Abstract
The recent investigations on the chemical structure of lecithin have resulted in many important contributions, all of which point to the correctness of the generally accepted view of its molecular structure, 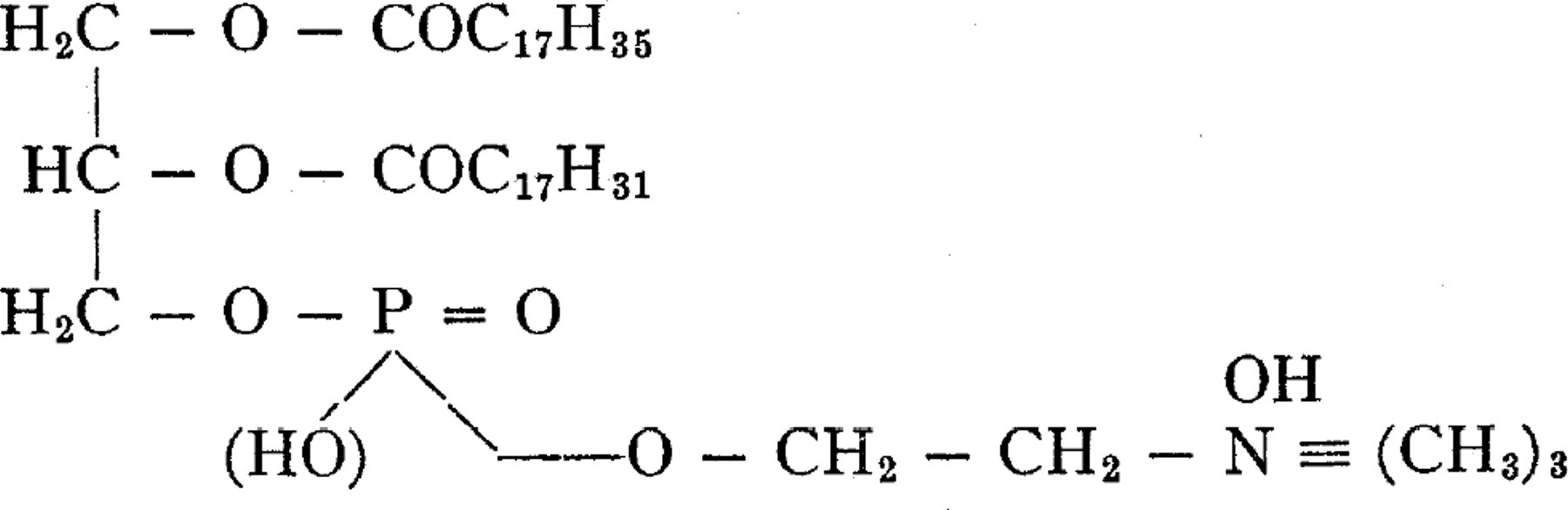
However, a scrutiny of all the work on lecithin reveals a remarkable incompleteness of each individual investigation. A rigorous proof of the accepted theory requires an elementary composition of C = 65.70, H = 10.79, N = 1.74, P = 3.86. It further requires the nitrogen of the molecule to be composed entirely of choline. Hence lecithin should not contain even a part of its nitrogen in the form of free amino groups.
The work up to the present has satisfied many of the requirements. The fatty acids and the glycerophosphoric acid have been identified; and MacLean has prepared, at least once, a sample of lecithin that was free of amino-nitrogen. However, this one sample was very incompletely analyzed. All other samples of lecithin prepared by various workers contained amino-nitrogen in their molecule, and from the standpoint of elementary analysis showed a marked disagreement with the theory. This is well illustrated by the following table :
The analytical data obtained by Ritter on hydrolecithin showed better agreement with the theory, and one might have been inclined to regard the material of Ritter as such that contained all the necessary and sufficient proof in favor of the conventional theory. Unfortunately, Ritter did not determine the aminonitrogen of the reduced lecithin, and hence failed to furnish definite proof of its purity.
Indeed, the present report contains data unmistakably proving that hydrolecithin of an elementary composition fully harmonizing with the theory may be and generally is impure, containing between 10 and 20 per cent. of its nitrogen in the form of aminonitrogen.
Get full access to this article
View all access options for this article.
