Abstract
There have recently been two notable investigations of the physico-chemical mechanism of bone salt formation. Holt, La Mer and Chown 1 concluded that blood serum is normally supersaturated with Ca3(PO4)2 to the extent of more than 200%. Sendroy and Hastings 2 also made careful determinations of K'spCa3(PO4)2 and of the solubility of this salt in serum, and obtained similar experimental results. They interpreted the results differently however, and concluded regarding supersaturation that “the evidence in every direction seems to be strongly opposed to this point of view,” that “the data indicate that the theory of supersaturation is inadequate,” that “the theory of complete dissociation of electrolytes … seems to fail as an adequate explanation for the solubility of calcium in serum.” These investigations have thus demonstrated that the behavior of calcium and inorganic phosphorus in serum and in solutions is inexplicable when K'spCa3(PO4)2 is applied as the criterion for the precipitation of calcium phosphate.
When their data are studied from another viewpoint, more plausible explanations result. The empirical Ca × P product of Howland and Kramer
3
has proved so valuable that it seemed to us that it might have theoretical validity. This empirical product would have theoretical validity if the calcium phosphate were deposited as a result of a second order reaction such as Ca++ + HPO4” 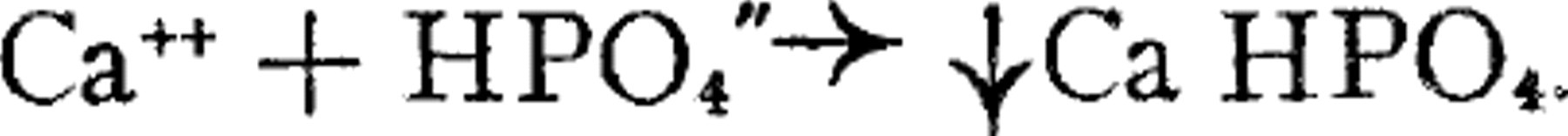 Ca HPO4.
Ca HPO4.
Get full access to this article
View all access options for this article.
