Abstract
There are at present four different views as to the nature of the absorption of oxygen by blood.
The first and oldest view is expressed by the reation, 1 mol. hemoglobin + 1 mol. oxygen 1 mol. oxyhemoglobin. According to the law of mass action one would have

in which a represents the concentration of hemoglobin in solution, b the oxygen concentration, c the concentration of oxyhemoglobin, and k a constant.
The second view is that of Bohr. Bohr assumed that hemoglobin in aqueous solution is hydrolytically split into globin + Fe-component, and that the Fe-component combines reversibly with oxygen. Bohr's formulation of this hypothesis leads to an equation of the fourth degree. Without going into details it will suffice to call attention to the main points of Bohr's work as far as we are concerned here. They are, in addition to the assumed hydrolysis of hemoglobin in aqueous solution, first, the inapplicability of formula (1) to his results and secondly, the variation of the ratio oxygen absorbed/total hemoglobin concentration with the total hemoglobin concentration 1 at constant pressure.
The third view is that of Wo. Ostwald who applied the older adsorption formula,
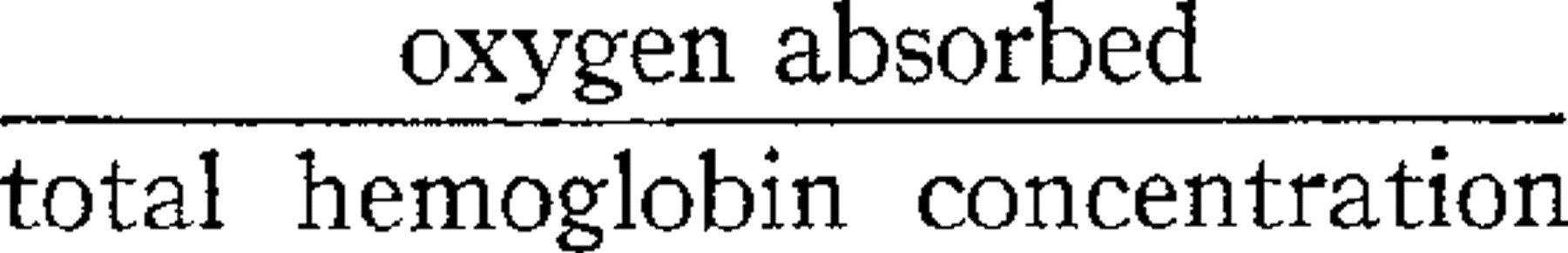
in which x = oxygen adsorbed, c = oxygen concentration, k and n are constants, to some of the data of Paul Bert and Loewy and found that log x and log c plotted in rectangular coördinates gave a straight line.
The fourth and last view is that brought forward by Manchot, who found that the quantity of oxygen or CO absorbed by 100 c.c. blood, laked or unlaked, varied with the dilution, i. e., with the total hemoglobin concentration. On 10-fold dilution this quantity approached a maximum value of 2 mol. oxygen to 1 mol. hemoglobin.
Get full access to this article
View all access options for this article.
