Abstract
Calcium metabolism in general is unquestionably connected with the solubility of Ca3(PO4)2 and this in turn with the ions of H3PO4 and Ca++ through the solubility product principle. This is particularly true for the deposition and absorption of bone as well as for the many related pathological conditions. Inasmuch as Ca3(PO4)2 cannot precipitate unless the ion product [Ca++]3X[PO4 —]2 exceeds the equilibrium value of this product (Ksp), it is necessary to evaluate Ksp under various conditions in order to determine the degree of saturation or undersaturation before proceeding to an investigation of the other factors involved in calcium metabolism. It appears from our studies that the Ksp of the tertiary phosphate of calcium is of more biological importance than the secondary phosphate CaHPO4. Data have been obtained on the latter and will be communicated later.
In a system containing orthophosphates, a knowledge of the hydrogen ion concentration and of the total phosphorus content makes it possible to calculate the concentration of (PO4) ion with reasonable accuracy, by using the formula:
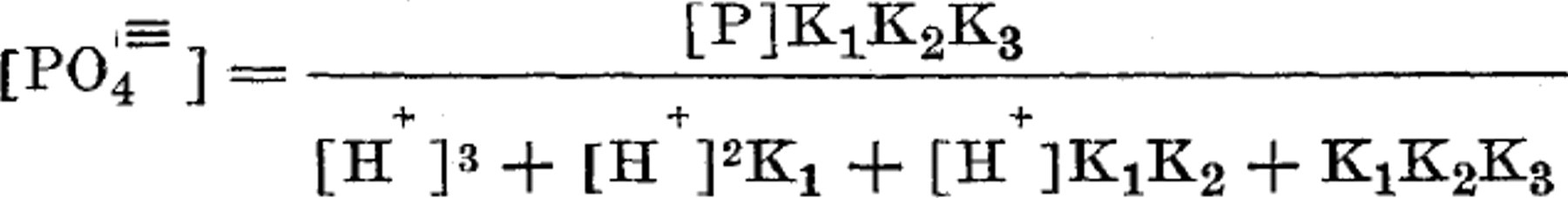
where [PI represents the molar concentration of phosphorus as phosphate, [H+]that of hydrogen ion, and K1, K2, and K3 the lst, Znd, and 3rd ionization constants of phosphoric acid, respectively. This formula is derived from the equations defining these three ionization constants :
which expresses the fact that the molar concentration of total phosphorus [PI is equal to the sum of the molar concentrations of the unionized and ionized forms of phosphoric acid.
A knowledge of the PO4 ion concentration and the Ca++ ion concentration enables one to calculate the value of the solubility product constant for tertiary calcium phosphate.
in systems which are in equilibriuni with solid Ca3(PO4)2. The system studied was prepared by titrating orthophosphoric acid with lime water, and determining the pH electronietrically after periods-extending in some cases to 8 months-had elapsed to insure equilibrium. Ca and P were determined for the solution; the composition of the solid phase was repeatedly'checked by chemical analyses of the precipitates in equilibrium with the solution.
Get full access to this article
View all access options for this article.
