Abstract
Since the initial communication of Starling, 1 , 2 it has been established that the serum proteins exert an osmotic pressure. The same investigator showed later that the colloid osmotic pressure is approximately proportional to the concentration of protein in the serum. It has been found that as serum is diluted the colloid osmotic pressure falls more sharply than does the concentration of the serum proteins. Verney 3 believes that the explanation of this phenomenon is to be found in the comparatively large molecular volume occupied by the colloidal particles, the line of reasoning being analagous to that employed in deriving the pressure-volume relationship of highly compressed gases. He finds that, where p is the colloid osmotic pressure of the proteins, v the reciprocal of the protein concentration, b is a constant depending on the particular conditions, the formula p(v—b) = k holds for dilutions up to 50% of that of normal serum. Thus he finds that the colloid osmotic pressure of the serum proteins varies with their dilution, within definite limits, in the manner that would be expected of an unionized colloidal solution in which the colloidal particles occupy an effective volume as large as 50% of the original.
It is thus known that the colloid osmotic pressure of the serum per gm. of protein becomes lower, the less the actual concentration of the protein in the serum. If we assume that the rate of change of the colloid osmotic pressure with respect to the concentration of the protein is proportional to the actual colloid osmotic pressure, we have the differential equation
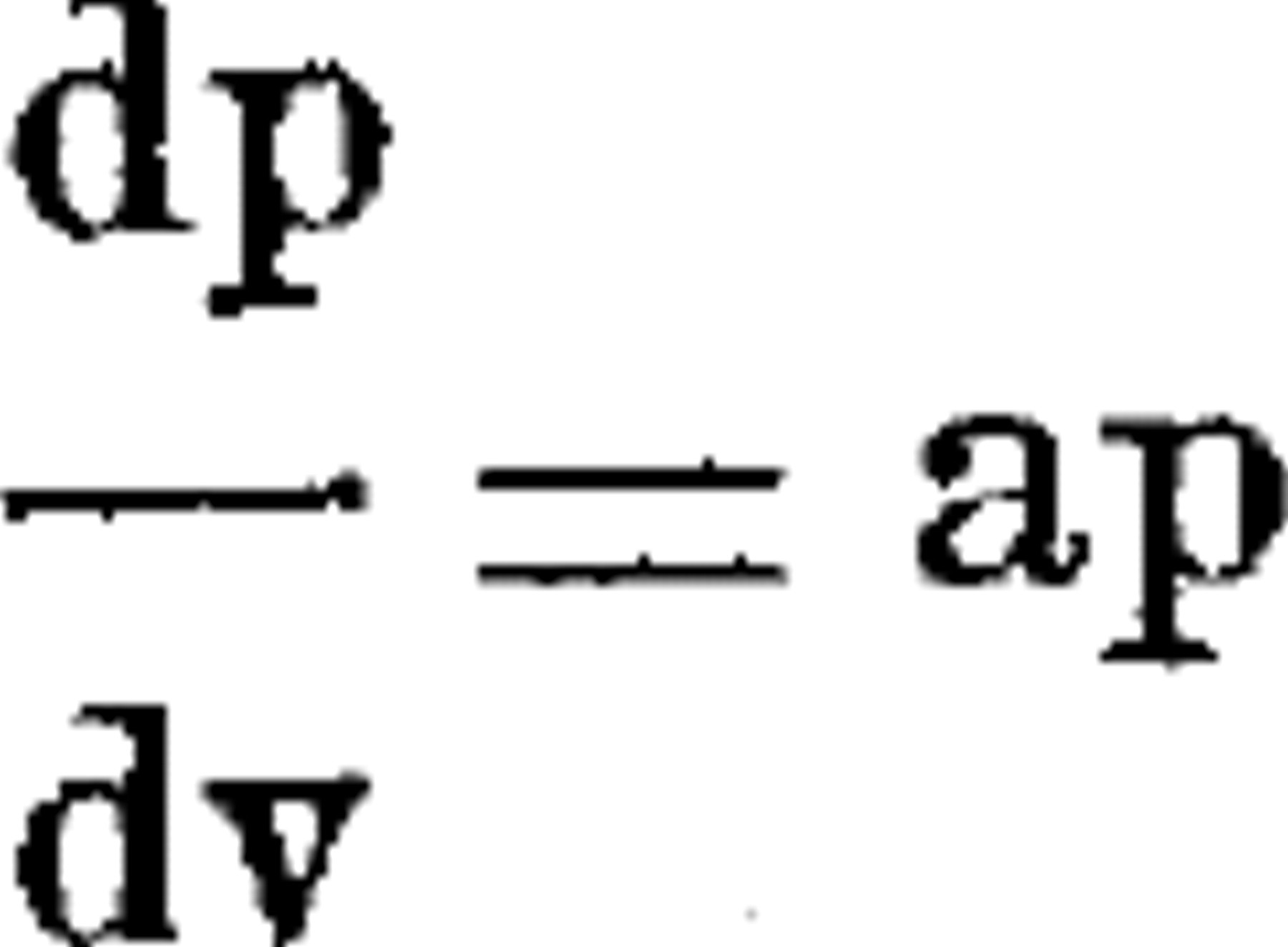
where p is the reciprocal of the colloid osmotic pressure and v is the reciprocal of the protein concentration.
Get full access to this article
View all access options for this article.
