Abstract
It has become common practice to express temperature effects on rates of biological reactions in terms of the Van't Hoff-Arrhenius relationship by the use of one of the following equations:
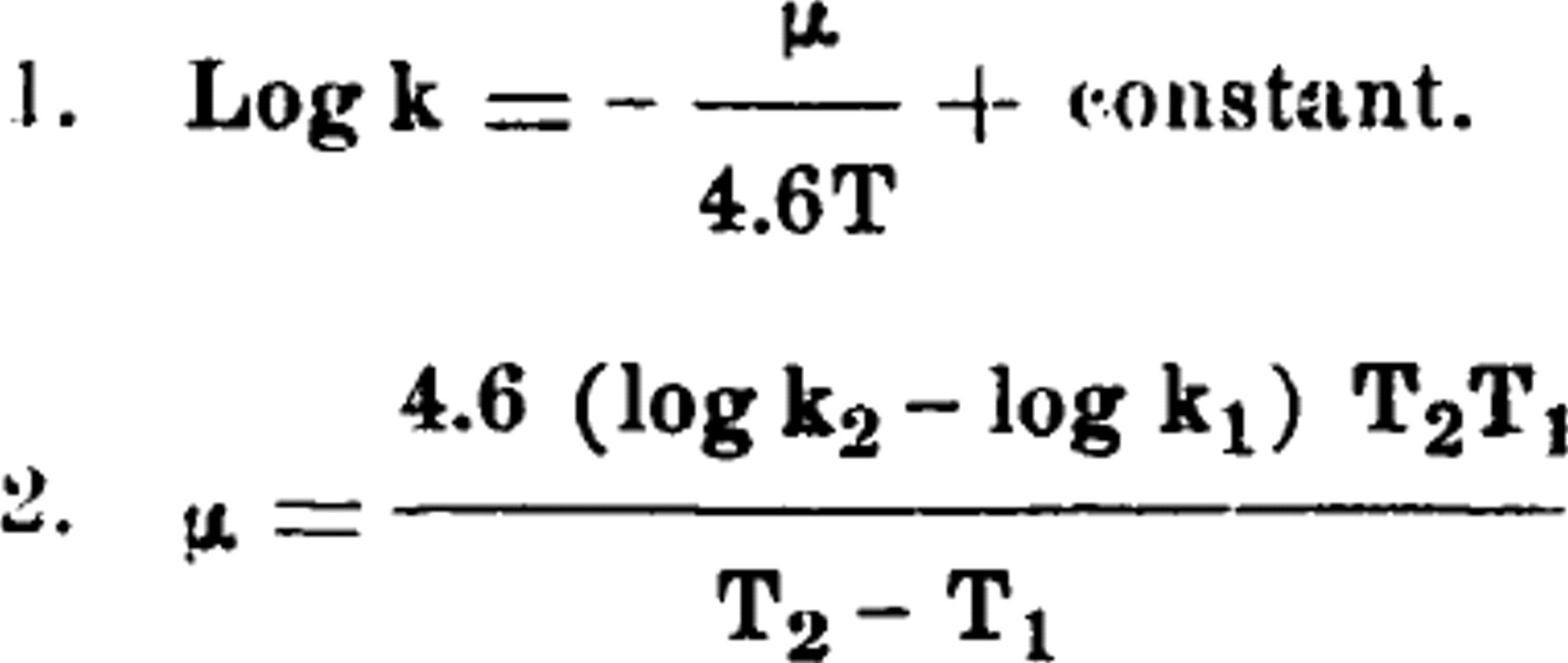
in which k2 and k1 are the velocity coefficients at absolute temperatures T2 and T1. The symbol μ has been called the thermal increment or thermal constant. The latter term is misleading since μ cannot be assumed to be a constant but its constancy must be proved for the temperature range under consideration. It is the purpose in this paper to discuss the value of μ as a function of temperature as calculated by means of Equation 2 for several biological processes and to point out instances where the use of Equation 1 has given misleading results.
In Table I are given values for μ calculated from data given by Sherwood and Fulmer 1 for the growth of Saccharomyces cerevisiae in various media. It is evident that μ decreases regularly with increase in temperature. In 3 of the media the value of μ is a linear function of the temperature. Moreover the medium used influences markedly the values of μ. It is of particular interest to note the influence of ammonium chloride both in the synthetic medium and in the beer wort. The notations μ10 and μ5 have been used; since μ decreased regularly with increase in temperature its value will necessarily vary with the temperature range employed in its calculation.
An analysis of data by Slator 2 on the fermentation of different sugars by several yeasts shows that μ decreased regularly with increase in temperature. The value of μ for sporulation rate of Saccharomyces pastorianus I as calculated from the data of Herzog 3 is a linear function of the temperature, i. e., μ = 44,100-1300t°.
Get full access to this article
View all access options for this article.
