Abstract
Studies which have examined the effects of ankle-foot orthoses (AFOs) on children with cerebral palsy (CP) often report insufficient detail about the participants, devices and testing protocols. The aim of this systematic review was to evaluate the level and quality of detail reported about these factors in order to generate best practice guidelines for reporting of future studies. A systematic search of the literature was conducted to identify studies which examined any outcome measure relating to AFO use in children with CP. A customized checklist was developed for data extraction and quality assessment. There was substantial variability in the level and quality of detail reported across the 41-paper yield. Many papers reported insufficient detail to allow synthesis of outcomes across studies. The findings of this review have been used to generate guidelines for best practice of reporting for AFO intervention studies. It is important to ensure homogeneity of gait pattern in a subject sample or to subdivide a sample to investigate the possibility that heterogeneity affected results. It is also important to describe the orthosis in sufficient detail that the device can be accurately replicated because differences in designs have been shown to affect outcomes. These guidelines will help researchers provide more systematic and detailed reports and thereby permit future reviewers to more accurately assess both the reporting and quality of orthotic interventions, and will facilitate synthesis of literature to enhance the evidence base.
Introduction
Cerebral palsy (CP) describes a group of permanent disorders of the development of movement and posture, causing activity limitation. 1 The resulting lower limb muscle imbalances, deformities and gait abnormalities are often managed by ankle-foot orthoses (AFOs). The evidence-base for the use of AFOs in children with CP has been repeatedly described as low quality. 2–4 These conclusions and recommendations for improvement are largely based on general methodological construct and minimising study bias, which were assessed using tools such as Sackett's Level of Evidence 5 or the PEDro scale. 6
Writing recently in the British Medical Journal, however, Herbert and Bo have argued that equally important in ensuring high quality research is the quality of the intervention itself.
7
This conclusion is also implicit in the recent publication in the Annals of Internal Medicine of an extension of the CONSORT statement to trials of non-pharmocologic treatment,
8
and is evident in the work done by the Equator Network (
Whether or not the AFO intervention has been administered in a way which is reliable, valid and clinically relevant, will affect the confidence that can be had in the study's findings and the degree to which the results are considered generalizable. Assessment of intervention quality relies on sufficient detail and transparency in trial reports. 7 Several authors have noted variable depth and breadth of detail provided by studies within the body of orthotic literature. 2,3,9 While there are general guidelines for reporting of intervention detail in randomized and non-randomized trials (e.g., the CONSORT statement, 8,10,11 and the TREND statement 12 ), there are currently no guidelines recommending the specific detail that should be reported in AFO intervention studies.
This is thus the first review to focus specifically on the level and quality of detail reported by AFO intervention studies on children with CP, about the participant samples, AFO interventions and testing protocol. It was anticipated that by focussing on examples of good practice within the literature, it would be possible to derive best practice guidelines for reporting of research in this area. These guidelines will improve the detail reported by future work and thereby the quality of this evidence-base.
Methods
Search strategy
An electronic search of the literature was conducted by one reviewer (ER) in April 2009 using the following databases: MEDLINE (1966–April 2009), CINAHL (1982– April 2009), RECAL legacy database (1991–2009), EMBASE (1988–2009 week 17), AMED (1985–April 2009), INSPEC (1987–April 2009), ISI Web of Knowledge (sci-expanded, SSCI, A&HCI), Informit Databases (with the sub-selection of health, technology, science, engineering; 1998–2009), the Cochrane Database of Systematic Reviews (1991–April 2009) and the Physiotherapy Evidence Database (PEDro). Medline was also accessed using the internet (PubMed). Databases were searched from database inception with no a priori exclusions, restrictions or limits. Reference lists from the relevant identified papers were also searched manually.
A search using MESH terms and free text words was performed using the search terms related to “cerebral palsy”, “child”, “adolescent”, “orthosis”, “brace” and “AFO”. Relevant truncation or wildcard symbols were used to retrieve all possible suffix variations of a root word. An example of the search strategy: (1) “cerebral pals$”; (2) “child$” or “adolescen$”; (3) “orthos$” or “orthot$” or “brace” or “AFO”; (4) Combine 1 AND 2 AND 3.
Inclusion criteria
Studies were included if they evaluated any outcome measure relating to AFO use in children or adolescents (aged 6–18 years) who had a primary diagnosis of CP. Only full papers from peer reviewed journals which were published in English were eligible for inclusion. As only experimental work was of interest, systematic reviews were excluded as were Level IV and V studies (case studies or opinion pieces). 5
An AFO is defined as an external supportive device which encompasses the ankle joint and the whole or part of the foot. 13 This review limited inclusion to AFOs designed to control unwanted ankle movement by use of physical application of force through a three point pressure system. Therefore, studies of non-rigid AFOs such as elastic wrap or lace up ankle braces, Lycra garments and supramalleolar orthoses were excluded. Studies which examined any of these devices in addition to other AFOs which matched the inclusion criteria were included. Because the term “dynamic ankle foot orthosis (DAFO)” is used ambiguously, devices so named were initially included and later excluded if further examination indicated that the devices were supramalleolar orthoses.
Data extraction and quality assessment
The title and abstract of each study identified from the search was assessed by one reviewer (E. Ridgewell) for inclusion or exclusion from the review. All papers that were initially discarded were checked by the second reviewer (F. Dobson) to ensure no papers had been accidentally excluded. Any paper initially included which upon full reading did not meet the inclusion criteria, was later excluded.
A customized quality checklist was used to conduct a systematic assessment of evidence quality. As no standardized or validated quality checklists were available for this type of review, a new checklist was designed which combined data extraction and quality assessment (Table I). Past literature reviews and studies in this area 2,3,9,14 and in other areas of orthotic management, 15–17 ISPO consensus conference documents, 4,18 systematic review and checklist guidelines 6,19–24 as well as examples of customized quality checklists previously used in other systematic reviews, 25–27 were used to guide design.
Customized data extraction and quality checklist.
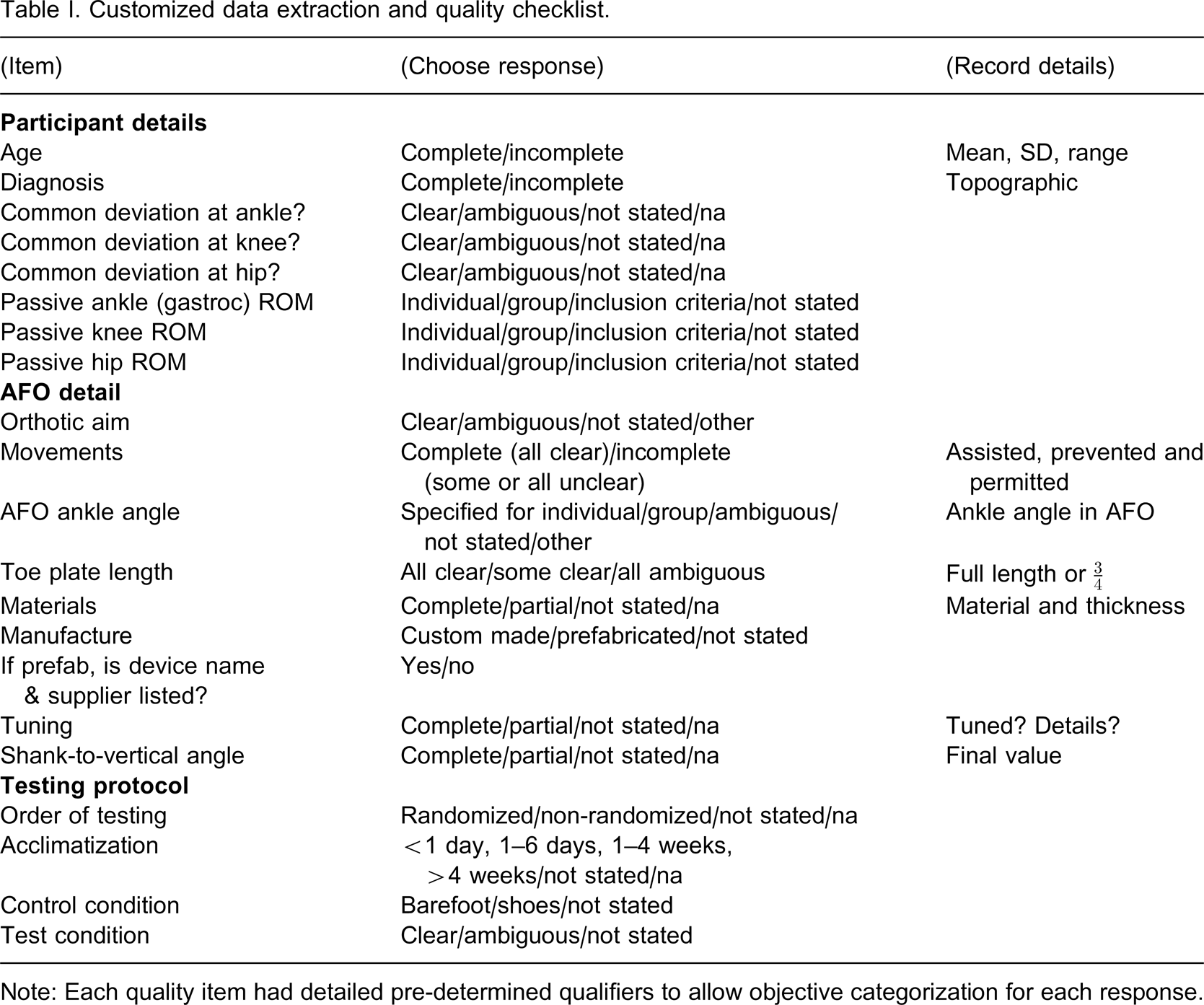
Note: Each quality item had detailed pre-determined qualifiers to allow objective categorization for each response.
There were three major themes to the checklist:
Participant details. This included reporting of topographical diagnosis, age, ankle, hip and knee passive joint range of motion (ROM) and the description of a common abnormality/indication within the group regarding a pattern of ankle, knee and hip movement.
AFO details. This included reporting of the orthotic aim, descriptions of movements permitted, prevented and assisted, AFO ankle angle, toe plate length, material type and thickness, AFO tuning, final AFO shank-to-vertical angle and whether the AFOs were described as custom made or prefabricated. The AFO shank-to-vertical angle is the angle between the lower leg and vertical while standing in the AFO. It is measured as the angle between the vertical and a line joining the knee joint centre and lateral malleolus in the sagittal plane.
28
Testing protocol. This included reporting of the control and test conditions, and details regarding randomization and acclimatization.
Both reviewers (E. Ridgewell; F. Dobson) piloted the data extraction and quality assessment form independently on a sub-group of papers to check the form content and reliability. Inter-rater agreement on full consensus items was 81%. The draft checklist was altered accordingly to provide improved clarity and instruction. Following independent review of all papers by both reviewers all non-consensus items were discussed until a consensus was reached. Any discrepancies were investigated using the original article to ascertain the correct response based on the objective a priori decision rules. Full consensus was reached on all items.
Results
The electronic search of selected databases identified 374 articles as having possible relevance to the use of AFOs in children with CP. Targeted searching of relevant on-line journals revealed 55 articles which were all later discarded as duplicates. After applying the inclusion criteria, 41 full papers were included in the review.
Demographics
Table II outlines the study details and demographics including participant diagnosis, number of participants, intervention, control, study design and primary activity/outcome examined. The total yield was published across 17 journals. A total of 1201 children with CP were examined over a period of 38 years with the majority of articles published after 1995. Eleven different devices or device designs were examined. The majority of studies examined gait while walking on a level surface. More studies examined diplegic children than hemiplegic children.
Demographic and descriptive aspects of all studies.
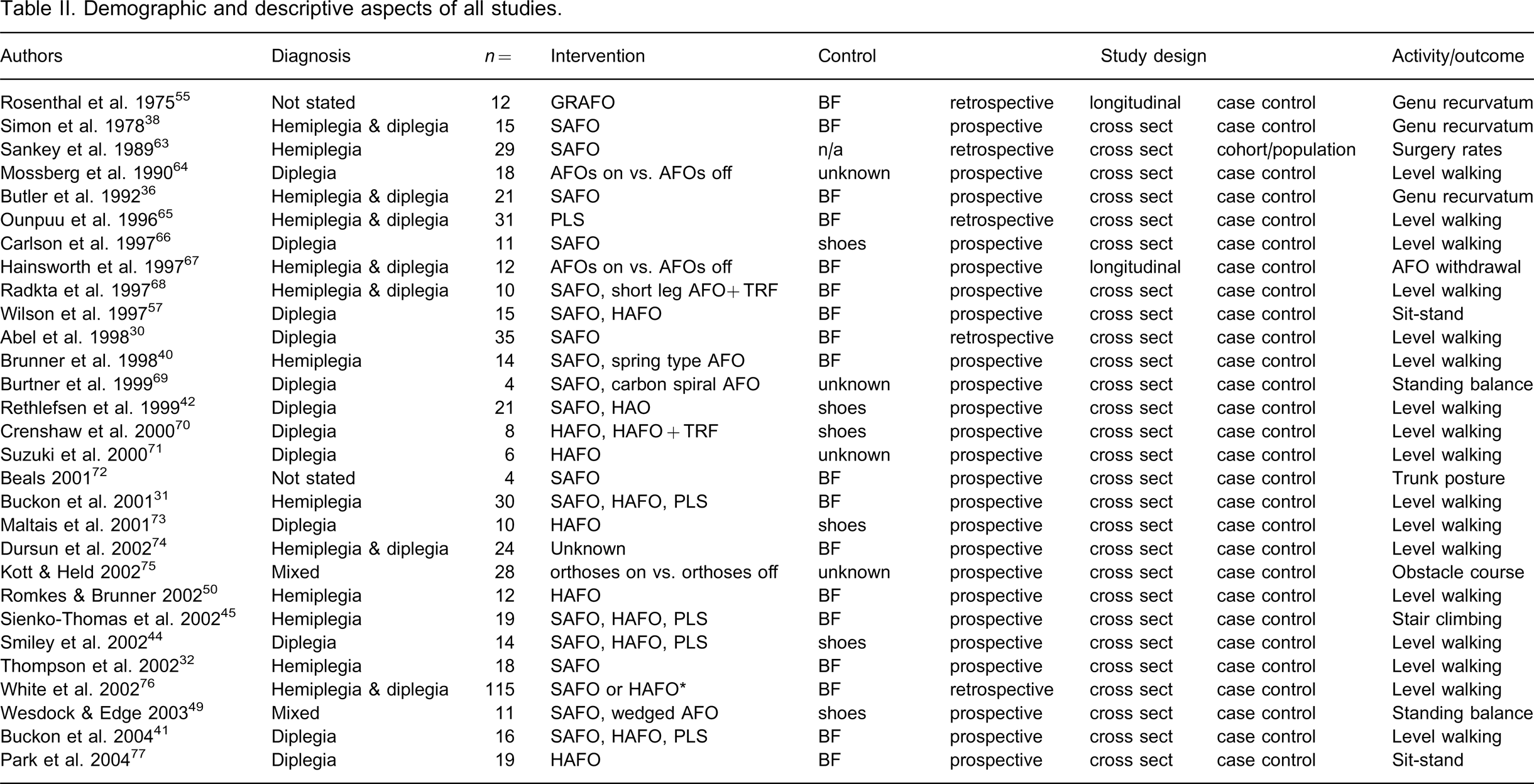
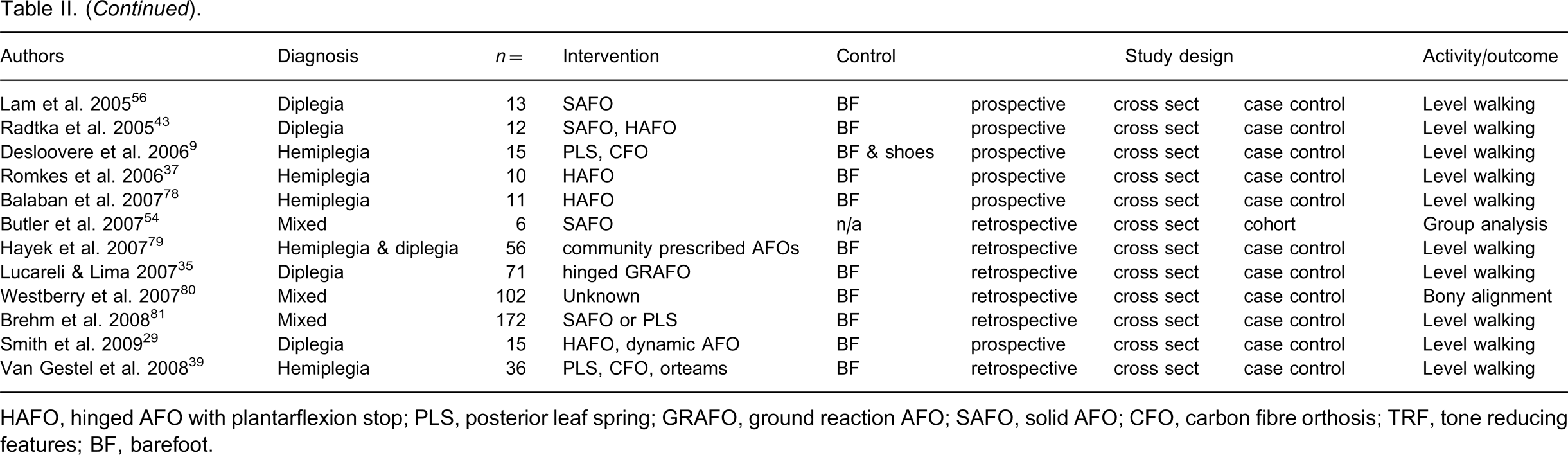
HAFO, hinged AFO with plantarflexion stop; PLS, posterior leaf spring; GRAFO, ground reaction AFO; SAFO, solid AFO; CFO, carbon fibre orthosis; TRF, tone reducing features; BF, barefoot.
Participant details
Table III provides a summary of the data extraction and quality outcomes across all studies. Table IV provides an overview of the responses of all studies to each item on the data extraction and quality assessment checklist.
Results of data extraction and quality outcomes for all studies.
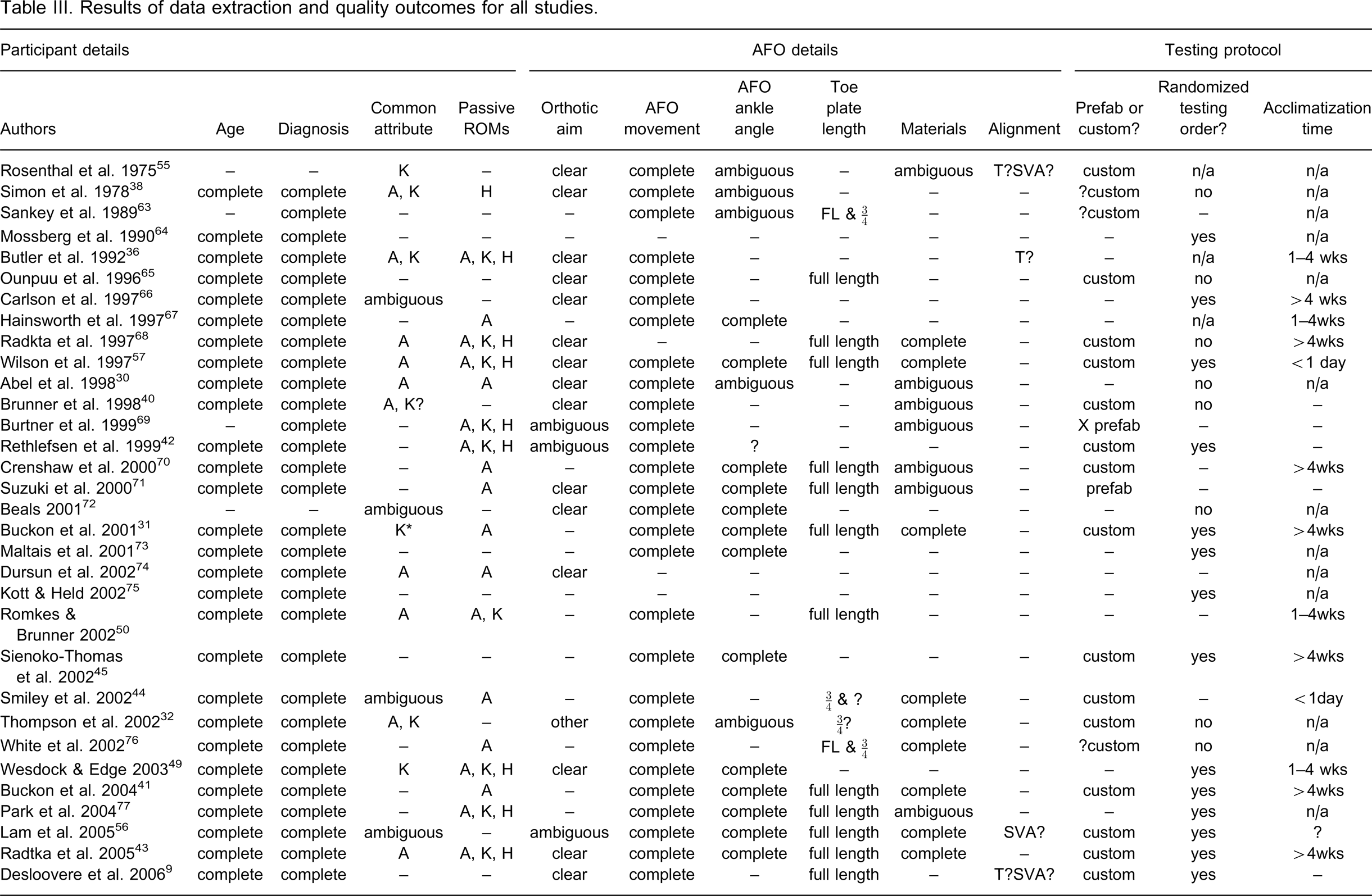
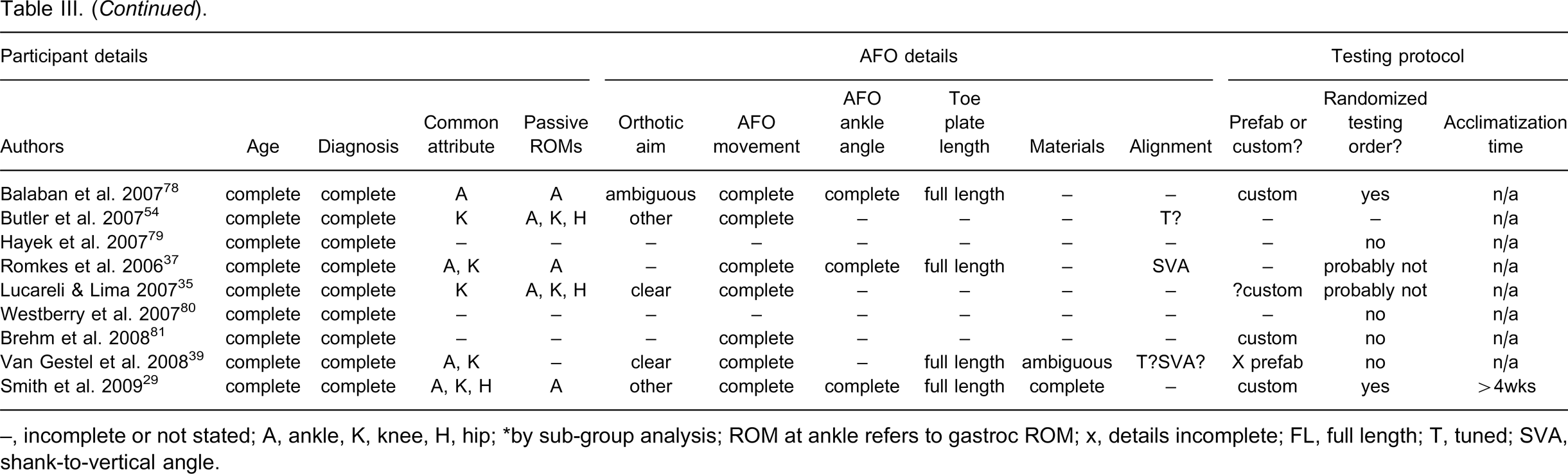
–, incomplete or not stated; A, ankle, K, knee, H, hip; ∗by sub-group analysis; ROM at ankle refers to gastroc ROM; x, details incomplete; FL, full length; T, tuned; SVA, shank-to-vertical angle.
Summary of responses to each quality item.

All unlisted papers were categorized as either ambiguous or not stated.
Topographical diagnosis and participant age were well reported. More studies provided information on passive ankle ROM (23/41) than knee (11/41) and hip ROM (10/41). Nineteen of the 41 studies made a clear attempt at describing a common pattern of abnormality or indication that was demonstrated by all participants. One study 29 clearly described a homogenous gait characteristic demonstrated by all participants. A homogenous mode of ankle movement was described most frequently (14/41), followed by knee movement (10/41) and three papers described a common pattern of hip movement.
AFO detail
Seventeen studies clearly stated the aim of the AFO intervention. A clear description of the type of AFO intervention was provided by most studies (35/41), as were clear descriptions of the movements prevented, assisted or permitted by the AFO. Sixteen studies clearly stated the AFO ankle angle. Nineteen studies gave clear descriptions of toe plate length for all devices tested. In some cases it was necessary to infer this information from photographs or diagrams. A full-length toe plate was used more often (18/41) than ¾-length toe plate (4/41). Ten papers provided complete detail on both material type and thickness, and eight more papers provided partial information.
Five studies reported that the AFOs were tuned prior to testing. One study provided the final shank-to-vertical angle of the AFOs tested and two provided partial information. Custom made devices were most commonly tested (18/41). Three studies tested prefabricated devices with the name of the device and supplier provided by one study. The remaining 20 studies did not state whether the device was custom made or prefabricated.
Testing protocol
More studies used a randomized order of testing (16/41) than non-random (12/41). The remaining studies either did not report this information or the item was not applicable. Twenty studies tested unfamiliar devices. Across these studies the acclimatization times ranged from less than one day (2/20), 1–4 weeks (4/20) or greater than four weeks (8/20). Most studies clearly stated the control condition with barefoot being the most common (28/41), followed by shoes (6/41) and both barefoot and shoes (1/41). The test condition was clearly stated in most studies.
Discussion
This review identified 41 full papers which examined the effect of AFO use on a diverse range of outcome measures in children with CP. In line with the conclusions of previous reviewers, 2,3 there was considerable variety in the level and quality of detail reported. In many cases this limits any assessment of intervention quality and the impact this may have on confidence in the findings. This variability also reduces the potential for meta-analyses to summarize results across studies to provide more substantial evidence of treatment practices. Incomplete reporting of data further reduces the potential to combine results across studies.
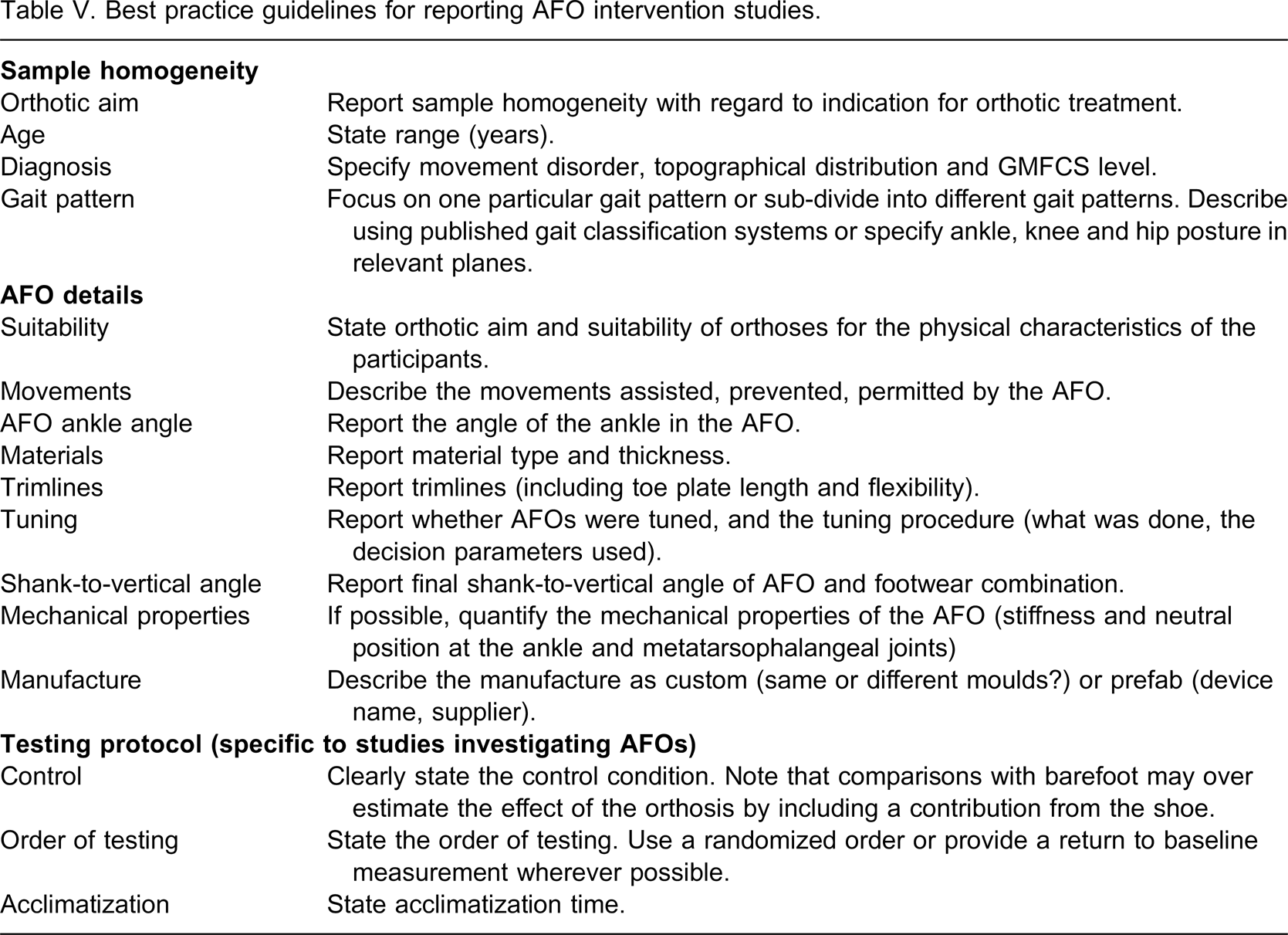
There were, however, sufficient examples of good quality interventions to enable best practice guidelines for future studies to be derived. These are discussed in full below and the recommendations for reporting of detail for AFO intervention studies are summarized in Table V.
Best practice guidelines for reporting AFO intervention studies.
Participant details
In a number of studies in which participants were subdivided according to gait pattern, differences in outcomes between the sub-divisions were observed. Abel and colleagues 30 found differences in the effect of a solid AFO on the gait of children with either equinus or plano-valgus feet. 30 Buckon and colleagues 31 found that normalization of knee motion was dependent upon the position of the knee during stance while barefoot, as well as the type of AFO. Thompson and colleagues 32 observed that responses varied in hemiplegic children with different Winters, Gage and Hicks (WGH) 33 gait classifications. Significant changes were found for the groups with Type I and Type II gait patterns, but not Type III.
Given these differences in outcomes according to the type of AFO tested and underlying gait pathology, it is clear that studies should focus on participant groups which exhibit some degree of homogeneity with regard to gait pattern, or should sub-divide groups to investigate the possibility that heterogeneity affected the results. At a minimum, this sub-division could include participant age, nature of the movement disorder, topographical distribution and GMFCS 34 level, but ideally would include a description according to a gait classification system. In a study of the effects of AFOs, it could be argued that homogeneity in the orthotic aim, the basis of which is largely dependent on gait pattern, is equally important.
There are good examples of studies which focused either on participants with a particular gait pattern 29,30 or sub-divided participants on the basis of gait pattern. 31,32,35 Several other studies ensured homogeneity by describing knee and ankle posture either with 29,36,37,39 or without 38 reference to published gait classification systems.
AFO details
Clearly reporting the design of the AFO is essential as differences between AFO designs have been shown to produce difference in outcomes in temporospatial parameters, 40–42 ankle kinematics 9,31,41–44 and knee kinematics 31 in straight line walking, as well as stair ascent and descent. 45 No studies have examined the effect of different AFO toe-plate lengths on children with CP, however one study of post-stroke adults 46 found differences in stance phase dorsiflexion as a result of toe-plate length. Variation in the material properties used in construction of the AFO may also influence the flexibility of so called “rigid” devices at the ankle and metatarsophalangeal joints. 47
The alignment of the AFO alone is described by the AFO ankle angle, the choice of which is based on clinical measures such as passive and dynamic gastrocnemius muscle length and tri-planar stability of the foot. Severe spasticity or contracture of this bi-articular muscle must be accommodated in the AFO ankle angle to avoid limiting maximum knee extension or compromising the tri-planar stability of the foot. 48
Reporting passive gastrocnemius length in addition to the AFO ankle angle permits the appropriate choice of ankle angle to be confirmed. It could be argued that reporting the evidence for the choice of AFO ankle angle (e.g., passive gastrocnemius length) is unnecessary. However, there were two studies 37,49 in which reported data suggested that the choice of AFO ankle angle did not consider passive gastrocnemius length. Similarly, three studies which evaluated the effects of free dorsiflexion AFOs on children who had or may have had limited gastrocnemius length. 37,40,50 In such cases, dorsiflexion of the ankle will only occur at the expense of knee extension or compromised foot position. Similar findings have been identified in the stroke literature, 17 which suggests that reporting this information may be a reasonable proposition.
When the AFO is combined with footwear, the AFO ankle angle may no longer describe the alignment of the device relative to vertical if there is a difference in height between the heel and forefoot of the footwear (heel-sole differential). In order to overcome this limitation, the alignment of the AFO and footwear is described by the shank-to-vertical angle. Differences in footwear heel-sole differential and therefore shank-to-vertical angle have been demonstrated to affect the alignment of the ground reaction force during quiet standing 52 , with suggestions that SVA can be modified to improve GRF orientation during walking 17,28,36,48,53,54 One study 37 reported the final shank-to-vertical angle of the AFO and footwear used, which was a standardized alignment for all participants. There is however some evidence suggesting that AFOs require individual adjustment, or tuning of the shank-to-vertical angle in order to obtain optimal function. 53 While the concept of tuning is not new, it has only recently become more widely recognized which may explain why only five papers 9,36,39,54,55 reported tuning of the AFO interventions.
Most studies in this review clearly reported the movements controlled by the AFO. There are several good examples of studies which have provided excellent descriptions of the physical characteristics of the AFO interventions. 29,31,41,43,56,57 Such descriptions do not, however, account for the differences in mechanical properties arising from small variations in AFO design, such as trimline position and choice of materials.e.g., 58–60 A new method of measuring the stiffness and neutral angle around the ankle and metatarsophalangeal joints has recently been described and demonstrated as reliable and clinically applicable. 58 Including such objective measurements in future clinical and research practice will improve our ability to compare AFO interventions.
To enable the quality of the AFO intervention to be more accurately assessed, future work should describe the movements prevented, assisted and permitted by the AFO, toe plate length and flexibility, trim-line position, materials and method of manufacture, AFO ankle angle, shank-to-vertical angle of the combined AFO and footwear, type of footwear worn and details of any tuning process undertaken. Testing of mechanical stiffness of the AFO and the combined AFO and footwear would further enhance objectivity. Transparent reporting permits replication of the study, and makes it possible to understand the variables that may affect intervention outcomes.
Testing protocol
All studies with the exception of one used either barefoot or shoes as the control comparison. This study 9 included both barefoot and shod conditions and found that shoes alone could have either a negative or positive effect on gait, thereby confirming findings from the stroke literature. 61 Future work should consider including both of these control conditions wherever possible to avoid attributing the effects of footwear to the AFO.
A randomized order of testing is desirable as it eliminates bias resulting from the order of testing. 21 This is particularly important in orthotic research as there are usually two or more conditions being compared over repeated trials of tasks such as walking. Use of a non-randomized order of testing introduces the risk of fatigue in the tasks performed last. Several studies used a non-randomized order of testing, which is often unavoidable in cases of retrospective analysis. Fifteen studies eliminated potential confounding series effects by randomising the testing order. A randomized order of testing should be used wherever possible, and in any event, the order of testing should be reported.
Acclimatization time to an unfamiliar device permits the novel nature of the device to be incorporated into the movement pattern thereby ensuring that the effects of the device accurately represent daily use. Many papers examined devices other than the device already worn by the participant. Most testing protocols permitted more than one week of acclimatization time. Only two studies permitted less than one day. 44,57
Future research
Previous reviewers have suggested that the quality of this body of literature could be improved by focussing on the execution of large scale RCTs 2 or on alternate study designs which overcome some of the difficulties of RCTs, for example cross-over and one-group interrupted time-series (single subject) designs. 3 However, equally important is a more systematic and detailed approach to reporting the participant sample, the AFO intervention and testing protocol. Use of the reporting guidelines presented in Table V will enable consistent reporting and will also permit a transparent assessment of study quality thereby improving the potential to combine results of several smaller studies using meta-analyses.
This review has identified several avenues of research which could benefit from focussed attention. For example, what is the most appropriate control condition for comparison with an AFO intervention? What is the minimum acclimatization required for an unfamiliar device? Do small differences in AFO design, stiffness and alignment have a significant effect on AFO effectiveness? Answering these questions might facilitate comparison across studies already published.
These reporting guidelines are in line with suggestions arising from the recent International Society for Prosthetics and Orthotics (ISPO) consensus conferences on the orthotic management of on CP 62 and stroke. 18 The principles on which these guidelines are based may also be applied to AFO research on other populations. However, certain elements may be more relevant in CP research due to the heterogeneity seen particularly in gait patterns.
Limitations
This review focused on an assessment of reporting detail and transparency regarding the participants, AFO intervention and testing protocol. An analysis of appropriate choice of outcome measures was not included as this requires decisions about which research questions are most important. Considering the type of outcome measures employed in each study is, without doubt, essential to future metanalyses as these are only possible between studies which have examined the same outcome measures.
This review did not rank or assign quality scores to studies; rather it focussed on examples of best practice with regard to different aspects of intervention quality specific to research into the effects of AFOs for children with CP. Issues relating to effect size, power or choice of statistical analysis are well described in general literature on research methodology.
Conclusion
Assessing the quality of individual studies and using studies in quantitative research synthesis requires transparent reporting. 12 While there was considerable variety in level and quality of detail provided by these studies, there were sufficient good examples of reporting detail and intervention quality. This enabled the generation of guidelines for reporting of detail in AFO intervention studies. These guidelines should also direct the design of future investigations in this area which will improve the synthesis of quantitative research and therefore the quality of this evidence-base.
Footnotes
Acknowledgements
