Abstract
In addition to procedures previously described, 1 we have adopted the principles of Myers' and Wardell's 2 blood cholesterol method for extraction of barbiturates.
A convenient amount of acidulated urine, blood (1 to 2 cc), or well-ground, liquified tissues is pipetted into a small mortar containing sufficient Plaster of Paris to form a dry mixture, which is then pulverized and transferred to a paper extraction shell. This shell is inserted into a perforated glass tube connected to a reflux condenser. Ten volumes of chloroform (10 to 20 cc.) are poured into the extraction flask, which in turn is heated to boiling on a water-bath for half an hour. The chloroform extract is then filtered and tested. If the test is negative the extract may be concentrated over a water-bath.
We have increased the sensitivity of our test previously described 3 by using cobalt acetate and, instead of the methyl alcohol solution of barium hydroxide, a 0.5% solution of lithium hydroxide in absolute methyl alcohol. Two or 3 drops of the 1% cobalt acetate and 0.5% lithium hydroxide reagents will produce a definite blue color in chloroform extracts of concentrations as low as 0.0025%. However, chloroform extracts can be concentrated 20 times, thus making the test sensitive to 1.25 parts per million.
We have described our test as colorimetric determination, but without the actual use of a colorimeter, because the colors produced by barium or other metallic hydroxides soon precipitate or fade if transferred to colorimeter cups. We have now succeeded in adopting the test for colorimetric readings with standard colorimeters. The procedure is as follows:
One cc. of the unknown chloroform extract is treated first with 0.05 cc. of a 1% cobalt acetate solution in absolute methyl alcohol and 0.3 cc. of a 5% (by volume) solution of iso-propyl amine (Research Laboratories, Eastman Kodak Co.) in absolute methyl alcohol. If barbiturates are present in the extract, a reddish-violet color develops, which is then compared in micro-cups with the color produced under the same conditions by barbital solutions of known strength in chloroform. We found that a 0.025% solution of barbital in chloroform usually suffices as a standard, against which unknown solutions over a wide range may be read. A simple formula, which may be used with any standard colorimeter, giving the number of milligrams of barbiturate per cc. of chloroform extract using the 0.025% solution as the standard is:
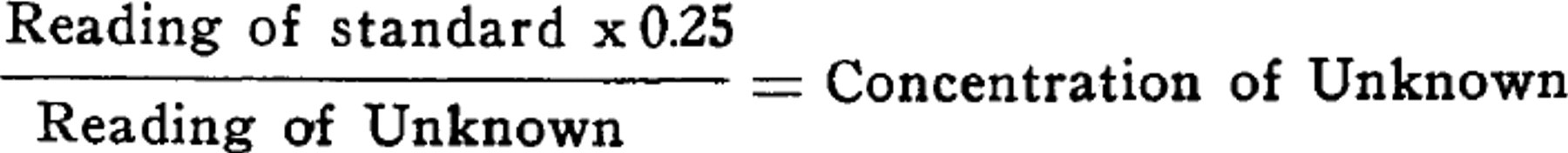
Get full access to this article
View all access options for this article.
