Abstract
It has been known for many years that epinephrine may be transported by electrophoresis into the skin where it produces its typical blanching in the intact dermis. As far as the writer is aware, this phenomenon has not been applied to the electrical transport of comparatively large quantities of epinephrine into the skin to establish locally in the skin reservoirs of epinephrine for the relief of allergic states. A method of administering epinephrine by electrophoresis to form these reservoirs has now been developed. This technic and its application to the relief of severe asthma form the subject of this paper.
Preparation of the solution: With the current densities needed, solutions of epinephrine hydrochloride were too acid to employ. A solution of epinephrine dihydrogen phosphate between pH 3 and 4 was tentatively adopted. This solution is prepared by simply adding equivalent quantities of epinephrine base and phosphoric acid so that the final concentration of epinephrine itself is 1% (with suitable adjustment of pH).
Quantity transported: This solution contains mainly the ionic species:
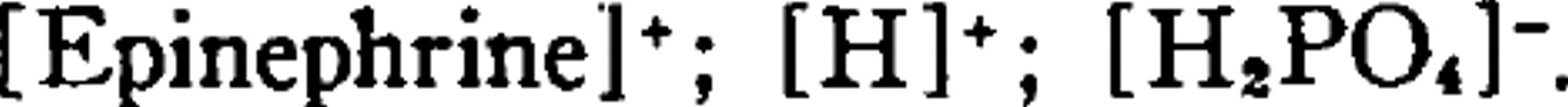
Although the electric mobility of H+ is much greater than either of the other 2 ions, its concentration is comparatively small even at pH 3.0. In the routine method now employed a single treatment lasts 30 minutes (occasionally more) with the current at 0.005 amp. Since 1.0 ampere is one coulomb per sec., approximately 9 coulombs are transported. Assuming that Faraday's laws hold here and that the electric mobility of the epinephrine ion is equal to that of [H2PO4]−, it can be readily shown that the epinephrine ion accounts for the passage of approximately 2 coulombs; therefore, more than 2 × 10−5 moles of epinephrine are electrically transported into the skin. Since the molecular weight of epinephrine is near 183, about 4 mg of epinephrine are probably deposited in the skin by the method outlined in a single treatment.
Get full access to this article
View all access options for this article.
