Abstract
The action of the generated ammonium carbonate in retarding the action of urease is due to the alkalinity of the carbonate. When the solution is kept neutral by a proper phosphate mixture the products have no effect on the velocity of the reaction. Elimination of the effect of the products makes urease a particularly favorable enzyme with which to study the reaction between enzyme and substrate. The results indicate that the action consists of two successive reactions: combination of enzyme and substrate in definite proportions; and decomposition of the compound, the urea being thrown off as ammonium carbonate; each of the two reactions consuming a definite portion of the total time. Formulation of these relations leads to the equation 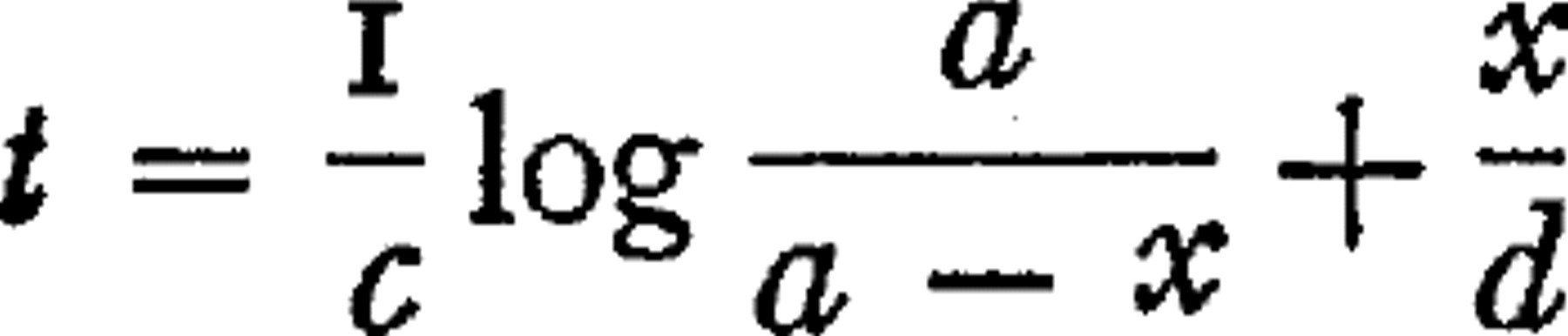 representing the time required for the decomposition of x amount of the initial substrate amount, a; c is a constant representing the velocity of combination of enzyme and substrate, d representing the velocity of decomposition of the complex. The values of c and d can be determined independently, and one can thereby determine whether changes in conditions affect the combination reaction, or that of decomposition. Neutral salts retard the combination. Alkaline reaction hastens it, but retards the decomposition. Slightly acid reaction greatly retards the combination, affecting the other reaction but little. The independent variation of the two phases of the process of enzyme action explains some previously obscure facts in regard to the effect of alkalies, acids, and other substances on enzyme action.
representing the time required for the decomposition of x amount of the initial substrate amount, a; c is a constant representing the velocity of combination of enzyme and substrate, d representing the velocity of decomposition of the complex. The values of c and d can be determined independently, and one can thereby determine whether changes in conditions affect the combination reaction, or that of decomposition. Neutral salts retard the combination. Alkaline reaction hastens it, but retards the decomposition. Slightly acid reaction greatly retards the combination, affecting the other reaction but little. The independent variation of the two phases of the process of enzyme action explains some previously obscure facts in regard to the effect of alkalies, acids, and other substances on enzyme action.
Get full access to this article
View all access options for this article.
