Abstract
In studying the mechanism of urease action, it would be of interest to find a derivative of urea that would be hydrolyzed in the presence of urease. Urease is very specific in its action and urea appears to be its only substrate. Closely related compounds such as amides or purines as well as simple derivatives of urea are not attacked by this enzyme. Armstrong and Horton
1
found that substitution in the urea molecule with methyl or ethyl groups invariably rendered the substituted urea inaccessible to urease. Schoorl
2
synthesized glucose-urea, in which one molecule of urea was united with one molecule of the sugar. This compound is very soluble in water and is stable in solution. When heated with acid it undergoes hydrolysis with the formation of 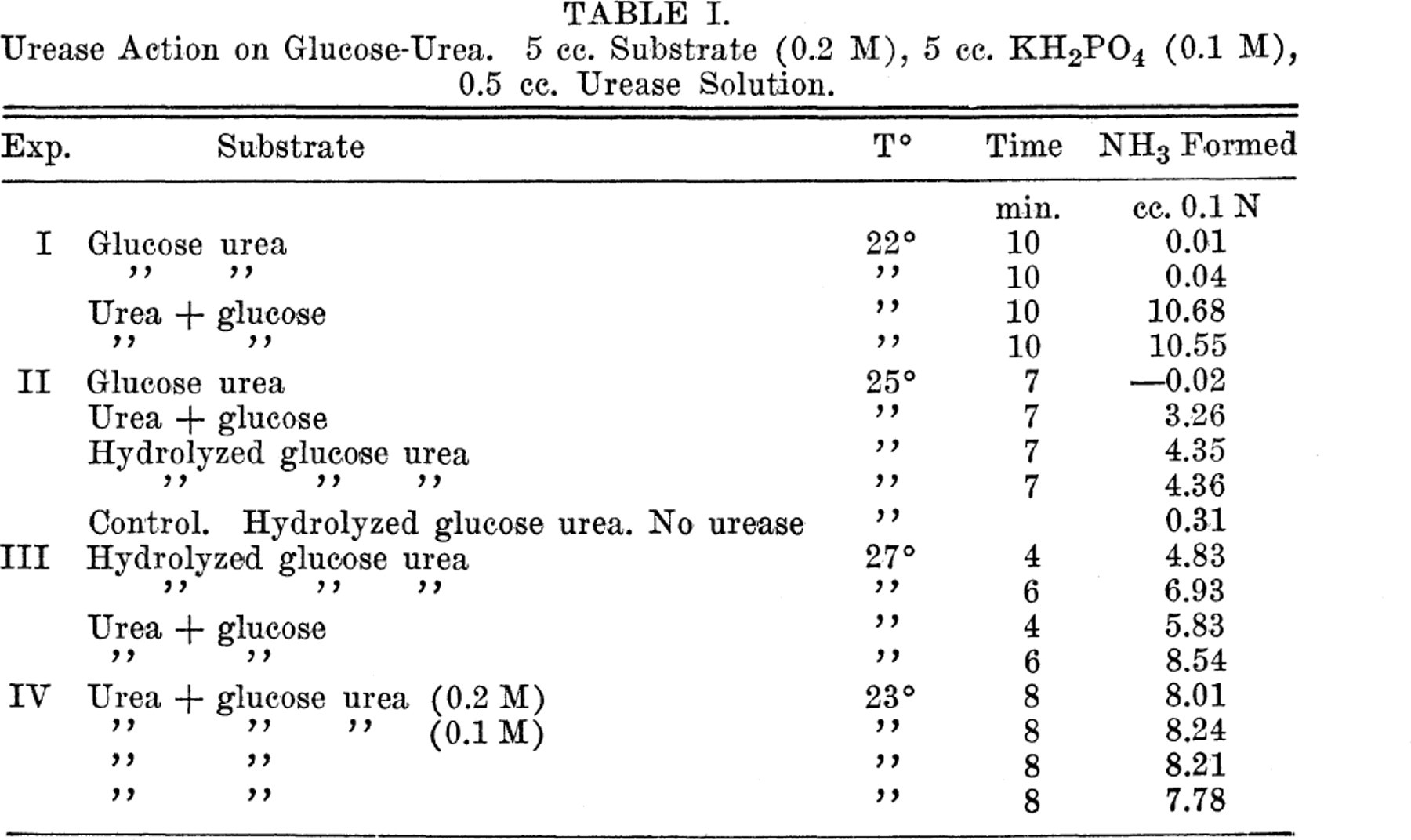 -glucose and urea. Johnson and Bergmann,
3
in their recent researches on nitrogenous glucosides have prepared glucose urea, and Dr. Johnson generously placed at our disposal a very pure sample of glucose urea for investigation. We have studied the action of urease on this urea derivative.
-glucose and urea. Johnson and Bergmann,
3
in their recent researches on nitrogenous glucosides have prepared glucose urea, and Dr. Johnson generously placed at our disposal a very pure sample of glucose urea for investigation. We have studied the action of urease on this urea derivative.
Jack bean urease was found to have no ability to decompose glucose urea. There was no ammonia production in 10 minutes when urease was added to 0.2 M solutions of this compound. However, if glucose-urea was hydrolyzed by acid prior to the addition of urease, ammonia production occurred at a rate comparable to that observed when a 0.2 M solution of urea and glucose was exposed to urease. The results (Table I) clearly demonstrate that the inability of urease to split glucose urea must be ascribed to the chemical make-up of the compound and cannot be the result of the presence, in the preparation, of substances that inactivate the enzyme.
Get full access to this article
View all access options for this article.
