Abstract
The albumin used in these studies was prepared from dog liver perfused in situ until free from blood. In all cases the animals had been well fed. The organ was excised, frozen in liquid air, powdered, and stored at −13°C. Preparation of the liver albumin followed upon extraction of the powdered liver with 0.5 M (NH4)2SO4, and salting out of the globulins in 2.1 M (NH4)2SO4. The albumin was salted out by increasing the concentration of ammonium sulphate to 3.5 M and was rendered free from any detectable amount of globulin by reprecipitation with ammonium sulphate. After 3 or 4 re-precipitations, the amount of material salted out in 2.1 M (NH4)2SO4 was reduced to the vanishing point. The pH was maintained at 6.5 to 7.0 throughout this treatment. The amount of albumin obtained from 100 g of liver was 0.9 to 1.5 g.
Electrophoretic studies were carried out in the Tiselius apparatus, using for observation of the boundaries the Toepler “Schlieren” method as modified by Longsworth. In a medium consisting of 0.1 M NaCl and 0.02 M KH2PO4/K2HPO4, following upon dialysis against a fluid of the same composition, electrophoretic analysis (pH 5.8 to 7.3) revealed 2 fractions; both moved towards the positive electrode. One fraction was considerably larger than the other and moved more slowly.∗ The other fraction, small in quantity, has shown much variability; in some cases it was practically absent. This fraction is isoelectric at pH 4.7 and we suspect it may consist of traces of serum albumin not washed out during perfusion of the liver.
The major fraction has always displayed much stability and homogeneity in media consisting of 0.1 M NaCl and 0.02 M in either 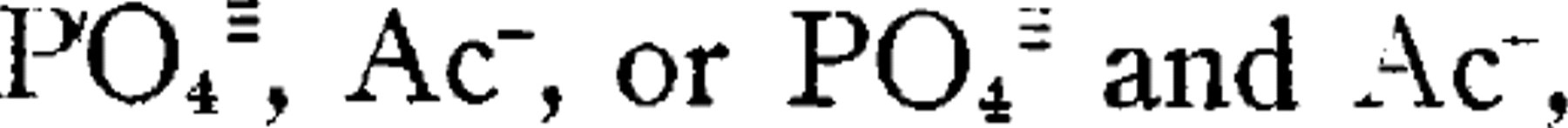 , according to the pH; many runs have been made over a wide range of pH (3.9 to 8.0) without evidence of heterogeneity.
, according to the pH; many runs have been made over a wide range of pH (3.9 to 8.0) without evidence of heterogeneity.
Get full access to this article
View all access options for this article.
