disorder was significantly related to their use.
METHODS
We analyzed data from MedWatch, the Food and Drug Administration (FDA) Safety Information and Adverse Event Reporting Program [6]. MedWatch was organized in 1993 to collect data regarding adverse events in healthcare. An adverse event is any undesirable experience associated with the use of a medical product. The MedWatch system collects reports of adverse reactions and quality problems, primarily due to drugs and medical devices, but also for other FDA-regulated products (e.g., dietary supplements, cosmetics, medical foods, and infant formulas).
MedWatch offers a choice between a voluntary reporting form, designed primarily for health care professionals and the general public, and a mandatory adverse event reporting service (AERS) form, available to manufacturers, importers, and medical product user facilities that manage and store medical products. The latter group is required by law to submit the mandatory form immediately upon discovery of a product malfunction. Printable mail-in forms are available as an alternative to the online submission system [7].
A MedWatch report of an adverse event does not establish causation. For any given report, there is no certainty that the drug in question caused the reaction. The adverse event may have been related to the underlying disease being treated, another drug being taken concurrently, or something else.
Machine-readable data from MedWatch, including adverse drug reaction reports from manufacturers, are part of a public database. We used the online tool OpenVigil 2.1 to query the database [8, 9]. OpenVigil data are exclusively from FDA and MedWatch, not from social media [10]. OpenVigil calculates proportional reporting ratios (PRRs) from adverse drug reaction reports to determine whether the combination of drug and adverse event are related, using the criteria of Evans et al. [11]. PRR = 2 indicates that the adverse reaction is two times more frequent in users of the drug than in the general population. According to the criteria of Evans et al. [11]
Ketoconazole, an anti- fungal, blocks production of androgens. Ketoconazole is most often used to treat men just diagnosed with advanced prostate cancer; it quickly reduces testosterone levels and can be administered if other forms of hormone therapy are no longer effective. To assess the relationship of ketoconazole and Alzheimer’s disease in prostate cancer, we restricted the OpenVigil analysis to men over age 60.
The MedWatch data are imperfect, with under- and over-reporting, missing denominator (that is, number of doses for a drug), wrong, duplicate and/or missing data in the database [8]. Consequently the total number of adverse event reports for all drugs and/or the drug in question from OpenVigil can vary slightly from drug to drug and for different adverse events related to the same drug. The flawed MedWatch data has presented a problem that all analytical software, such as OpenVigil, has been forced to confront [12]. For example, the OpenVigil report for leuprolide indicates that MedWatch had received a total of 8,803 adverse event reports relevant to leuprolide. Of these, 6 were of dementia Alzheimer’s type (0.068%). In comparison, for all drugs in MedWatch there were 1988 reports of Alzheimer’s disease among a total number of 3,792,386 adverse events. But in the case of cognitive disorder, 18 adverse event reports were of leuprolide and cognitive disorder (0.20%). In comparison, for all drugs in MedWatch there were 10,052 reports of cognitive disorder among a total number of 3,794,374 adverse events.
RESULTS
Data to evaluate the criteria of Evans et al. for Alzheimer’s disease and androgen deprivation drugs are listed in Table 1. Alzheimer’s disease, according to the criteria, is unrelated to any of the drugs.
Data to evaluate the criteria of Evans et al. for Alzheimer’s disease and androgen deprivation drugs
Alzheimer’s disease, according to these criteria (
Data to evaluate the criteria of Evans et al. for cognitive disorder disease and androgen deprivation drugs: Cognitive disorder, according to these criteria, is unrelated to any of the drugs
*Ketoconazole analysis restricted to males over 60.
Data to evaluate the criteria of Evans et al. for cognitive disorder disease and androgen deprivation drugs are listed in Tables 1 and 2. Cognitive disorder, according to the criteria, is unrelated to any of the drugs.
Proportional reporting ratios for Alzheimer’s disease and cognitive disorder for the individual drugs are summarized in Fig. 1.
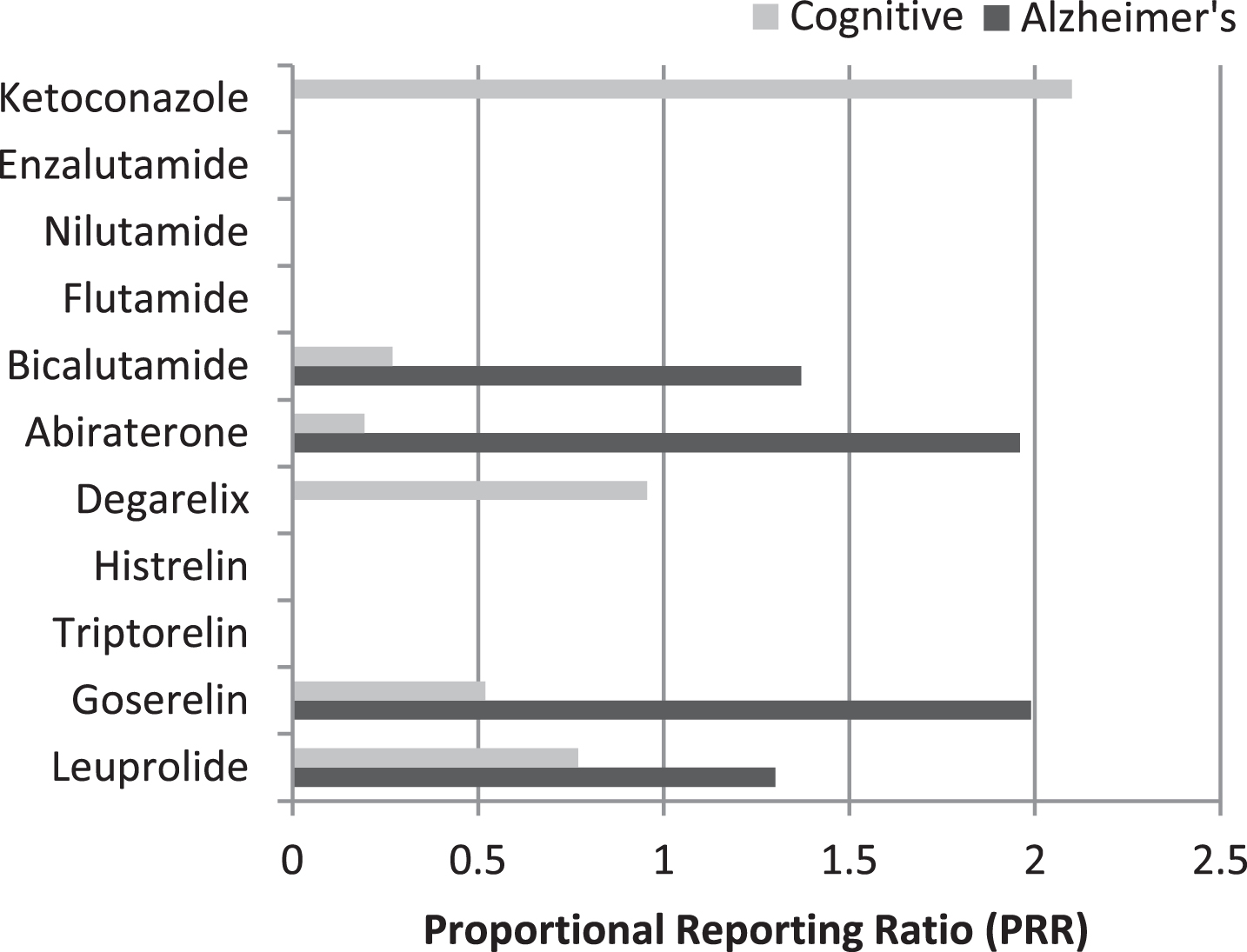
Proportional Reporting Ratios (PRR) for Alzheimer’s disease and cognitive disorder of androgen-deprivation drugs.
We performed a second analysis looking at all androgen deprivation drugs taken together compared to the entire MedWatch database. Because reports of Alzheimer’s disease in patients below the age of 60 are uncommon, we restricted the analysis to male patients over 60.
MedWatch received a total of 6,041 adverse event reports for all androgen deprivation drugs. Of these, 7 were of dementia Alzheimer’s type (0.12%). In comparison, for all drugs in MedWatch there were 361 reports of Alzheimer’s disease among 346,170 adverse events. Chi-Squared with Yates’ correction: 0.006. PRR and 95% confidence interval (lower bound; upper bound): 1.1 (0.53; 2.35). According to the criteria of Evans et al. the combination of anti-androgenic drugs and Alzheimer’s disease is probably not related.
Of the 6,041 adverse event reports for all androgen deprivation drugs, 7 were of cognitive disorder (0.12%). In comparison, for all drugs in MedWatch there were 659 reports of cognitive disorder among 346,170 adverse events. Chi-Squared with Yates’ correction: 1.37. PRR and 95% confidence interval (lower bound; upper bound): 0.61 (0.29; 1.28). According to the criteria of Evans et al. the combination of anti-androgenic drugs and cognitive disorder is probably not related.
DISCUSSION
Testosterone is related to cognition. Sex hormones affect brain development. Androgens modify neural activity needed for learning and memory, are neuroprotective during aging, protect against Alzheimer’s disease in mouse models [13] and, hypothetically, humans [14]. The androgen receptor directly mediates neuroprotection [15]. But testosterone apparently does not affect those parts of the brain that demonstrate sex differences in performance; and no one knows whether testosterone is necessary to maintain intellect throughout life [16].
Testosterone deprivation may impair memory in older men [17]; while testosterone supplementation can augment memory and spatial perception. Studies of prostate cancer demonstrate that androgen deprivation drugs adversely affect cognition [18], which returned to baseline when drugs were withdrawn [19]. Moreover, LHRH agonist use as compared with no use in men with prostate cancer was associated with a decreased risk of death from Alzheimer’s disease [20].
Nevertheless, two large studies failed to confirm any effect of androgen deprivation on cognition or Alzheimer’s disease in men with prostate cancer [1, 2]. Our analysis of FDA MedWatch adverse event data reports, likewise, does not support the idea that androgen deprivation therapy
A weakness in our MedWatch analysis is that it represents an uncontrolled epidemiological study. A major improvement in design would be a prospective randomized trial. A greater design improvement would be repeated measures comparing the same subject under different conditions.
Does androgen deprivation result in Alzheimer’s disease and cognitive dysfunction in prostate cancer patients? More studies are needed for a definite answer.
