Abstract
Little is known concerning the exact mode of action of most natural antibiotics, other than that they produce several general changes in bacterial morphology and respiration. Since biological activity can often be referred to specific groups in the molecule, metabolic studies involving smaller moieties of antibiotic molecules might possibly provide an approach to this problem. A preliminary attempt along these lines involving the nucleus of the penicillin molecule is described in this paper.
Experimental. 4-Thiazolidinecarboxylic acid, thioproline, was synthesized by the method of Armstrong and du Vigneaud(1). The organism, 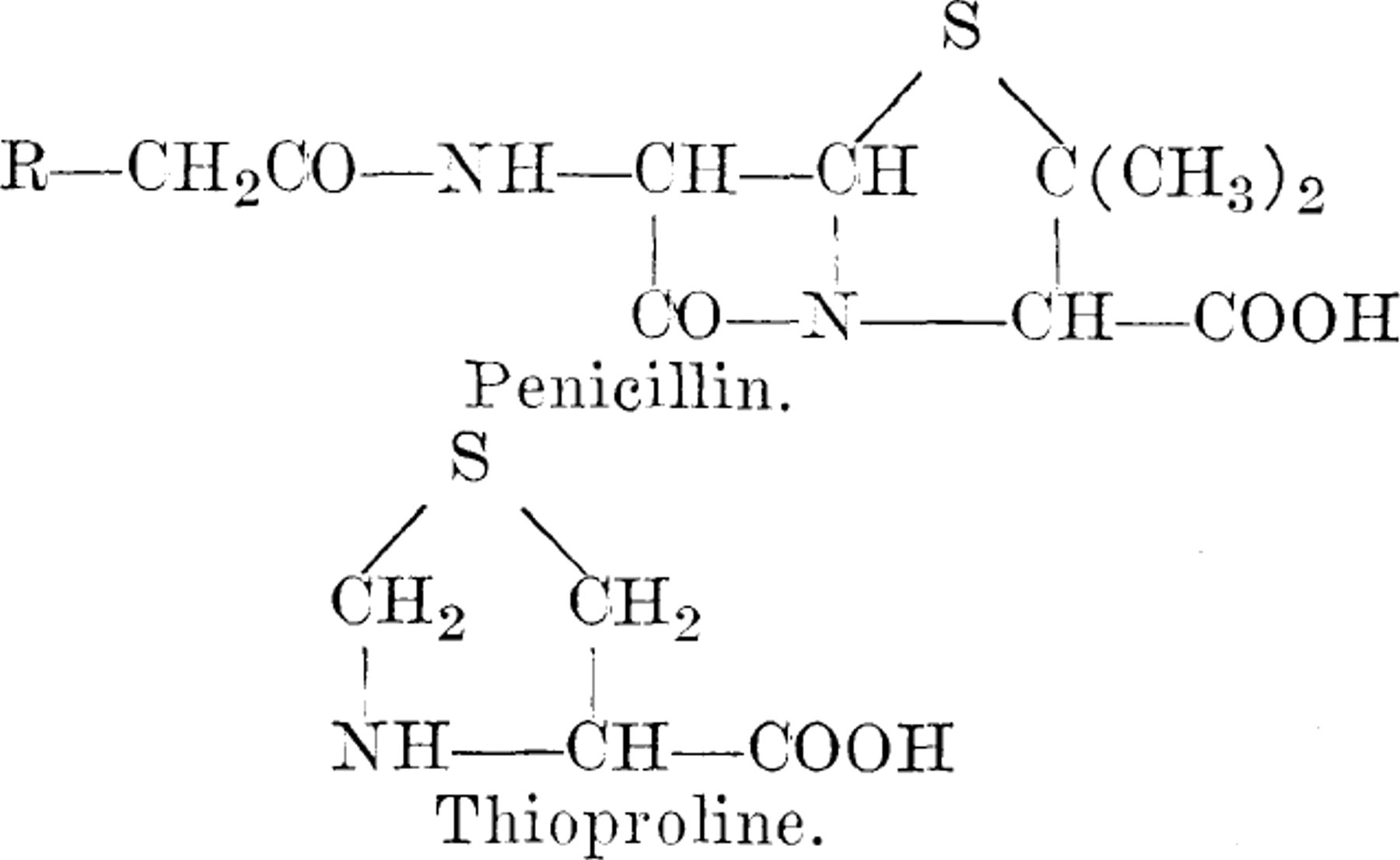 medium, and technics employed were those previously described and extensively used in analogue-metabolite inhibition studies (2). The results are summarized in Table I. Thioproline effectively inhibits the growth of Escherichia coli in mineral salts-glucose medium, and the inhibition is reversed to a limited extent by proline, despite the fact that the inhibitor is not isosteric with proline. To a somewhat lesser extent, a large number of other amino acids can also reverse the inhibition. Vitamins, purines, pyrimidines, and increased levels of glucose, however, are inactive in this regard. Combinations of amino acids manifest an even greater reversing ability than any single substance.
medium, and technics employed were those previously described and extensively used in analogue-metabolite inhibition studies (2). The results are summarized in Table I. Thioproline effectively inhibits the growth of Escherichia coli in mineral salts-glucose medium, and the inhibition is reversed to a limited extent by proline, despite the fact that the inhibitor is not isosteric with proline. To a somewhat lesser extent, a large number of other amino acids can also reverse the inhibition. Vitamins, purines, pyrimidines, and increased levels of glucose, however, are inactive in this regard. Combinations of amino acids manifest an even greater reversing ability than any single substance.
Discussion. A consideration of the data suggests that thioproline may inhibit bacterial growth by preventing the conversion of a large number of amino acids to some product or products essential for growth. It is apparently most effective in preventing proline utilization, perhaps because of its structural similarity to this amino acid. The known facts regarding penicillin action are not out of line with the possibility that penicillin prevents bacterial protein synthesis.
Get full access to this article
View all access options for this article.
