Abstract
To elucidate the relationship between the chemical structure and the pharmacological action in the barbital series, over 50 derivatives were investigated. These compounds were all 5, 5-substituted barbituric acids, having the general formula: 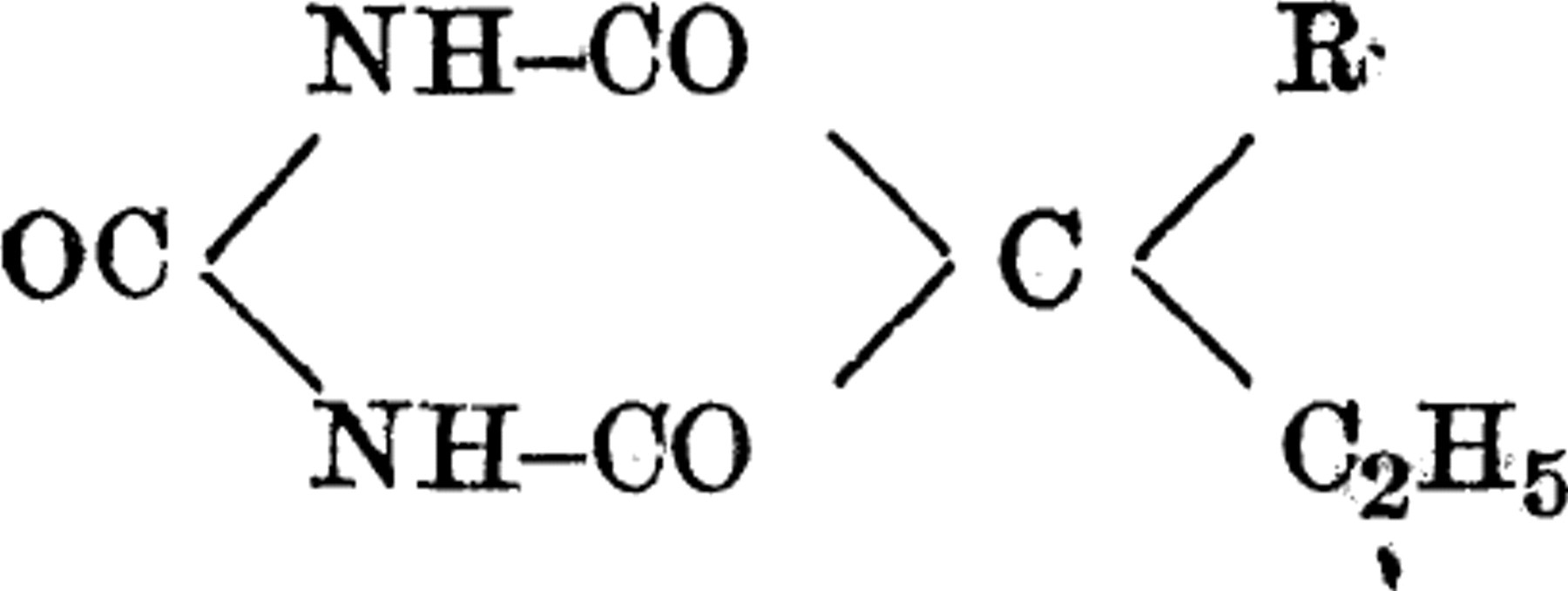
wherein R-alkyl radical (normal or secondary with 2 to 9 C-atoms). Several of them were new compounds synthesized for the first time. 1
Albino rats weighing from 71 to 126 gm. (average 96 gm.) were employed in this study. Solutions of the sodium salts of the compounds were injected intraperitoneally. The minimal anesthetic dose (M.A.D.), the duration of action, and the minimal lethal dose (M.L.D) were determined by using 5 animals for each dose level.
Since space does not permit a presentation of detailed results, only a few salient points will be discussed here. In Figs. 1 and 2, the “primary alkyls” refer to those compounds having a normal alkyl group. It should be noticed from Fig. 1 that with the increase in number of C-atoms in the alkyl group, either normal or secondary, both the M.A.D. and M.L.D. grow relatively smaller, but when the alkyl radical is longer than 5 C-atoms, the amount required to anesthetize or kill rats again increases. The therapeutic index, or the ratio between the M.L.D. and M.A.D., appears to be gradually greater as the alkyl chain lengthens.
In general, the duration of action shows similar features; that is, it is shorter when the alkyl group becomes lengthened (Fig. 2). In the series of normal alkyl derivatives, the critical compound is the one that possesses 6 C-atoms at R; and in the series of secondary alkyl derivatives, the critical compound is the one having 7 C-atoms at R, beyond which the duration of action begins to increase.
Get full access to this article
View all access options for this article.
