Abstract
Several investigators 1-4 described in preliminary form the synthesis of 2-methyl-3-phytyl-1,4-naphthoquinone and its identification with vitamin K1. In regard to their respective biological activities, general statements have been made to the effect that both compounds have equal potency. 3-6
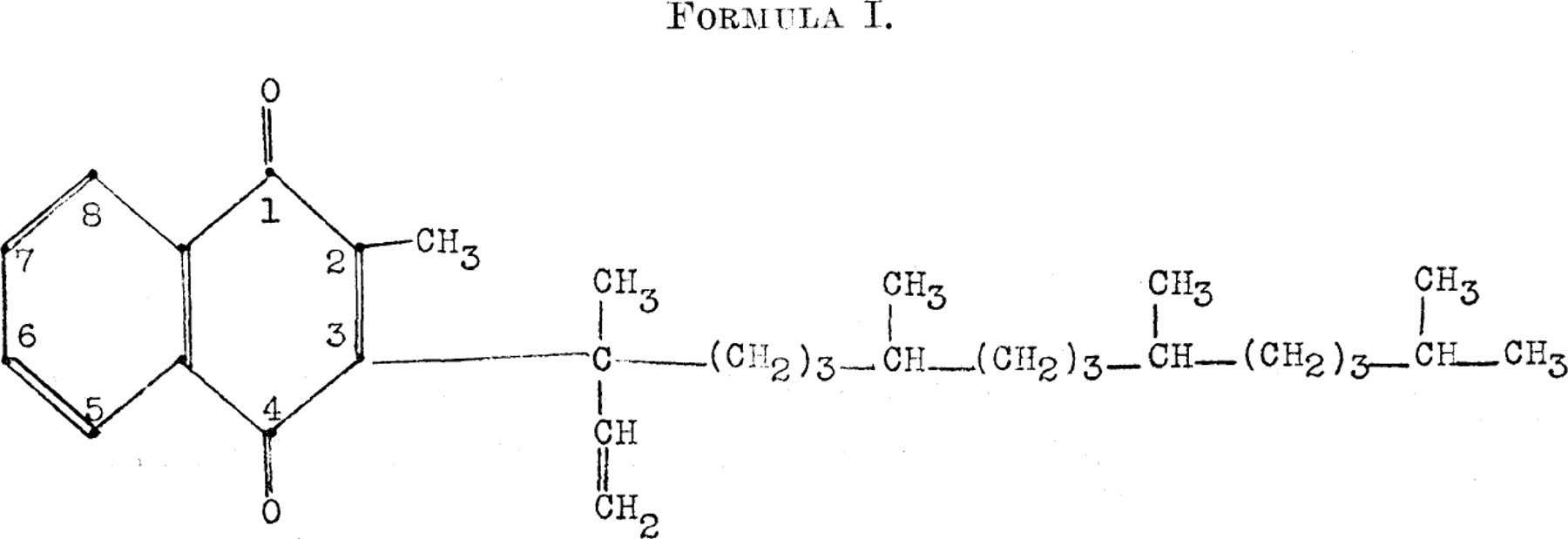
We have prepared 2-methyl-3-phytyl-1,4-naphthoquinone by a modified Fieser 3 procedure with final purification by chromatographic adsorption on calcium sulfate. Using the method of Claisen and Eisleb, 7 we synthesized an isomeric compound, 2-methyl-3-“iso-phytyl”-1,4-naphthoquinone (Formula I). The biological assays of these compounds are shown in Table I together with those of the Synthetic Vitamin K1 (Merck),∗ of the 2-methyl-1,4-naphthoqui-none and the diacetate of the corresponding hydroquinone, and of the diacetate of the 2-methyl-3-phytyl-1,4-naphthohydroquinone. The data presented permit the conclusion that the 2-methyl-3-phytyl-1,4-naphthoquinone has a potency of 1 unit 8 in about 71/2 γ, its diacetate derivative 1 unit in approximately 15 γ. Furthermore, the Synthetic Vitamin K1 (Merck), the isomeric compound and 2-methyl-3-dihy-drophytyl-1,4-naphthoquinone showed 1 unit in about 15 γ, whereas the methylnaphthoquinone and the diacetate of its corresponding hydroquinone had 1 unit in about 1/2 γ and lγ, respectively, as already reported. 9
The phytyl derivatives appear to have considerably more potency, when the test period is prolonged to 18 hours, as shown in Table II; The speed of action of the unit of 2-methyl-1,4-naphthoquinone is practically identical with that of the unit of the natural vitamin K. However, the phytyl derivatives are not as rapidly utilized by the animal body, as a comparison of Table I with Table II and Table III shows.
Get full access to this article
View all access options for this article.
