Abstract
The absorption spectra of oxyhemoglobin, carboxyhemoglobin, cyarihemoglobin, methemoglobin (pH 5.9) and methemoglobin (pH 9.2) have been studied quantitatively in both the visible and ultraviolet regions.
These pigments have complex absorption patterns. The absorption curves are all very different in the visible region, but exhibit a general similarity of shape in the ultraviolet where most of the light absorption is evident.
The absorption curves of certain far simpler substances have yielded to an analysis which resolves the complex of peaks and troughs into a series of curves (bands), whose summation gives the observed absorption pattern. 1 Bands whose peaks are at equal frequency distances from each other may be considered to possess an intimate relationship, and probably represent the same fundamental disturbance in the molecule caused by the absorption of energy. This is an important deduction since it greatly simplifies the interpretation of a complex absorption curve.
It is noteworthy that this type of analysis may be applied to the absorption curves of such complex molecules as these various hemoglobin derivatives. The spectrum of cyanhemoglobin is composed of a single series of bands, spaced at regular intervals. The absorption curves of the other pigments studied possess bands which belong to the same series, although they also show other bands. Cyan-hemoglobin and oxyhemoglobin may be used as examples (Table I).
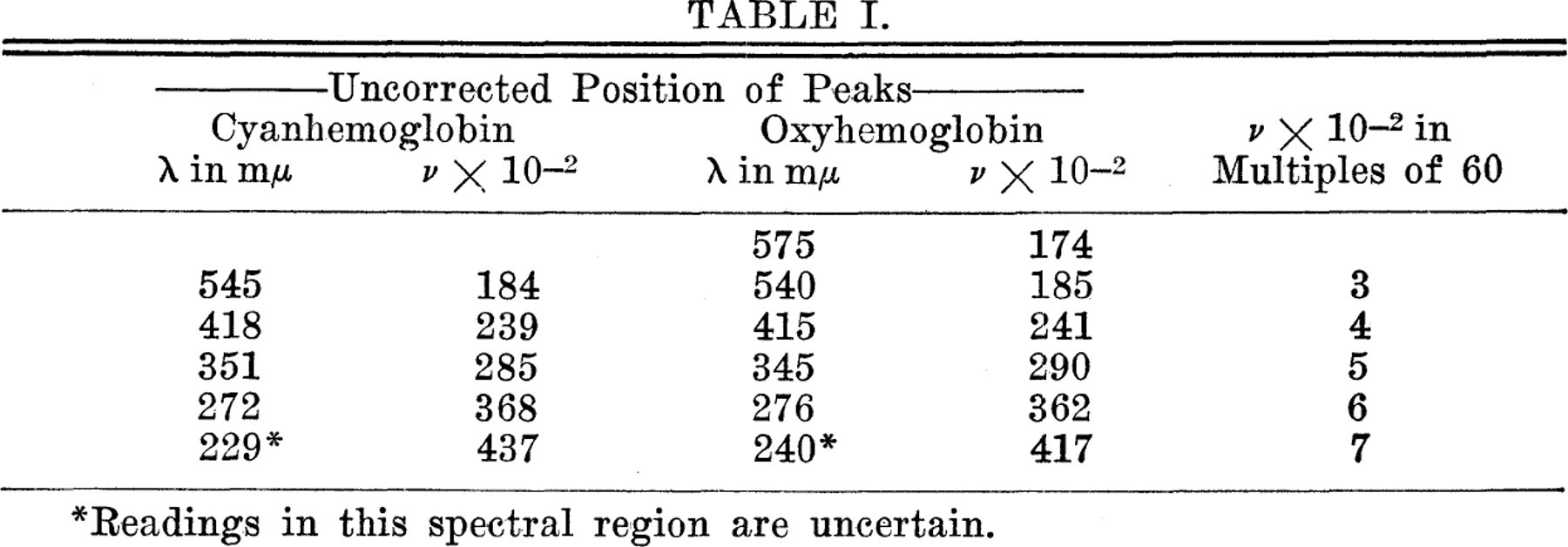
This preliminary analysis permits the following tentative deductions to be drawn: All the hemoglobin derivatives have bands which belong to a single series, the members of which may be expressed by n = v ×10−2/60. n represents a simple integer, such as 3, 4, 5, 6, and 7, which are demonstrable in the regions of the spectra studied. This series of bands is probably related to the general structure of the hemoglobin molecule.
The so-called α-band of oxyhemoglobin (peak at λ 575 mμ.) probably belongs to another series.
Get full access to this article
View all access options for this article.
