Abstract
Bioimpedance analysis was used to measure conductive tissue extracellular fluid (ECF) volume changes in the residual limbs of four unilateral transtibial amputee subjects during standing and walking conditions. Results showed that all residual limbs experienced ECF volume decreases during 5 min standing intervals. During 5 min of walking immediately after a standing interval, the residual limbs of healthy subjects increased in ECF volume while those of diseased subjects, one with peripheral vascular disease and another with cardiovascular insufficiency and a high-salt intake, decreased. One subject demonstrated less absolute value ECF volume change during standing and walking at 12 months post-surgical revision compared with at six months. Presentation of bioimpedance data to patients improved compliance to practitioner recommendations and patient understanding. Results were useful towards clinical assessment, patient education, and decision-making about treatment.
Introduction
Diurnal residual limb volume change is an important clinical challenge in lower-limb prosthetics. Volume reduction over time induces a loose socket fit. If no accommodation is performed, the result is concentrated stresses in soft tissue, particularly at bony prominences. Volume expansion can increase tissue pressures in the residual limb and cause blood flow occlusion, restricting venous return and causing a build-up of cell waste products in the limb. Both conditions can lead to soft tissue injury.
Residual limb volume fluctuations can be challenging to manage. Diurnal volume change is often difficult to predict, and it can be hard for amputees to decide how many socks to add or remove or what other compensations to perform. Additionally, patients often have difficulty understanding how their behaviour (e.g., diet, activity, and smoking) affects limb volume fluctuation. Establishing when a residual limb has achieved a stable limb size after amputation surgery so as to fabricate a definitive socket is another important clinical issue.
We have shown bioimpedance analysis to have sufficient sensitivity in prosthetics for the measurement of residual limb conductive tissue extracellular fluid (ECF) volume change. 1 Bioimpedance is a non-invasive means for assessing ECF volume and intracellular fluid volume within living tissue. It is the ECF that is the primary source of limb volume fluctuation in amputee prosthesis-users. 2 ECF includes blood and interstitial fluid. Interstitial fluid is plasma that enters and leaves the interstitial space. Bioimpedance analysis has been used as a tool principally for body composition/body fat analysis 3–8 and in the assessment of fluid imbalance in haemodialysis patients. 9–12
Bioimpedance analysis functions by measuring biological conductivity. Biological conductivity in the residual limb occurs primarily through fat-free tissues and fluids 13 as opposed to bone or adipose tissue. It is changes in the fat-free tissues and fluids, particularly the ECF, that cause amputee diurnal socket fit problems. To conduct a bioimpedance test, a very low electrical current (<700 μA) is injected into the residual limb through two outer pair electrodes, while voltage potential is measured across two inner pair electrodes.
Current is injected over a range of frequencies (between 5 kHz and 1 MHz) each second. The lower frequencies tend to travel through the ECF since cell membranes are not well-penetrated by low frequency signals. At high frequencies, however, current travels through both the ECF and intracellular fluid. Using data at different frequencies in a modelling strategy known as Cole modelling,
14
we can calculate ECF resistance. Tissue is modelled as an equivalent electrical circuit consisting of an extracellular resistance (RECF
) in parallel with an intracellular resistance and cell membrane capacitance. Nonlinear weighted least-squares curve-fitting applied to the multifrequency impedance spectrum is used to extrapolate RECF
and total resistance at the low and high-frequency limits. Resistance data is then converted to volume data through modelling based on the principle of volume conduction:
4,15
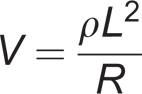
where V is limb fluid volume, R is the fluid resistance, L is the limb segment length, and ρ is the specific resistivity of the biofluid. For segmental bioimpedance analysis (assessment within a limb segment, like the residual limb), mixture theory can be used to account for the presence of nonconducting elements within the limb:
16–18
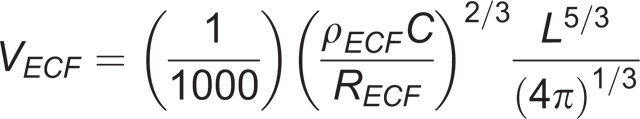
where the limb segment is assumed to be a cylinder with an average circumference C and length L.
Bioimpedance measurement is potentially attractive for clinical prosthetics use in that it provides a very sensitive quantitative assessment of ECF volume change, measuring volume alterations as small as those over the course of a 5-min standing interval. The purpose of this paper is to present four clinical cases where residual limb ECF volume change measurement using bioimpedance analysis was useful towards clinical assessment. We present these cases and demonstrate how ECF volume change was assessed, and discuss how the findings were useful in treatment.
Methods
Subjects
All four subjects were individuals with unilateral transtibial amputation who had their amputations at least six months prior and could walk on a treadmill without assistive devices for 5-min intervals. A human subject application was approved by an institutional review board, and informed consent was obtained before any study procedures were initiated.
Instrumentation
A commercial bioimpedance analyzer that measured impedances at 50 frequencies between 5 kHz and 1 MHz each second was used (Hydra 4200, Xitron Technologies, San Diego, California). Four strip electrodes (77 × 20 mm contact surface, 0.81 mm thickness) were implemented where the outer two electrodes delivered current (<700 μA) and the inner two electrodes sensed voltage (Figure 1). This is a standard configuration used for bioimpedance analysis. 19 Much care was taken to prepare the electrodes for measurement. Thin multi-stranded wires were soldered to the electrode tabs and strain-relieved at their attachments. A custom connector was designed to attach the electrode wires to the instrument cable to ensure a stable attachment.
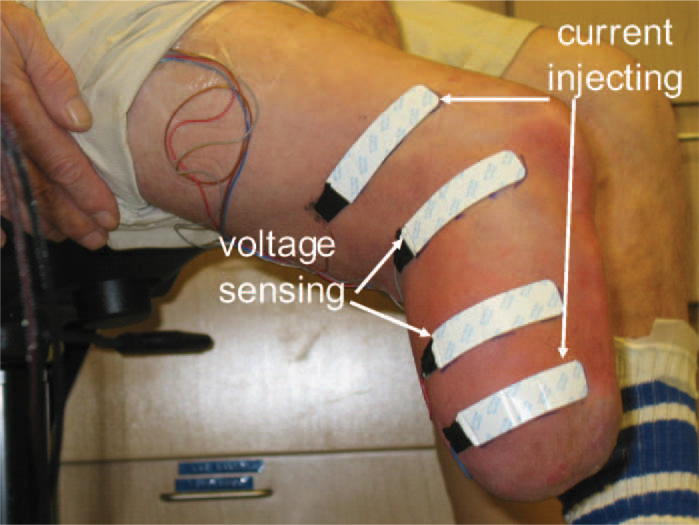
Subject ready for bioimpedance analysis testing. Current injecting and voltage sensing electrodes were positioned on the residual limb and the attached wires strain relieved.
Electrodes were positioned on the lateral and posterior aspects of the residual limb. These locations typically cover thick soft tissue regions of the residual limb. The proximal voltage sensing electrode was at the level of the patellar tendon, proximal of the fibular head. The distance between the voltage sensing electrodes was 7.0–9.0 cm depending on the limb length. The distal current injecting electrode was at least 3.0 cm distal to the distal voltage sensing electrode and was positioned on the relatively cylindrical portion of the residual limb. A spacing of at least 3.0 cm was used to enhance the likelihood of well-distributed current and thus measurement through the entire cross-section. These distances are shorter than those suggested in the Xitron Hydra 4200 instrument manual (≥10.0 cm voltage/voltage electrode spacing, and ≥5.0 cm voltage/current electrode spacing). However, since it was the change in ECF volume that was of interest in this research, as opposed to absolute ECF volume measurements, and since our data well fitted the models, our parameter thresholds were considered acceptable for this application. The proximal current injecting electrode was at least 6.5 cm proximal to the proximal voltage sensing electrode. Custom Matlab (Mathworks, Natick, Massachusetts, USA) code was written on a laptop (D620 Latitude, Dell, Round Rock, Texas, USA) to display bioimpedance data from the instrument in approximately real time (3 s delay). Our algorithm was similar to that described by De Lorenzo. 16 However, our error criteria were slightly relaxed so that the algorithm ran faster, albeit with slightly less accuracy. Since the visualization data were used only to ascertain performance of the instrumentation and to identify gross changes in ECF volume, the algorithm's performance was considered acceptable. A different algorithm 20 was used for post-processing the data for presentation here.
To determine if our bioimpedance measurement technique produced results on a non-diseased limb consistent with volume change measurements on normals described in the literature 21–23 bioimpedance measurements were taken from the contralateral limb of a healthy unilateral amputee subject (Case 1 below). These data were collected on a different day from that of residual limb bioimpedance measurement. The protocol was the same as that described below for residual limb testing, excluding the socket donning and doffing portions.
Data collection protocol
The data collection protocol involved intervals of sitting, standing, and walking. After the subject's mass was measured and his or her self-selected walking speed determined, the electrode locations were prepared by gently rubbing the skin with sandpaper. Sandpaper was used for cleaning rather than a cleaning agent since it does not dehydrate the skin. 19 A thin layer of coupling gel (Couplant D, Panametrics, NDT, Waltham, Massachusetts, USA) was applied to the undersurface of the electrodes to facilitate electrical coupling. Electrode locations were determined, and reference marks were made on the skin. The electrodes were positioned on the residual limb. Data collection was initiated at a 1 Hz sampling rate. After a stable limb volume signal was demonstrated the subject donned the prosthesis. He/she then sat quietly for 2 min. ECF volume measured at the end of the 2-min interval was used as a reference. The subject then stepped onto a 6.4 cm high platform with an electronic scale (349KLX Health-O-Meter, Pelstar, Bridgeview, Illinois, USA) embedded in the surface so that weight on the instrumented limb was measured. The subject was asked to maintain equal weight bearing. Weight was monitored continuously at a 1 Hz sampling rate. The subject was instructed to shift his/her weight appropriately if a change of more than 10% of half the subject's body weight occurred. After a 5-min standing interval, the subject stepped onto a treadmill (Clubtrack, Quinton Instrument Company, Bothell, Washington, USA) and walked at his or her self-selected walking speed for a 5-min interval. The subject then sat and rested for 2 min. The stand and walk portions were then repeated again. After the second walk period, the subject sat and doffed the prosthesis, liner, and socks.
Data processing
Collected bioimpedance data were post processed using software provided by the manufacturer (v2.2, Xitron). The algorithm used a non-linear least square error of both magnitude and phase where the weighting was established by the published accuracy specifications of the Xitron instrument. A spectral frequency was removed if its inclusion in the analysis significantly decreased the total weighted least square error, with limitations enforced so as to prevent the programme from deleting frequencies solely to force fit the Cole model. 20 Less than 4% of the data collected in this study were rejected. The data were converted to ECF volume using a well accepted model, 18 and then expressed as a percentage of the ECF volume measured after the initial 2 min sit interval with the prosthesis donned. It is important to note that only ECF volume change (i.e., not including bone and adipose tissue) was characterized in this analysis. Thus the data are presented as a percentage of ECF volume, not as a percentage of the entire residual limb volume.
To facilitate inspection of the data, a 10-point moving mean of the percent change in ECF volume was plotted. Since there was a time delay caused by the moving average filter, this curve was moved back 10 points so as to realign it with the original data. This curve was particularly useful for analysis of the walking segments of the trials. Those sections had a lot of fluctuation because of the change in ECF volume over the course of each step, high during swing and low during stance. Because the sampling rate was less than the walking frequency, it was not possible to measure the ECF volume change during each step. However, in this analysis it was the change over the course of the walking interval that was of interest. Thus use of the moving mean curve was appropriate. In the results presented below, we present only the moving mean curve, not the raw data, so as to simplify visualization of the data.
Results
Instrumentation evaluation
Data collected from the intact contralateral limb of the healthy subject presented as Case 1 below demonstrated an ECF volume increase during standing (average of 0.6%) and a decrease during subsequent walking (average of 0.7%) (Figure 2).
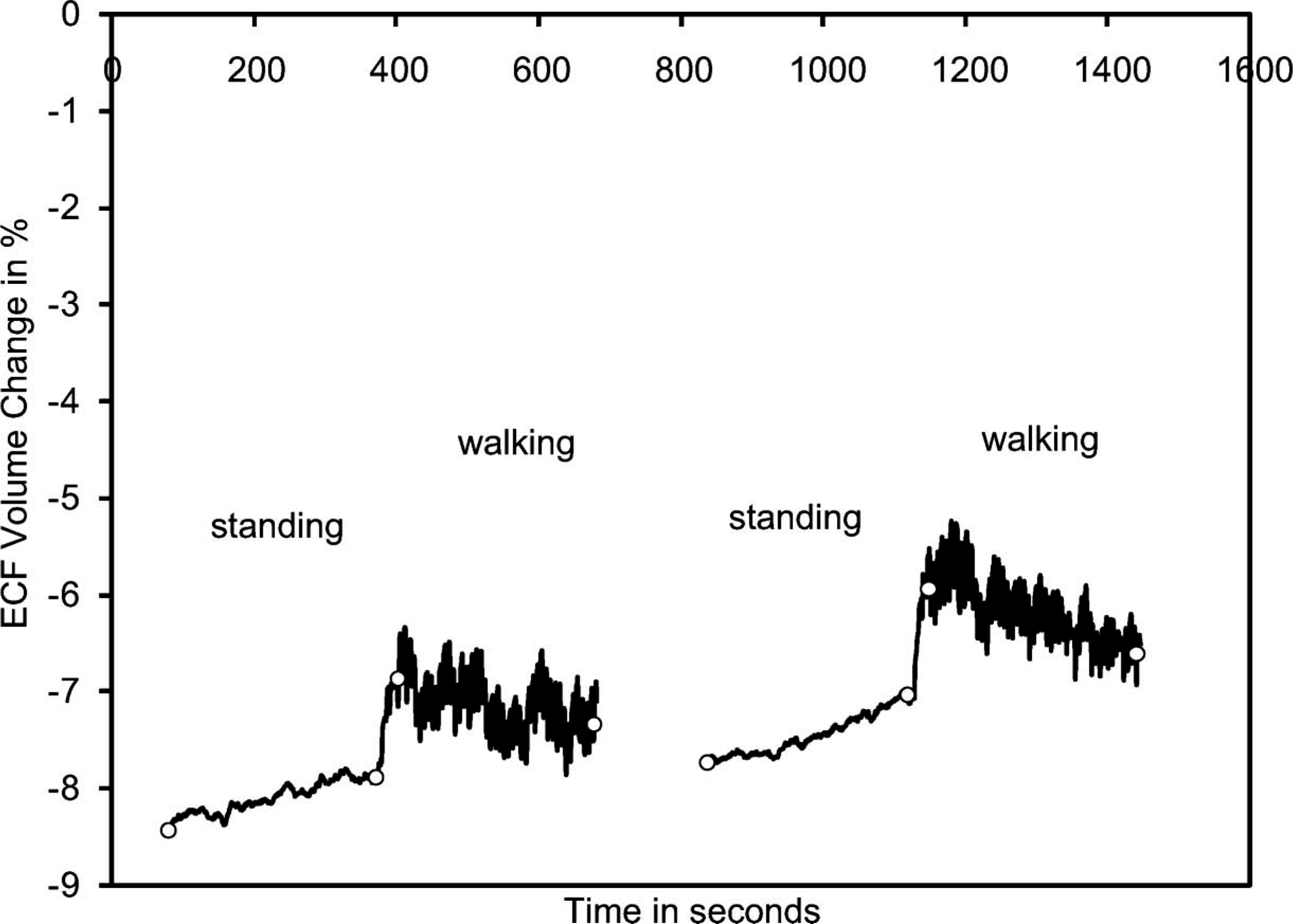
Instrumentation evaluation: Results from the intact limb of a healthy subject (Case 1). For clarity, only data from the standing and walking segments are shown. The beginnings and endings of the standing and walking segments are marked with open circles.
Case 1
This was a healthy subject with a comfortable prosthesis and no reported residual limb problems. His case is presented so as to provide a base for comparison with the other three subjects (Cases 2, 3, and 4) described below.
This male subject was 60 years of age and had his amputation four years prior due to traumatic injury. He was 72.7 kg in mass and 175 cm in height, and had no abnormalities other than his unilateral amputation. He used a transtibial socket with an elastomeric liner with a locking pin. A K-4 level ambulator, using K-level definitions from the traditional clinical characterization of Medicare Functional Classification Levels, 24 this subject regularly walked and played golf and racquet sports. He did not add socks or perform any prosthesis modification over the course of the day to accommodate limb volume reduction except under extreme physical exertion. In that condition he would add one sock.
The subject's residual limb demonstrated an ECF volume decrease during standing and increase during subsequent walking (Figure 3). Results from the first stand/walk segment were similar to those from the second stand/walk segment. The ECF volume decrease during the standing intervals averaged 1.3%. ECF volume increase during the walking intervals averaged 0.9%.
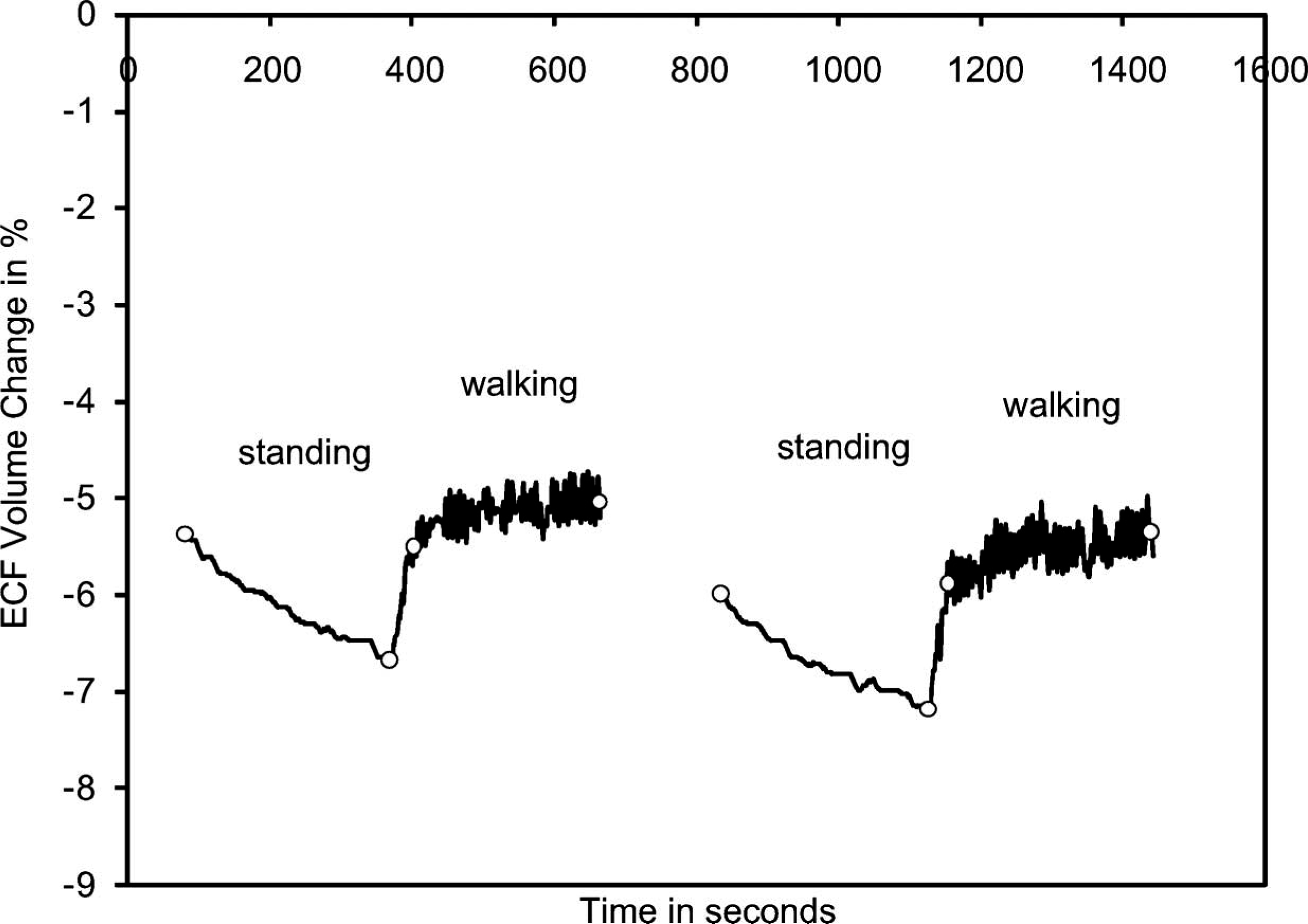
Results from the residual limb of a healthy subject (Case 1). For clarity, only data from the standing and walking segments are shown. The beginnings and endings of the standing and walking segments are marked with open circles.
Case 2
This was a 69-year-old male who had been an amputee for 23 years. He was 105.0 kg in mass and 185 cm in height, and had his amputation due to vascular disease and gangrene. For the past 2½ years he was diabetic. A K-2 level ambulator, this individual used a transtibial socket with an expanded polyethylene foam liner and neoprene suspension sleeve. He was retired and used a bicycle for transportation instead of walking. Results from this subject (Figure 4) were much different from those of the healthy subject (Figure 3). There was a gradual ECF volume decrease over the session. Most notably the subject's limb did not enlarge during the walking intervals. Instead he kept on decreasing in volume.
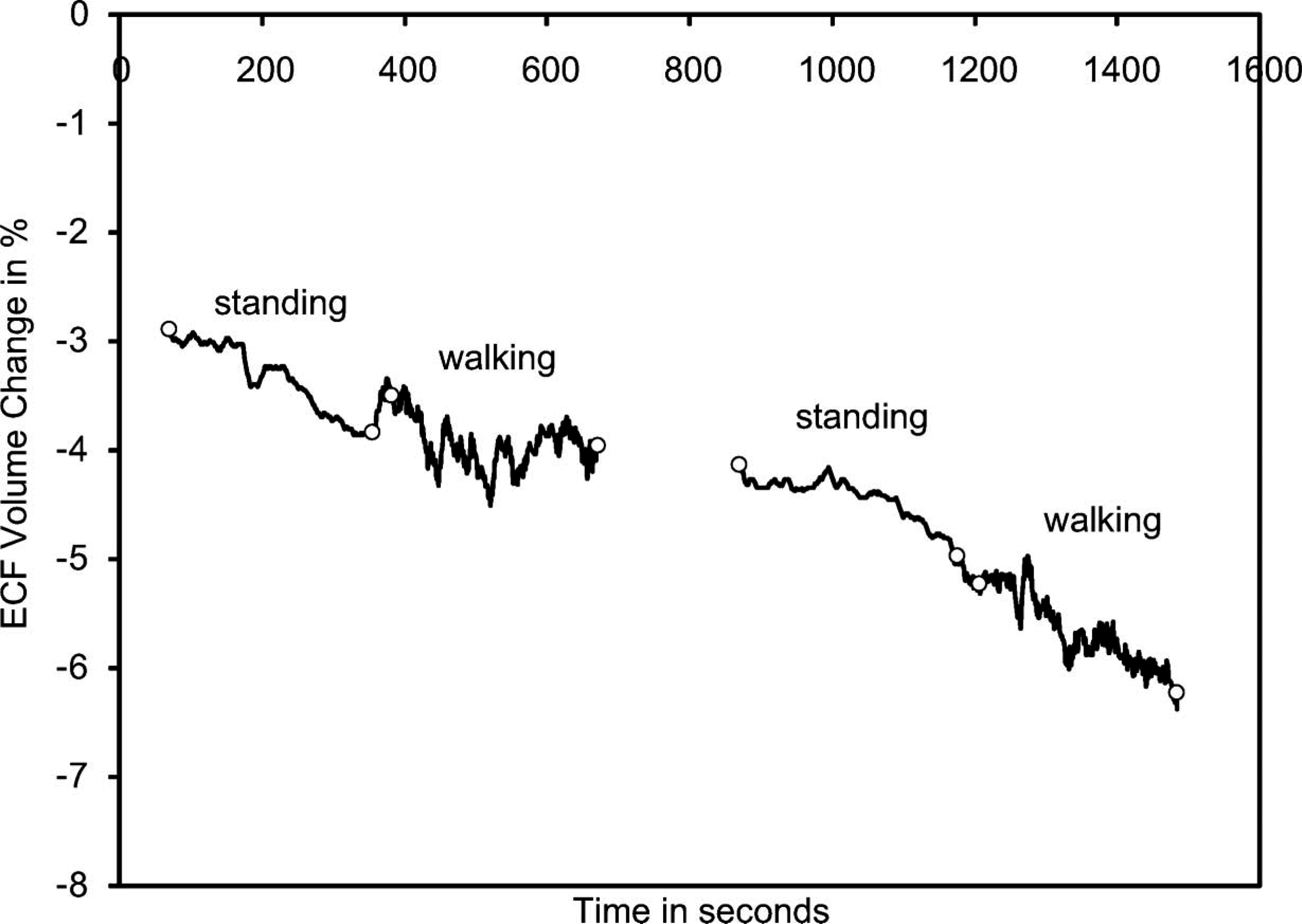
Results from the residual limb of a subject with vascular insufficiency (Case 2). For clarity, only data from the standing and walking segments are shown. The beginnings and endings of the standing and walking segments are marked with open circles.
Of particular note in this case was the subject's interpretation of his fitting. He was adamant that over the course of the session his residual limb was increasing in volume, not decreasing. He was sure that he was experiencing distal discomfort because his residual limb was swelling so excessively in the socket. He felt that the induced tightness was causing a slight throbbing sensation.
After two sessions with similar data collected on the subject, the research practitioner showed this subject the bioimpedance data. The subject was surprised that his limb was actually decreasing in ECF volume over the session. Interestingly, he quickly accepted the data as valid. In the subsequent session one month later, he was using a smaller socket and clinically his limb was much healthier. The improvement in his limb tissue health was marked. In the research practitioner's view, this attitude change to accept the new socket was due to his being shown the bioimpedance ECF volume change data. For months he had refused to use the new prosthesis with the smaller socket. Showing and interpreting for him the bioimpedance data helped to convince him that using the new socket was in his best interest.
Case 3
This subject was a 25-year-old female who had an amputation revision six months prior. Her original traumatic injury was two years earlier. She was in good health, a K-4 level ambulator, and an avid long-distance runner. She was 58.2 kg in mass and 160 cm in height. She used a transtibial socket with a silicone liner and locking pin.
Testing results on this subject showed substantial ECF volume decreases during standing (1.4%) and moderate increases during walking (0.5%) (Figure 5a). At 12 months post revision surgery, however, her ECF volume decrease during standing was less (0.9%) and her increase during walking was less (0.2%) (Figure 5b). Clinically, she had transitioned from adding two sock ply midday to not adding any ply during the day. Instead the additional two ply were put on in the morning when she initially donned her prosthesis.
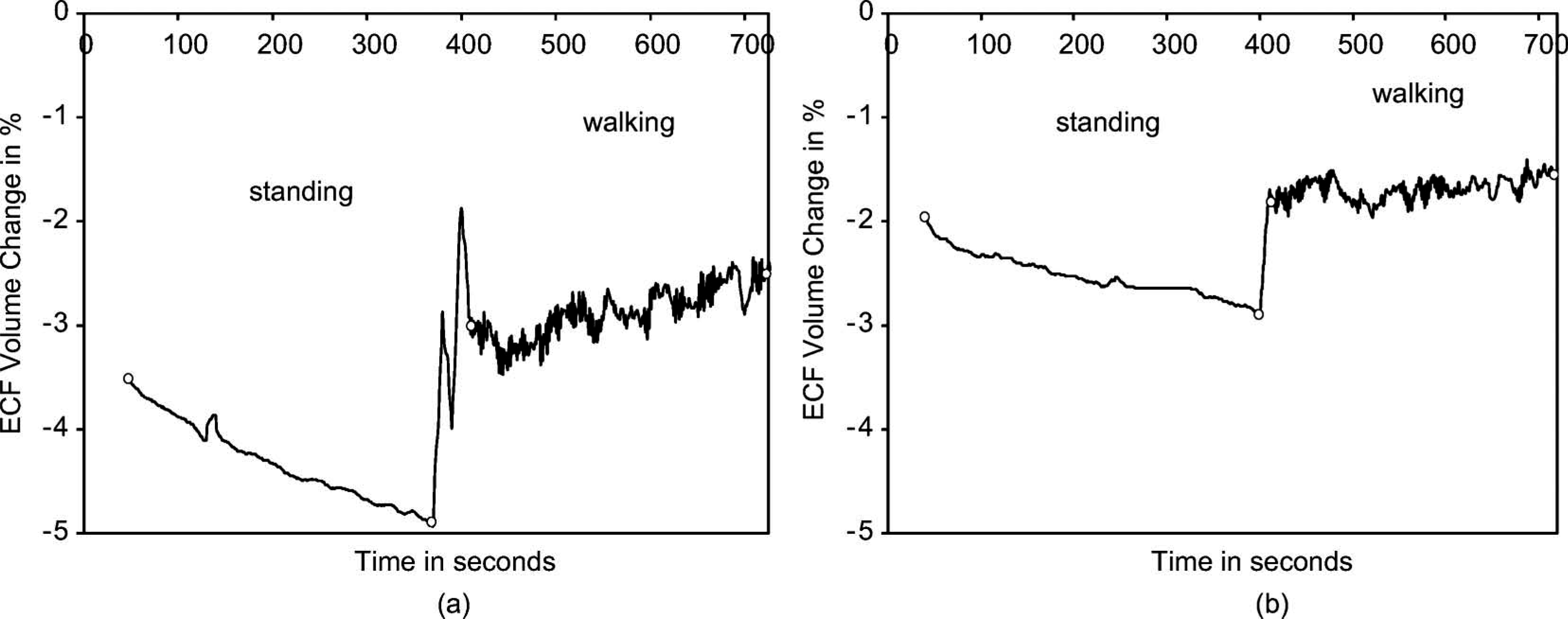
Results from the residual limb of a subject transitioning to a stable limb volume (Case 3). Only one set of stand and walk segments is shown in each plot. (a) Six months after surgical revision. (b) 12 months after surgical revision. The beginnings and endings of the standing and walking segments are marked with open circles.
Her bioimpedance results are consistent with those of the healthy subject who had a stable residual limb (Figure 3). After she stabilized, much less absolute value ECF volume change during both the standing and walking intervals was measured. Thus the bioimpedance data helped to reassure the subject that her change to no midday sock addition, consistent with the study practitioner's clinical interpretation, was due to limb volume stabilization.
Case 4
This subject was a 64-year-old male unilateral amputee who had his amputation 42 years prior. His cause of amputation was trauma. He was 90.5 kg in mass and 183 cm in height, and was a K-3 level ambulator. Though he was retired, he reported walking frequently. He used a transtibial socket with a wool sock, an expanded polyethylene foam liner, and neoprene suspension. He had congestive heart failure and was taking medication for it.
Results from sessions for this subject over many months showed very stable and consistent ECF volume changes. Results from a typical session shown in Figure 6a demonstrate a 1.3% ECF volume decrease during standing and a 0.2% decrease during walking. Results were essentially identical over a 16-month period.
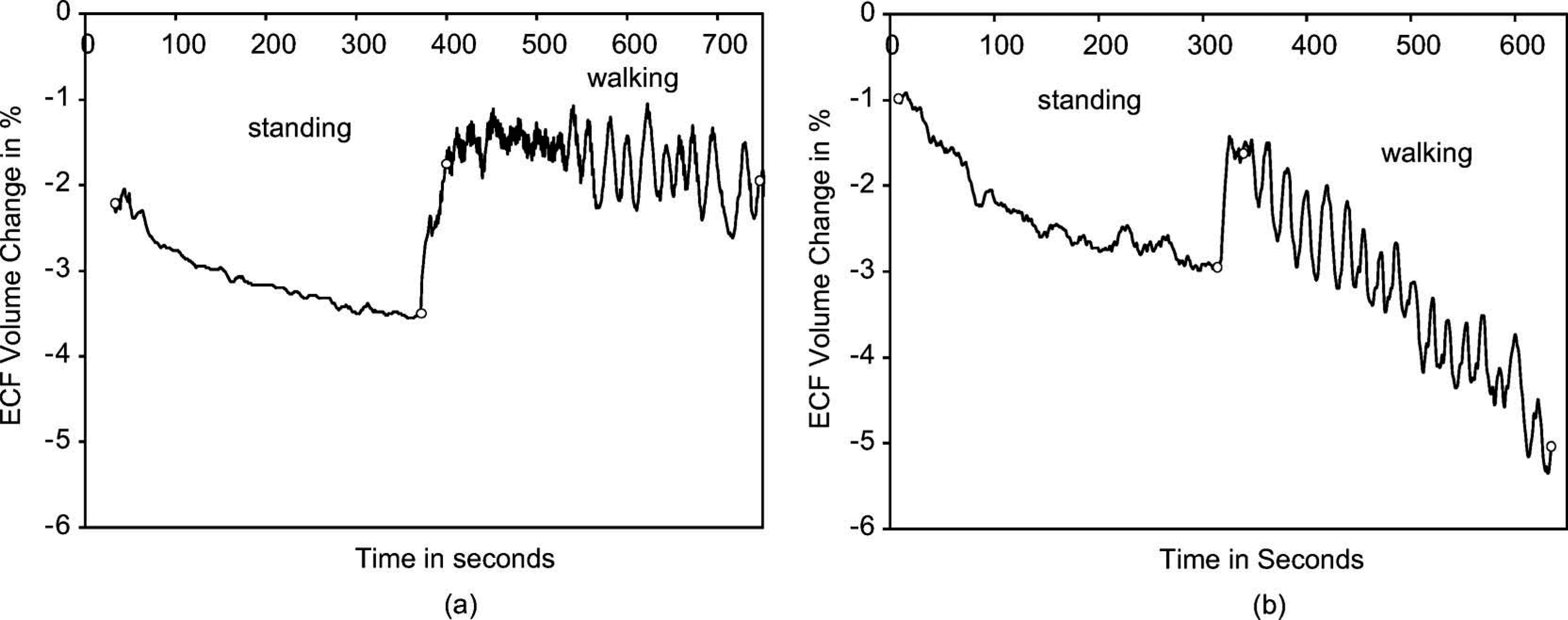
Results from the residual limb of a congestive heart failure patient changing the salt in his diet (Case 4). Only one set of stand and walk segments is shown in each plot. (a) On a healthy diet. (b) After three weeks on a high-salt diet. The beginnings and endings of the standing and walking segments are marked with open circles.
Drastically different results were obtained in a subsequent session. Instead of showing stable curves, his residual limb ECF volume decreased during the session, particularly during the walking interval (2.0% and 3.5% ECF volume decreases during standing and walking intervals, respectively) (Figure 6b), similar to results for Case 2 above. Upon querying by the study practitioner about recent changes to his diet or daily routine, the subject admitted that he had eaten a lot of ham and other salty foods over the past three weeks, unlike his usual low-salt diet.
Discussion
Measurement of residual limb ECF volume fluctuation achieved with bioimpedance analysis is potentially useful in clinical prosthetics practice. Because data are collected while the residual limb is within the prosthetic socket and while the subject is ambulating, insight into the effects of different activities on residual limb ECF volume change can be obtained. With an appropriate study protocol, we can gain insight relevant to clinical treatment.
The percentage change in the ECF volume is presented here as opposed to percentage change relative to the entire limb, i.e., including bone and adipose tissue. The percentages are higher than if the entire limb volume were used in the calculation. We present the data in this format not only because it is the convention in bioimpedance analysis but also because it is more appropriate for comparisons among subjects.
The visualization tool developed to present data in approximately real time (3 s delay) proved to be of important clinical utility during data collection. Commercial instruments (Hydra 4200, Xitron; SFB7, Impedimed, San Diego, California, USA) do not provide continuous display of fluid volume data. The ‘real time’ advantage allowed us to, for example, identify when a patient sat too long in a chair, circulation became compromised, and repositioning was needed. The capability to evaluate ECF volume change in real time facilitated individualised assessment.
Use of strain-gauge plethysmography was considered for this study instead of bioimpedance analysis. Strain-gauge plethysmography involves placing an instrumented wire around the proximal calf to measure limb circumference during activity. 21–23 Assuming the cross-section is approximately circular and that cross-sectional area is proportional to volume, limb regional volume change can be estimated. One limitation with this technology for use in external prosthetics is that the sensor will be compressed between the limb and socket during use which might detrimentally affect its performance. Second, depending of the limb-socket interface stress distribution, the strain-gauge wire might not remain in one plane during use but instead move proximally in one region and distally in another. This distortion and that prosthetic sockets are typically not right circular cylinders in shape, will confound the meaning of the measurement. Because of these potential limitations, we chose to use bioimpedance analysis in this research instead.
Presentation of total fluid data, the sum of intracellular and extracellular fluid, rather than exclusively extracellular fluid data was considered for presentation here. However, the intracellular fluid calculation required low error in the high frequency end of the spectrum of the Cole model. 14 In our collected data, there was greater noise in this region and thus greater error in the data than in the low frequency region where extracellular fluid resistance was determined. Thus the potential to introduce greater error coupled with the extracellular fluid volume change being of greater clinical interest in this application led us to present only extracellular fluid data.
Results from the contralateral limb of a healthy amputee subject (Case 1) (Figure 2) are consistent with results from normal subject limbs presented in the literature. 21–23 Using plethysmographic techniques, researchers demonstrated an increase in the volume of lower limb segments during quiet standing 21,22 and a decrease during normal walking at a 3 km/h speed. 23 During standing, gravity draws blood and interstitial fluid into the lower extremities, resulting in fluid accumulation and swelling. During walking, fluid flow out of the limb is induced via a muscular pumping action. 25–27 Also, interstitial fluid transport from the arterial vasculature into the limb is reduced, due to the increase in interstitial pressure from muscle contraction. 28,29 The overall result is a decrease in limb volume during walking.
Results from the residual limb of a healthy amputee subject (Case 1) during standing and walking showed trends opposite to those of his contralateral limb, and of the limbs of normal subjects reported in the literature. 21–23 The ECF volume decreased upon standing and increased upon walking (Figure 3). The main reason for the difference was most certainly due to the presence of the prosthetic socket. The prosthetic socket applies radial compression during standing, increasing interstitial fluid pressure and driving fluid out of the residual limb. It is reasonable to conclude that this interstitial pressure increase had a more substantial influence increasing transport out of the residual limb than did the influence of gravity on increasing transport into the residual limb. The result was a residual limb volume decrease during standing.
The presence of the prosthetic socket was also expected to be the main reason for the differences seen between the residual limb and healthy limb ECF volume changes during walking (Case 1). The ECF volume increase in the residual limb during walking was likely due to the release of the constant interstitial pressure from standing as well as an increased arterial fluid drive as the subject began moving. Arterial fluid drive into the interstitial space increases during walking due to the increased cardiovascular activity. The interstitial pressure was elevated as during standing, but it was now applied cyclically instead of continuously due to the cyclic action of the prosthesis on the residual limb tissues. The result was an arterial to interstitial fluid increase which in the short-term resulted in a residual limb volume increase.
We recognize that over a longer time interval than that investigated here a residual limb will typically decrease in volume. 30 It is reasonable to expect that this decrease results from the continual elevation of interstitial fluid pressure during socket use. The elevated interstitial fluid pressure in the long term would be expected to drive fluid out of the residual limb and dominate over limb volume increase induced by arterial to interstitial fluid flow.
For all of the presented cases, ECF volume change data measured during 5 min of standing followed by 5-min walking proved to give clinically useful information. This protocol was essentially a stress test. During standing, interstitial fluid pressure increased as expected, as a result of the increased radial compressive stress applied by the prosthetic socket on the residual limb during standing, and thus increased fluid transport from the interstitial space into the venous vasculature, decreasing the ECF volume of the residual limb. All subjects in this study showed residual limb ECF volume decreases during standing. During subsequent walking, arterial pressure increased but the limb ECF volume response varied depending on the health of the subject. In healthy subjects (Cases 1 and 3) residual limb ECF volume increased, we expect due to the increased fluid transport from the arterial vasculature into the interstitial space. These subjects' arterial fluid drives were strong and intact. The bioimpedance data for Cases 2 and 4, however, suggest a weak arterial fluid drive, consistent with their health conditions – vascular disease/diabetes for Case 2 and congestive heart failure coupled with a high-salt diet for Case 4. There was an imbalance between fluid leaving and entering the residual limb.
It is reasonable to expect that ECF volume changes during standing with equal weight-bearing reflect the porosity of the residual limb. A limb that experiences much transport from the interstitial space into the venous vasculature during standing, thus undergoes much ECF volume decrease, is very porous. A limb with minimal interstitial to venous transport during standing is not so porous. The results from Case 3 are consistent with this interpretation. After amputation, as her residual limb matured it decreased in volume and became firmer. The amount of fluid transport during standing decreased. The bioimpedance data quantified this maturation process. An interesting next step would be to use bioimpedance measurement to monitor subjects postoperatively to determine if the results can be used to establish the timing of definitive socket fabrication. Bioimpedance investigation might also prove useful towards evaluation of treatments to promote limb stabilization.
Case 4 presented in this report demonstrated that a useful application of bioimpedance analysis was in establishing when a subject's condition had changed. Because we had collected data on this subject previously we immediately recognized when a different response was measured. That difference prompted investigation of the source, which led to a clinically relevant issue concerning salt consumption in this cardiovascular insufficiency patient. We expect that the high salt intake coupled with his congestive heart failure condition caused him to retain much interstitial fluid, providing greater resistance to fluid transport from the arterial compartment into the interstitial space. This reduced transport would explain the decrease in residual limb ECF volume during walking. These results point to the potential value of bioimpedance analysis as a diagnostic tool in prosthetics practice.
The use of quantitative measurement to help demonstrate to a subject his or her status and the need for modification is a powerful tool in biomedicine. As demonstrated in Case 2, patients are more likely to accept a practitioner's diagnosis if shown data. Bioimpedance analysis potentially adds this very useful capability to a prosthetist's toolset.
This case (Case 2) also illustrates the importance of immediate data display towards use of the bioimpedance instrument. Practitioners and patients can immediately see the influence of their actions on ECF volume. The immediate feedback makes clear how prosthesis refinements, position changes, and activities influence the ECF volume change results.
A potential extension of the bioimpedance measurement and analysis methods described here would be to take measurements from both the residual limb and contralateral limb simultaneously. Because limb volume change data has been used effectively to identify vascular abnormalities, 31 the contralateral limb data could be used here to help distinguish systemic functional issues from those exclusive to the residual limb. Strain-gauge plethysmography instead of bioimpedance analysis could be used for the contralateral limb so as to avoid electrical interference problems between the two limbs.
Currently the bioimpedance analysis system used in this research is an experimental research tool. Interesting future research studies include investigation of the influence of suspension systems (e.g., suction sockets, vacuum-assist devices), socket design, and componentry selection on residual limb ECF volume change. Possibly bioimpedance analysis could be used in feedback systems to control and optimise prosthesis adjustments. To take this system to a clinical product level, practical concerns need to be resolved. Principally, logistical issues concerning electrode preparation and attachment need to be simplified, and the system needs to be made easier to use. There is potential to create new protocols to provide different insights into a patient's status and appropriate treatments, depending on the clinical need. It will be through extensive amputee subject testing and careful interpretation of the data that the clinical potential of bioimpedance analysis in prosthetics will be realized.
Conclusion
Bioimpedance analysis provided clinically useful insight towards understanding and managing limb ECF volume change in four case study patients with transtibial amputation.
Footnotes
Acknowledgements
This material was based on work supported by the National Institutes of Health (grant R01EB-004329).
