Abstract
Individuals with schizophrenia have a 20% shorter life expectancy than the general population [1] with the majority of this excess mortality from physical causes such as cardiovascular disease rather than from suicide [2]. Obesity and abnormalities in glucose and lipid metabolism are known to be risk factors for cardiovascular disease and are especially prevalent in those individuals with psychotic illnesses such as schizophrenia [3]. Individuals with psychotic disorders such as schizophrenia also appear to have poor access to adequate physical health care and poorer treatment of physical health problems [4,5]. In addition to this, there is increasing recognition and concern about the metabolic side-effects of second generation antipsychotics (SGAs). Individuals with schizophrenia who take such medications have an increased risk of significant weight gain, diabetes mellitus, and an atherogenic lipid profile [6–9].
It appears that individuals experiencing their first episode of psychosis (FEP) could be particularly susceptible to weight gain and metabolic dysfunction when taking antipsychotics, especially SGAs [6,10]. Concern over weight gain interferes with medication adherence [11,12], which may in turn increase relapse and impact negatively on quality of life [13,14], impacting on the early recovery process [15]. A recent systematic review has also highlighted that the use of antipsychotic medications is associated with increased cardiovascular risk in first episode psychosis populations [16]. At the same time, there is evidence that various pharmacological [17] and non-pharmacological interventions can prevent and manage this weight gain and could benefit this population if offered early [18–20]. Therefore, monitoring and addressing this metabolic risk is essential in terms of preventing long-term negative consequences.
Clinical guidelines have been produced recommending regular monitoring of weight gain and metabolic profiles in those prescribed antipsychotic medications [21]. In Australia such a consensus-based guideline was published in 2004 [22]. Despite these guidelines, the level of ‘metabolic monitoring’ is invariably poor in most clinical services. Data from the UK report low rates of monitoring in assertive outreach teams; for example only 17% of individuals had a recorded measure of obesity and 28% a recording of blood glucose in the previous year [23]. Similar low rates of monitoring are reported in the USA and Australia [24,25]. The limited data on levels of monitoring in FEP suggests that this is also the case in this population [26].
Evidence suggests that incorporation of evidence-based guidelines into every day clinical care does not necessarily follow on from the dissemination of guidelines [27,28]. A range of strategies has been used to disseminate and implement guidelines into clinical practice. The evidence in this area does not suggest any one single intervention as most useful but highlights the importance of using interventions that are specific to the setting and are theoretically driven [29]. The importance of identifying and addressing specific barriers and enablers for behaviour change is consistently highlighted in this literature [27,30,31].
Attempts to improve monitoring in other clinical services
It appears that legislative processes and practice recommendations such as the US Food and Drug Administration (FDA) department warnings and the American Psychiatric Association (APA) guidelines about metabolic effects of SGAs have had limited effect on practice [32]. More specific implementation strategies to improve monitoring for metabolic syndrome have been described by an implementation research initiative in the UK. Barnes and colleagues describe an intervention consisting of audit and feedback as well as provision of educational tools and physical health and lifestyle information for patients. There were attempts to individualize interventions to services, and to address some pre-identified barriers to monitoring. They demonstrated a considerable improvement in monitoring but noted that even after the intervention only a minority of patients received screening in accordance with best practice recommendations [33]. In Australia, others have attempted to outline potential barriers to improving practice [34] and have proposed the use of monitoring algorithms [35]. However, we were unable to identify any approaches to improving monitoring within a younger first episode psychosis population, a population that appears at particular risk for significant metabolic problems.
Concern about the incorporation of best practice regarding the management of side-effects from SGAs arose early in 2006 at our first episode psychosis programme the Early Psychosis Prevention and Intervention Centre (EPPIC) in Melbourne. At that time, clinicians reported that limited monitoring of weight gain and metabolic side-effects was occurring. We therefore aimed to investigate the level of monitoring in our service and whether an intervention, targeted at identified barriers and enablers to performing monitoring, could subsequently improve levels of monitoring.
Specifically, the aim of this study was to investigate whether a targeted implementation strategy could improve 1) general ‘screening’ of metabolic parameters in a first episode psychosis population, and 2) clinical guideline concordant metabolic ‘monitoring’ for those young people initiating antipsychotic medication. In addition, we also wanted to compare our current rates of screening for various indices with other services.
Materials and methods
Setting
Orygen Youth Health (OYH) is a comprehensive public youth mental health service for those aged 15–25 years old living in western Melbourne. The service has a number of continuing care teams, a dedicated crisis and assessment team and an inpatient facility. The first point of contact with the service is the crisis and assessment team or inpatient unit, and then, depending on the clinical presentation, the patient is transferred to the appropriate continuing care team (CCT). EPPIC is a separate CCT within this service and accepts patients with FEP. To be accepted into this clinic, patients must have a diagnosed psychotic disorder and have not previously received more than 6 months of treatment for this. Patients receive a maximum of 2 years treatment with the EPPIC CCT. The EPPIC clinical team has been described in detail elsewhere [36]. EPPIC receives around 300 new referrals a year. Patients accepted by EPPIC are assigned an individual ‘case manager’ and a treating psychiatrist. There are six psychiatrists and ten case managers (mixture of psychiatric nurses, social workers, psychologists and occupational therapists).
Trial design
The trial was a quasi-experimental before-and-after study, with data collection taking place at baseline or pre-intervention and 2½ years later following the targeted practice improvement intervention (post intervention).
Eligibility and recruitment
Inclusion/exclusion criteria
The intervention was aimed at the entire cohort of clinicians (case managers and psychiatrists) currently working in the EPPIC clinic as well as those working on the inpatient unit and the crisis/assessment team during a 6-month period between January 2008 and July 2008. There were no exclusion criteria for clinicians as this was a service-wide intervention. The files of all consecutive patients entering the EPPIC clinic during the pre-intervention and post-intervention time points were audited. Files where patients were not prescribed antipsychotic medication during their treatment at EPPIC were excluded.
Assessment of monitoring practice
The audit of metabolic monitoring practice was performed using a predesigned audit form. The time points specified for monitoring (and therefore audited) were derived from our guideline (see Box 1). The audit format differed slightly between pre-intervention and post-intervention. This was because the service monitoring practice guideline was modified in the time between the two audits and the second audit needed to capture these changes. The audits were directly comparable in terms of time points for monitoring and the groups of metabolic measures undertaken.
The pre-intervention audit was undertaken on the files of all EPPIC patients presenting consecutively to the clinic for the first time between 1 January 2006 and 30 June 2006. Data on metabolic monitoring/screening in the preceding 18 months was extracted from these files. The results of this pre-intervention audit were also used to inform the intervention strategies (see below). A post-intervention audit was performed on patients consecutively presenting to the EPPIC clinic between 1 September 2008 and 28 February 2009. Data on metabolic monitoring/screening in the preceding 6 months was extracted from these files. This was 6 months only because the post-intervention audit was performed in December 2009 and therefore information was only available for 6 months for all patients included in the audit period.
Box 1. Guideline for monitoring of weight and metabolic side-effects developed at the service
Approved EPPIC guidelines for standard clinical practice for weight and metabolic monitoring (developed on the basis of guidelines and in consultation with multi-site experts in the field and clinical team and approval from clinical management group)
Indices to be assessed:
Height and weight to estimate BMI
Systolic and diastolic blood pressure
Waist and hip circumference (to obtain waist-hip ratio)
Fasting blood glucose
Full fasting blood lipid profile (including total cholesterol, low and high density lipoprotein and triglycerides)
Number of cigarettes smoked daily
Level of daily exercise
Assessment time points:
Baseline
1 month
3 months
6 months
12 months
18 months
Each file was individually examined for evidence of completion of metabolic monitoring at each time point using all sections of the clinical file, including ongoing progress notes. Three of the authors performed the first audit (SH, AP and MAJ). All these investigators re-audited a random selection of 10 files from this audit; the reliability of assessments for the main outcome measures was 100%. Four student researchers performed the second audit. A reliability check on five randomly chosen files was performed early in the audit process. One student failed to identify data for a main outcome measure and was subject to further training using the audit tool.
Ethics
Ethical approval for the project was received from the Melbourne Health ethics and quality assurance committee.
Outcome measures
Guideline concordant metabolic monitoring
This was defined as completion of all recommended monitoring indicated in Box 1 at baseline, 1 month, 3 months and 6 months (post-intervention data on the 12 and 18 month time points was not available). Our guideline recommendations were substantially stricter than those from other bodies [3,22], for example, we specifically added a 1 month time period to monitor the early weight gain identified by studies in FEP populations [18]. Therefore, we also defined additional minimum monitoring and screening standards as outcome measures.
Minimum metabolic screening
This was defined as the completion of a full ‘set’ of metabolic measures including obesity measures (BMI or weight and height or waist-hip ratio); and metabolic blood tests (lipids and glucose) at some point within 6 months of being prescribed an antipsychotic
Minimum metabolic monitoring
This was defined as the completion of full baseline measures including both obesity measure (BMI/waist-hip ratio/or weight) and metabolic blood tests plus the completion of full measures at between 1 and 6 months following initiation of antipsychotic medication (or 1 to 6 months after baseline).
Secondary outcome measure
Additionally, we aimed to compare our post-intervention monitoring rates with those of other services. Therefore, we calculated monitoring/screening rates of four separate metabolic indices individually from the post-intervention audit both at baseline and at some point in first 1–6 months following antipsychotic initiation. This enabled direct comparison of our results with those of previous studies from other services. These four metabolic indices were 1) obesity measures (BMI/waist-hip circumference/weight), 2) blood pressure, 3) glucose measure (glucose or Hb1Ac, fasting or random), 4) blood lipids (fasting or random). This information was only available from the post-intervention audit.
Other outcomes
We compared the rates of documented interventions for metabolic/weight disturbance offered by clinicians pre- and post intervention.
The intervention
Barrier and enabler analysis
The intervention was developed on the basis of interviews conducted with psychiatrists at EPPIC following the baseline audit. The interviews have been described in detail elsewhere [37]. Interpretation of the interview data using thematic analysis allowed identification of barriers and enablers to implementing monitoring guidelines that were mapped to theoretical domains of behaviour change.
Based on the barriers and enablers identified in the interviews by psychiatrists [37], a multifaceted intervention using a range of behaviour change strategies to target the identified constructs and implement routine monitoring was developed. The choice of interventions was informed by evidence of effectiveness and expert opinion [29]. The intervention was also informed by the pre-intervention audit; for example, we were able to identify which parts of the service were better or worse at performing monitoring and therefore where to focus particular parts of our intervention.
Components of intervention
1) Development of local guidelines (see Box 1).
We developed local consensus evidence-based monitoring guidelines for the service, and distributed this to all clinicians as a wall poster. This guideline is shown in Box 1. These were based on a number of the guidelines including the Australian consensus statement [22]. For further information regarding this see Hetrick et al. [37]. The existing form that was being used to record metabolic measures was adapted to reflect the guidelines for monitoring and to serve as a reminder of what and when to monitor. It was placed in all files by medical records staff.
2) Educational intervention
A series of didactic and interactive seminars were conducted, using persuasive communication to highlight the long-term consequences of metabolic disturbance and weight gain in young people taking SGAs. The seminars were also used to disseminate the recommendations from the locally developed guidelines. In addition, the seminars provided feedback about the service-wide levels of monitoring from the pre-intervention audit of current clinical practice.
3) Service/structural changes
Structural change was also part of the intervention. Prompts were provided in the form of the paper-based monitoring sheet being placed in every patients’ file. Regular reviews of a patient's metabolic status were built into the EPPIC clinical review process (which occurs on a 3-monthly basis for all clients) overseen by a metabolic ‘champion’ responsible for reminding clinicians. This ‘champion’ was a member of the research team and was a source for further consultation if there were difficulties in monitoring. A service policy on metabolic monitoring was also developed and distributed to all psychiatrists in the service.
4) Provision of monitoring equipment
We ensured the equipment required to undertake monitoring (e.g. scales, tape measures, blood pressure cuffs) was located in each psychiatrist's room. Stamps that indicated the necessary blood tests for monitoring were placed in the psychiatrists’ rooms to aid ordering and completion of the correct blood investigations.
This range of interventions was initiated over a 6 month time frame beginning on 1 January 2008 and ending on 30 June 2008 with the expectation that they were subsequently integrated into the standard clinical infrastructure and pathways of the service.
Statistical analysis
We compared the monitoring at the two time points (pre- and post intervention) on the three main outcome measures of guideline concordant monitoring, minimum metabolic screening (MS) and minimum metabolic monitoring (MM) as well as the secondary outcome of the number of interventions offered to clients. The data was analysed using SPSS version 18 (SPPS, Chicago, IL). The differences in rates between pre- and post intervention were examined using t-tests for continuous data and Pearson's and Mantel-Haenszel chi for categorical data.
Results
Of the 119 patients presenting to EPPIC for the pre-intervention audit, only 118 files could be obtained for audit as one file was missing. Of these 118 files, only 106 were audited, as the other 12 patients were not prescribed antipsychotics during their treatment. For the post-intervention audit there were 106 patients presenting to EPPIC at this time. The files on two of these patients could not be located and 18 of these patients were not prescribed antipsychotics; 86 were therefore eligible for audit. There were no differences in entry criteria into the service between the two audit time points. There was no difference in gender distribution of patients in the two audits (37.3% female in pre-intervention and 29.9% in post-intervention audit, p = 0.26). The mean age of patients in the post-intervention audit was significantly lower than those in the pre-intervention audit (20.2 years, standard deviation (SD) 2.5 compared to 21.1 years, SD 2.7, p = 0.02).
Difference in guideline concordant metabolic monitoring, metabolic screening and metabolic monitoring pre- and post intervention
No patient had all the strict guideline concordant metabolic monitoring completed for the 6 months following starting antipsychotic medication at either audit time point. Based on our defined criteria, minimum metabolic screening (or completion of full metabolic measures at some point within 6 months of being prescribed an antipsychotic) was completed on 22.2% (n = 24) of patients at pre-intervention. A significant improvement to 81.4% (n = 70) was found post intervention (Mantel-Haenszel chi = 8.171, p = < 0.001). The rate of minimum metabolic monitoring (completion of full baseline measures plus the completion of full measures at between 1–6 months following initiation of antipsychotic medication) was low at both time points but did improve significantly from 1.7% (n = 2) to 39.5% (n = 34) post intervention (Mantel-Haenszel chi = 6.897, p = < 0.001). The rates pre- and post intervention are shown in Figure 1.
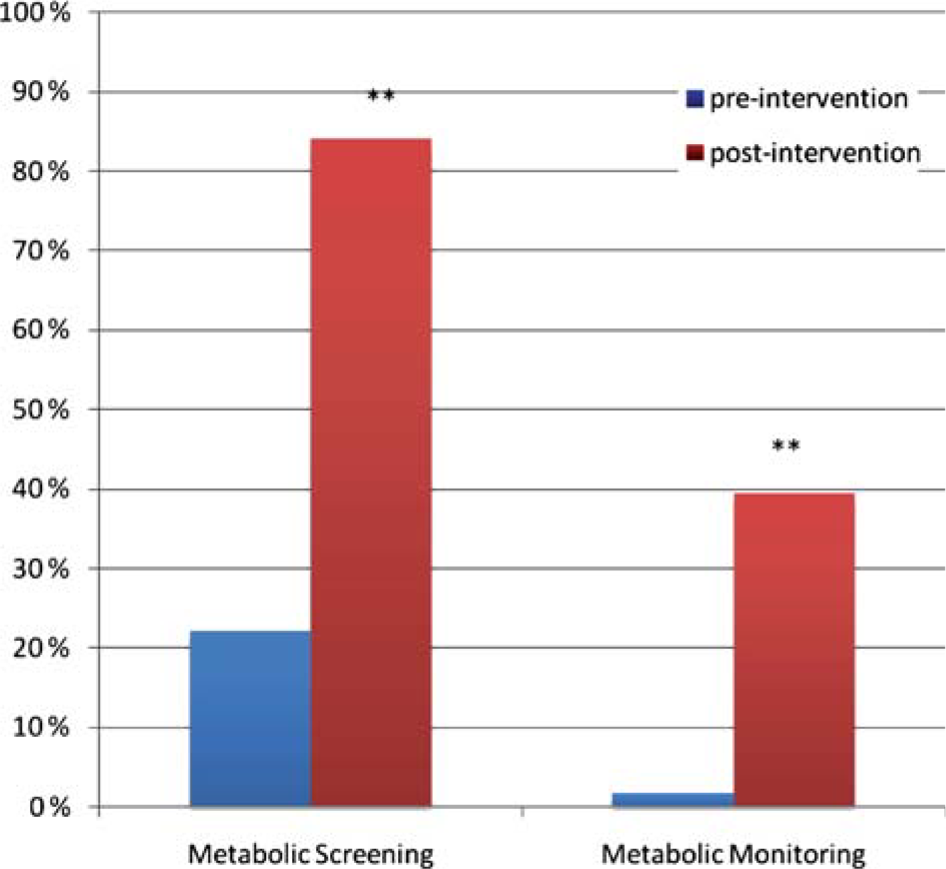
Percentage of clients with minimum screening (MS) and minimum monitoring (MM) at pre- (n = 106) and post-intervention (n = 86) time points. **p < 0.001
Rates of metabolic screening and metabolic monitoring (following antipsychotic initiation) over a 6 month period for four separate metabolic indices at the post-intervention time point and comparison with a UK audit over a 12 month period [33]
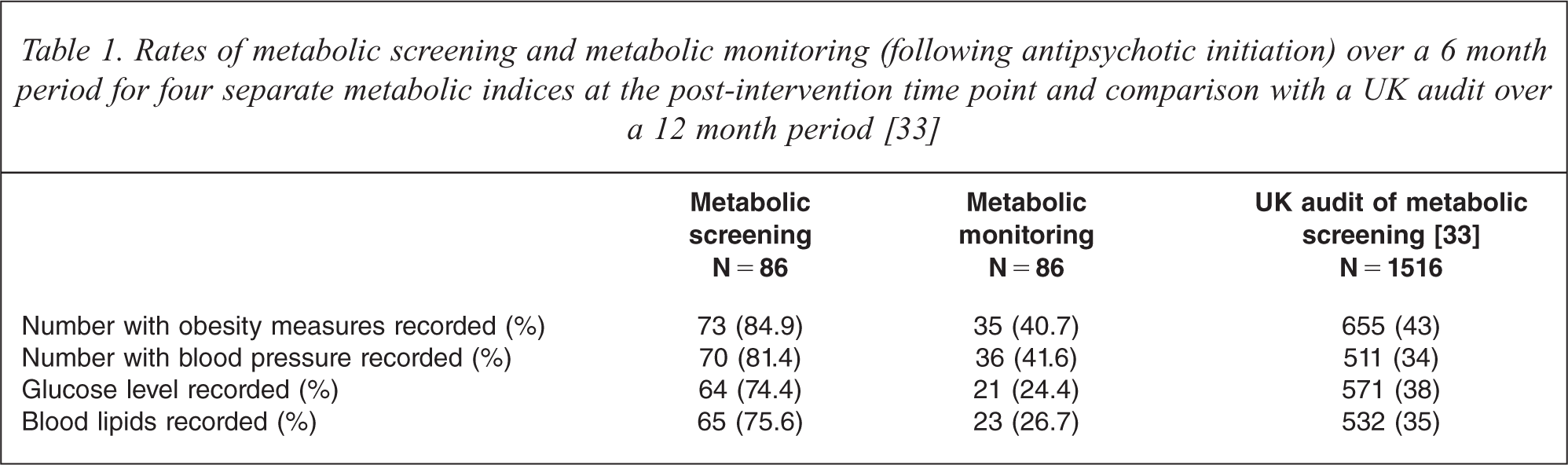
Post intervention rates of monitoring/screening for the four separate metabolic indices
The monitoring and screening rates (as previously described) for the individual metabolic indices of obesity measures, blood pressure, glucose measure and blood lipids are shown in Table 1. The individual rates were higher for the screening outcomes (74.4% to 84.9%) than for the monitoring outcomes, which ranged between 17.4% for blood lipids to 34.9% for obesity measures.
Rates of preventative interventions
Finally, with a view to implementing service-wide targeted preventative interventions, the number of clinicians intervening to prevent patients’ potential weight gain and metabolic side-effects was analysed. Overall, there were only eight (7.2%) instances where clinicians documented they intervened with their patients in the pre-intervention sample. Post intervention the number of patients where clinicians had documented interventions was 22 (29.3%). This was significantly higher than pre-intervention (Mantel-Haenszel chi = 4.069, p = < 0.001).
Discussion
Summary of results
Following the implementation of a targeted implementation strategy that addressed barriers and enablers, there was a significant improvement in both the overall screening and initial monitoring of metabolic indices. There was also an improvement in number of interventions for metabolic problems offered by clinicians. The rates of screening for the four specific areas of metabolic problems (weight, blood pressure, glucose and lipids) were relatively high post intervention. However, despite the implementation strategy, the rates of monitoring metabolic disturbances following initiation of SGA medications were still far from concordant with current clinical guidelines.
Comparison of screening rates with other services
The screening rates in the post-intervention audit presented here (Table 1) are considerably higher than a recent UK audit of a FEP service in the UK [26]. A small study in the USA reported similar rates of screening in the month after starting antipsychotic treatment for FEP patients in a specialized metabolic clinic and an inpatient setting, but lower rates than ours in a standard outpatient setting within the service [38]. Our rates are also higher than those given for screening from adult psychiatry services in the UK [33,39]. The results we present in Table 1 are derived from very similar variables to the two UK studies of Barnes and colleagues [23,33], the main difference being that the screening rates from our study are taken over a 6 month period as opposed to a 12 month period in these studies (and therefore our results may be higher than if we looked at a 12 month period). Despite this, screening rates remain over twice as high in our post-intervention sample than in this study, as shown in this table. Direct comparisons with previous research for metabolic monitoring following initiation of antipsychotic medication are more difficult to make. However, our rates of baseline testing on initiation of medication compare favourably to those for baseline testing in relation to new prescriptions for antipsychotics in the USA [24].
Comparison with other quality improvement programmes
Other similar quality improvement programmes have managed to change practice in this area. However, these have been in older populations, and generally employed less targeted approaches to the implementation strategies used that did not specifically address local barriers and enablers to change [33,40]. It also appears that the first episode population, given the relatively younger age than general adult psychiatry patients, may suffer from additional difficulties with regard to accessing and prioritizing physical health care compared to those in adult mental health services. For example, in our service, at least 60% of the clients are unable to identify a regular General Practitioner (GP). Some monitoring, such as blood tests, were not routinely done on site and relied on a young person accessing a local pathology service or a GP. Low monitoring of baseline metabolic blood levels have been reported in other first episode psychosis populations [26,38]. It is therefore not unexpected that these rates are relatively low, but calls for more creative strategies to address this particular problem such as the headspace initiative [41].
Limitations
This was a naturalistic study examining the effects of a series of implementation strategies on current practice. Given the non-randomized nature of the trial we cannot be sure that the change was not simply an effect of time and an increase in profile of metabolic problems in this population. There was also some acknowledged turnover of staff, and the clinicians whose behaviour was subject to the first audit were not exactly the same as the follow-up. However, whilst the rotation of junior psychiatrists is an inevitable consequence of psychiatric training in public hospitals, the majority of the senior staff and ‘culture carriers’ were the same at the two time points. We collected metabolic monitoring data from our service clinical files alone. It is possible that patients may have occasionally had tests ordered by other physicians such as GPs and that the results were not directly communicated to our service. Given that the number of these tests would be relatively small and unlikely to be intended for the purposes of metabolic monitoring of antipsychotic treatment directly, we feel this does not significantly bias our results. The post-intervention sample was statistically younger than the follow up sample, but given the small difference (0.9 years) and the fact that we audited consecutive cases presenting to the service over a time period, we do not feel this would have influenced our results.
Future directions
We are encouraged that the interventions were able to change practice in a number of areas. However, practice is still far from concordant with our guidelines or even our less stringent minimum guidelines. We were aware, both early in the study, and from our final results, that our recommended guidelines were particularly strict for clinicians to follow (especially the 1 month time point) in relation to other guidelines. Despite this, further intervention approaches are obviously needed to improve levels of monitoring in our service. Audit and feedback have been shown to be effective when baseline concordance with recommendations is low and when the audit and feedback are continuous [42]. We plan to make more continuous audit and feedback possible by a specifically designed electronic database to capture routine metabolic monitoring data for each client.
We are also investigating the effectiveness of changes to service delivery; we are considering trialling the use of a dedicated nurse being available for physical consultation in the same building. Other researchers have highlighted success in monitoring when using a dedicated physical health nurse to co-ordinate and administer monitoring [43] or a specialised metabolic clinic [38]. However, both approaches have financial implications and would need evaluation of their cost benefit.
Conclusions
Using a targeted implementation strategy we were able to substantially improve the routine screening and monitoring of weight and metabolic indices in a first episode psychosis service. Implementing guideline-concordant routine monitoring is essential to ensure better outcomes for patients. Services should endeavour to use such targeted implementation strategies in other areas where practice does not conform to guideline standards.
Footnotes
Acknowledgments
The authors would like to thank all the clinicians from the service who participated in the quality improvement programme, especially Wei Soong, Karen O'Connor, Yang Yun and Cameron Duff. We would also like to thank Kurt Zhao and Sandy Carmichael for their input into data collection.
