Abstract
Borderline personality disorder (BPD), it is increasingly recognized, is an important clinical and communal problem. It is a condition with a high mortality, approaching 10% in some studies [1,2] and a morbidity that imposes a severe burden on the health system. It is more common than was once supposed, having a lifetime prevalence of 5.9% in the general population [3]. It can be diagnosed in about 20% of inpatients and 10% of outpatients [4]. It has a significant comorbidity with all major Axis I disorders and to a greater extent than other personality disorders [5]. BPD is thus important not only in itself as a serious condition but also because of its effect on the duration, recurrence and outcome of Axis 1 disorders [6,7]. These findings lead to the view that work towards a more effective means of treating BPD, must be a significant component in building a modern mental health system. This achievement will depend, to a great extent, upon an understanding of the basic pathophysiology of BPD, upon which there is no general agreement among those who have devised treatment approaches for the condition. Such an understanding should contribute to a refinement and development of these approaches and current therapies.
This study derives from two related proposals about the origin of borderline personality disorder (BPD). The first is the basis of one current therapy for BPD, the conversational model [8–10]. It suggests that BPD is a manifestation of a deficiency in the maturation of that state of mind which Hughlings Jackson [11] and William James called ‘self’ [12], and which Edelman calls ‘higher order consciousness’ [13]. The identifying feature of this complex form of consciousness is a reflective awareness of inner events [12, p.297]. Although they do not approach directly the subject of self, the hypothesis of Bateman and Fonagy regarding a failure of ‘mentalization’ in BPD resembles this proposal [14].
It is proposed that a particular kind of interplay, mediated by conversation, or in infancy, proto-conversation [15], is necessary to the maturation of this form of consciousness [9,16]. A deficiency in this form of relatedness, together with the effect of traumatic impacts upon the psychic system, is considered to impair such maturation, consistent with evidence that family background and exposure to abuse are independent predictors of the development of BPD [17]. This is a more specific formulation of a hypothesis which was put forward by Stern (1938) [18] in his pioneering paper which gave the first outline of BPD. He suggested that an aspect of his patients’ maturation had been impeded due to ‘psychic starvation and insecurity due to lack of parental, chiefly maternal, affection’ (p.470). He also pointed out that ‘actual cruelty, neglect and brutality by the parents of many years duration are factors found in these patients’ (p.470).
The second proposal concerns a failure of inhibitory control in BPD which is predicted by a developmental model of ‘self’ based on the theories of Hughlings Jackson [19,20] and is supported by observations such as those of Silbersweig et al. [21]. Schore has suggested that inadequacy of inhibitory control over limbic output, central to BPD, is likely to be manifest right-hemispherically [22,23]. He proposed that a particular disturbance of mother–child relatedness leading to ‘traumatic attachments’ results in ‘a deficit of the higher right brain regulation and lower right cortically driven aggressive states’ [23, p.299]. This study tests the hypothesis that borderline personality involves a relative failure of higher order, presumably prefrontally connected, inhibitory mechanisms which will be especially evident in the right hemisphere.
The hypothesis is tested by means of a study of P3a, an early component of the late positive complex of event-related potentials (ERP), or P3(00), associated with the 300 ms latency range.
The P3 is one of the most investigated endogenous brain potentials in psychiatric research. Studies of BPD patients revealed the reduction in amplitude and latency prolongation of auditory P3 [24–26]. However, the characteristics of the P3a were not revealed in these BPD studies because the ability of the conventional method of ERP averaging to identify the P3a are quite limited.
A principal methodological difficulty is that reliable identification of the P3a from single trial records necessitates effective tools for significant improvement of the signal to noise ratio. To meet this condition most previous techniques neglected the existence of the P3a and used a single template for reliable identification of the larger and more prominent P3b [27]. In the new method of single trial analysis supported by multiple templates, no such simplifications are necessary and the P3a and P3b reconstructions are reliably realized [28,29].
A comprehensive evaluation of P3a and P3b as distinct ERP components offers significant promise for investigation of the inhibitory aspect of the attention process. P3a is frontally connected and makes up the early part of P3. P3b arises somewhat later and is particularly parietally connected [30].
P3a reflects novelty detection [31] and orientating [32] while P3b is less clearly understood. Verlerger and his colleagues suggest that it is akin to a response set [33]. Soltani and Knight [31] see P3a as an automatic and P3b a more volitional response.
Polich [34] has suggested neuroinhibition ‘as an overarching theoretical mechanism for P300’. Inhibitory activity is necessarily recruited in the switching of attention from one event to another. This inhibitory function is reflected in the impairment of P3a, particularly its habituation [35]. P3a is underpinned by dopaminergic pathways while P3b depends upon norepiphrenic neurotransmission [32]. Parkinsonian subjects, who are relatively dopamine depleted, have difficulty in switching attention and exhibit impaired habituation of P3a [36].
The inhibitory aspect of the attentional process, as reflected in P3a, develops as the individual matures [37]. In borderline patients P3a is abnormally large in amplitude, suggesting that this maturation is impeded. In this study, we investigate the possibility that this impairment is particularly right sided. We believe this report to be the first, apart from our previous study, concerned with attentional processes in BPD as reflected in P3a.
Subjects
Seventeen patients with BPD (4 males and 13 females; mean age 31.6 years, SD 7.9, range 20–44 years) participated in the study. The BPD patients came from an ongoing programme for the treatment and evaluation of BPD patients. Patients were free of medication for at least 30 days at the time of the study. The sample is relatively small since the majority of BPD patients were heavily medicated at first presentation, the medications almost invariably including antidepressants. The subjects were consecutive admissions to the programme of those patients who were medication free. The data were collected over a two-year period. The diagnosis was made by two independent raters (psychiatrist and psychologist) in a clinical assessment and according to DSM-III-R criteria in a diagnostic interview that included the Diagnostic Interview for Borderline Patients. The subjects were not considered by their referring doctors or at the clinical assessment to be suffering major depressive disorder.
The control group included 17 (4 males and 13 females; mean age 34.3 years, standard deviation (SD) 8.6, range 20–47 years) age and sex matched healthy subjects recruited from the general western Sydney community. For both groups, exclusion criteria were left-handedness; a recent history of substance abuse, epilepsy, or other neurological disorders; and head injury, which were assessed with section M from the Composite International Diagnostic Interview [38] and the Westmead Hospital clinical information base [39]. Subjects were asked to refrain from smoking or drinking caffeine for three hours prior to the recording session. Written consent was obtained from all subjects prior to testing in accordance with National Health and Medical Research Council guidelines. Control subjects were also screened for the history of psychiatric illness (themselves or first degree relatives).
Procedure and ERP recording
The ERP data were collected according to a standard auditory ‘oddball’ paradigm in a method similar to that used in several previous studies [29,37]. In brief, auditory tones (50 ms in duration, 10 ms of rise and fall time) were presented pseudo-randomly via stereo headphones to both ears at 60 dB above each individual subject's auditory threshold; 15% were target tones presented at 1500 Hz, the remaining 85% were background tones delivered at 1000 Hz with the constraint that there were no successive target stimuli.
The subjects were instructed to ignore background tones and to press a reaction-time button to target tones with the first finger of each hand. The identification of each target was regarded as correct if a button press response was obtained within 1 s of the target tone. Reaction time was recorded for each button press. EEGs were recorded from 19 electrode sites according to the 10–20 international system with linked ears as a reference using a DC acquisition system. An additional two channels were used for detecting horizontal and vertical eye-movement potentials. Impedance of all electrodes was less than 5 kΩ. The voltages were continuously digitized at 250 Hz and digitally stored. EOG correction using a technique based on [40] was carried out off-line. The successive EEG segments time locked to correctly detected target tones were then tested on the presence of excessive artefacts. An accepted trial was then digitally filtered. A 251 time-points time series extracted from 0.2 s pre-stimulus to 0.8 s post-stimulus was digitally stored and the procedure continued until 40 eligible segments were acquired.
Data analysis
Late component ERPs were identified in single trial ERP (recordings) using the fragmentary decomposition (FD) method described in detail elsewhere [27,28]. The method is a further development of conventional single trial screening procedures with a component template. Using the positive half cycle of a 2 Hz sine wave as a template, the previous technique was addressed to a single P3 component [27]. The FD-based technique uses adaptive segmentation and high resolution FD which resolves component temporal overlap [28]. This advanced signal processing technique provides means to match an individual template to both the P3a and P3b components detected in a single trial record. Within the latency windows of 80–120, 160–220, 180–235, 240–299 and 300–360 ms, the N1, P2, N2, P3a and P3b components, respectively, were identified as positive (P) or negative (N) waveforms with the maximum of absolute peak amplitude.
An important aspect of the variability of single trial parameters is a non-Gaussian character of frequency distributions of the peak amplitudes of the late ERP components [28]. Accordingly, conventional parametric estimates have been validated using a non-parametric Mann-Whitney U-test for inter-group comparisons.
Lateralization of brain potentials has been quantified by comparing the potentials on homologous electrodes (e.g. locations F3 and F4) over both hemispheres. The inter-hemispheric relationships were estimated using the pairs of quantities (AL, AR), where AL and AR denote P3a peak amplitudes at the right and left homologous electrodes, respectively. The difference between the peak amplitudes, ΔA = AR − AL, was taken as a measure of lateralization.
Using sequence sensitivity of ERP single trial analysis we tested relevance of identified single trial ERP components to the novelty processing in the context of a habituation. The decrease or increase of peak amplitude with the target stimulus repetition was evaluated using regression analysis. Habituation is associated with negative values of the slope.
Results
Inter-hemispheric asymmetry of P3a voltages in BPD patients
Consistent with the previous reports [29,37], single trial records with identified P3a and P3b components comprise three major types of activity patterns illustrated by Figure 1. Between-group differences in the mean peak latencies of the P3a are statistically insignificant for all electrode sites. This observation supports peak amplitudes as adequate measures of inter-hemispheric relationships.
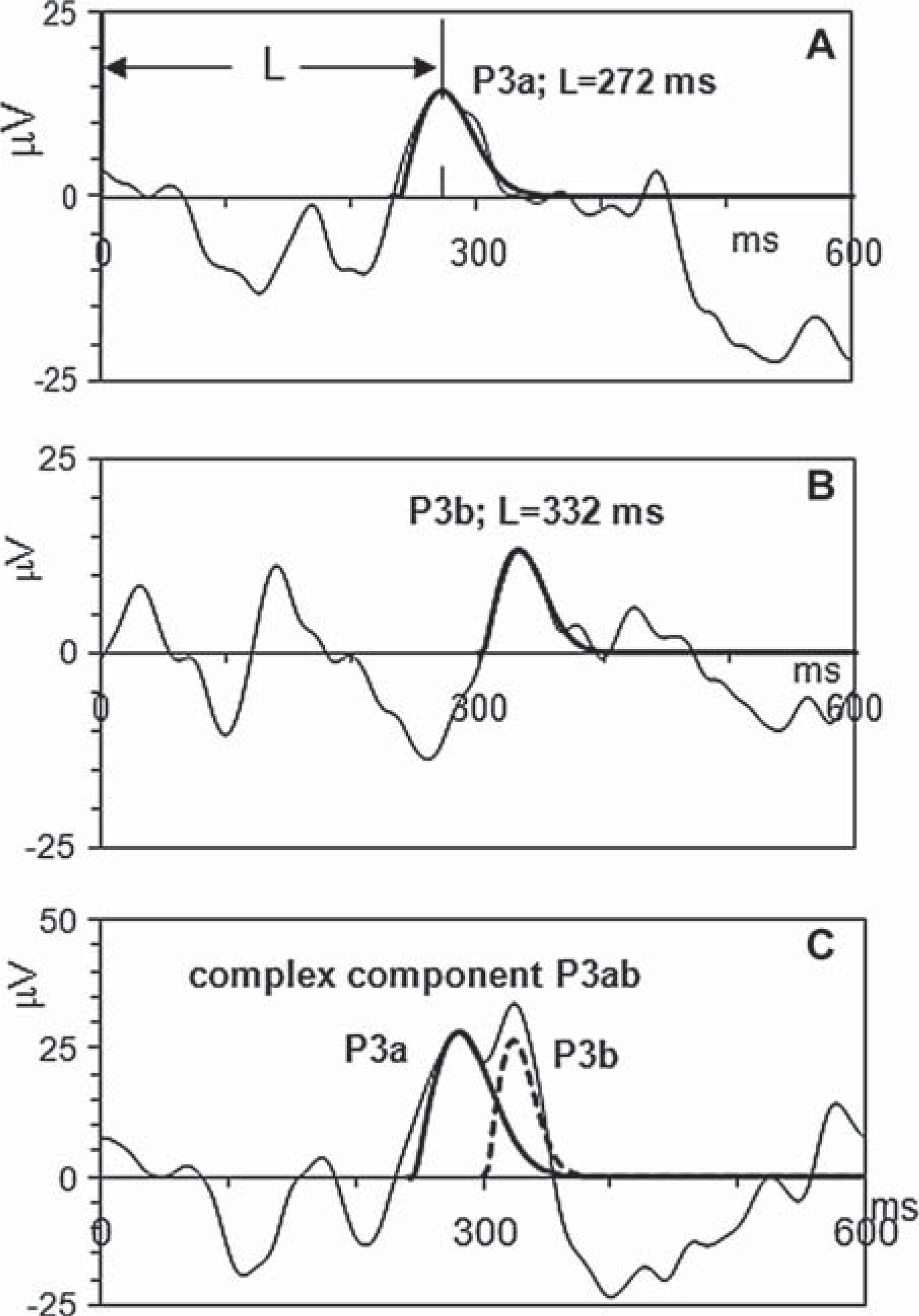
The light lines indicate event related potentials elicited in different single trials. The darker lines show templates which identify P3a and P3b components of single trial ERPs. The x-axis units are milliseconds, and the y-axis units are microvolts. L indicates the peak latencies of P3a and P3b in A and B, respectively.
The results of comparing P3a over homologous electrodes have shown that peak amplitudes of P3a in the left and right hemispheres of BPD patients are significantly different. Figure 2 shows the means of amplitude differences between homologous electrodes in the right and left hemispheres. Positive values of statistically significant differences reflect larger P3a amplitudes in the right hemisphere.
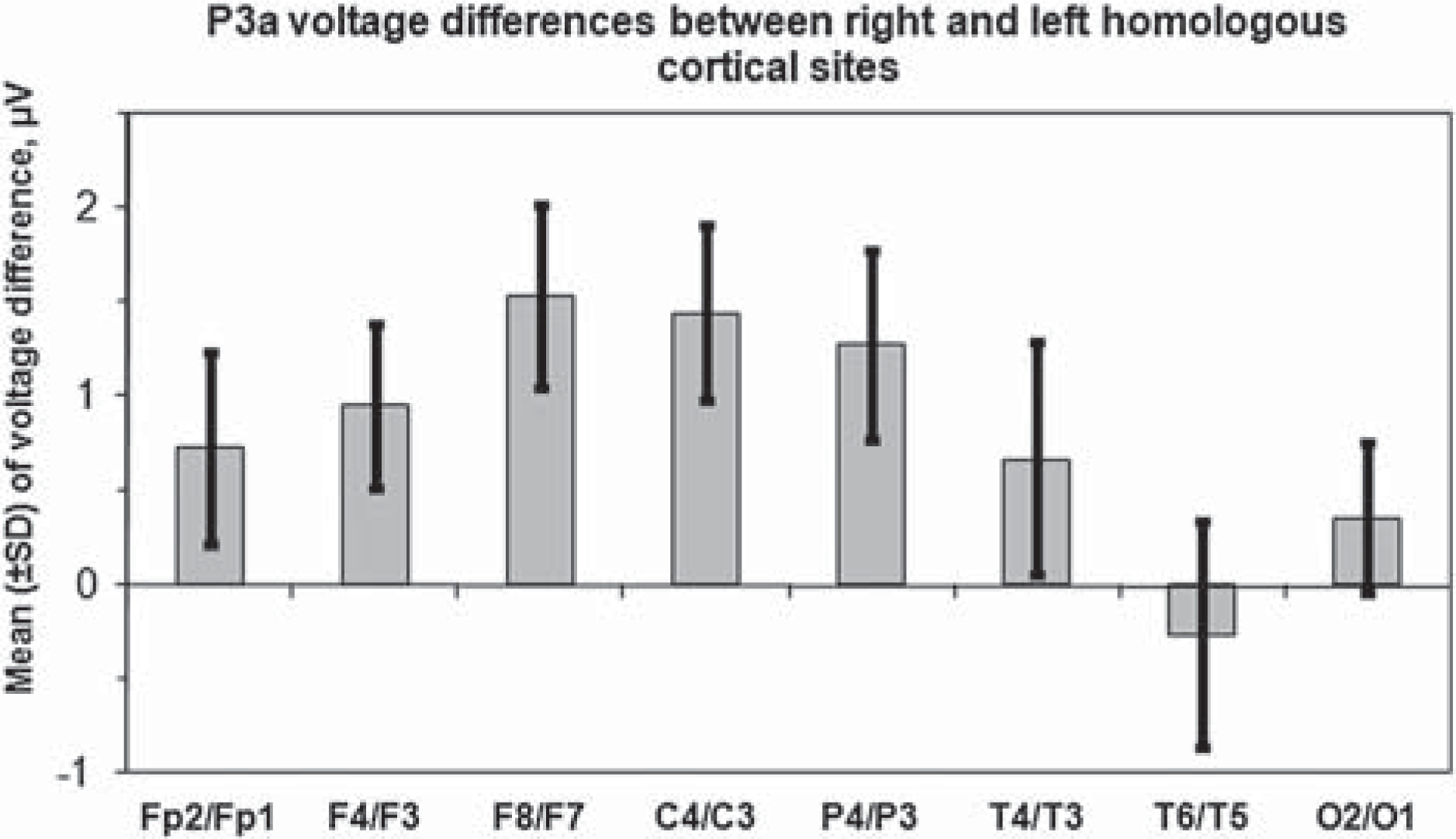
P3a voltage differences between right and left homologous cortical sites in BPD patients.
BPD versus controls results
Within-group analysis revealed that inter-hemispheric asymmetry of P3a voltage differences found in BPD patients is absent in the control group. Accordingly, the Δ A in BPD patients is significantly higher in relation to the healthy comparison subjects at F4/F3, F8/F7, C4/C3 and P4/P3 pairs of homologous sites (p < 0.001 for all pairs). At the right-hemispheric locations of these pairs, i.e. F4, F8, C4 and P4 cortical sites, and midline (Fz, Cz and Pz) cortical sites the BPD patients showed a significantly increased magnitude of P3a voltages as compared with the controls. By contrast, the P3a data from the left hemisphere demonstrated no statistically significant inter-group changes of P3a voltages. Major characteristic aspects of this hemispheric asymmetry of P3a voltages are presented in Table 1 for frontal recording sites. In the control group the P3a voltages are nearly equal at F4/F3 and F8/F7 homologous cortical sites. By contrast, in BPD patients the P3a amplitudes at the same pairs of cortical sites show significant asymmetry with larger voltages in the right hemisphere. A non-parametric Mann-Whitney U-test demonstrates a highly significant increase of P3a voltages in BPD patients as compared with controls at F4 (p < 0.001) and F8 (p < 0.002) in the right hemisphere. The increased P3a amplitude in BPD patients is also seen at the middle site Fz (p < 0.001).
Comparisons of P3a voltages of BPD patients and normal subjects at frontal recording sites
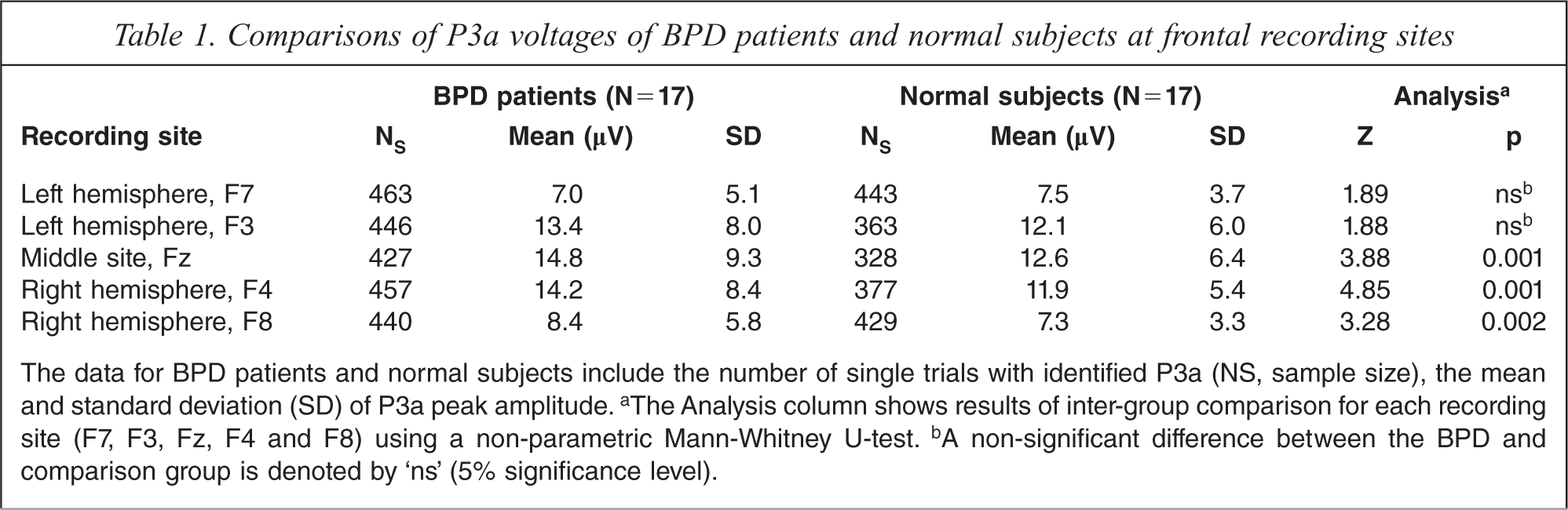
The data for BPD patients and normal subjects include the number of single trials with identified P3a (NS, sample size), the mean and standard deviation (SD) of P3a peak amplitude.a The Analysis column shows results of inter-group comparison for each recording site (F7, F3, Fz, F4 and F8) using a non-parametric Mann-Whitney U-test.b A non-significant difference between the BPD and comparison group is denoted by ‘ns’ (5% significance level).
Habituation data
The tests of habituation were separately applied to P3a and P3b for each individual electrode site. The regression analysis conducted for both groups revealed no statistically significant effect of target stimulus repetition on ERP amplitude in the group of BPD patients.
Similar results were obtained by regression analysis of P3b in normal subjects. By contrast, the P3a data in these subjects revealed statistically significant voltage decline (5% significance level) at nearly all recording sites with the exception of Fp1, Fp2, F7, F8 and O1. The decline rates (regression coefficient β in microvolt per stimulus), sample sizes (N) and significance levels (p) at midline Fz, Cz and Pz recording sites were −0.088 (N = 328, p < 0.01), −0.106 (363, 0.002) and −0.103 (390, 0.01), respectively. The sample size corresponds to the number of trials in which the P3a was identified.
Comparison of habituation on homologous electrodes revealed no significant effect of lateralization on a decline rate.
Discussion
In this study, borderline patients were shown to differ from controls in two ways. First, the BPD patients show significant larger P3a amplitudes in the right hemisphere, particularly frontally and centrally, than in the left. On the other hand, there were no significant differences between BPD and controls in P3a amplitudes at electrode sites in the left hemisphere (Table 1). Secondly, unlike controls, in whom habituation of P3a is evident, habituation of P3a fails in borderline subjects. These findings, taken together, are suggestive of a deficiency of higher order inhibitory activity, more evident right hemispherically in BPD. These findings are consistent with observations that the right hemisphere is particularly implicated in inhibitory control [41] and that failure of inhibition in a social context is significantly associated with brain lesions leading to ‘dysfunction of orbitofrontal and basotemporal cortices of the right hemisphere’ [42].
The findings regarding the large P3a amplitudes exhibited by BPD subjects give support to the possibility that BPD symptomatology is the manifestation of a certain kind of maturational deficit. The capacity to inhibit responses grows with maturation [43,44]. Young subjects without psychiatric disability have large P3a amplitudes that decline with age [37].
Posner and his colleagues [45] propose an attentional basis of BPD with a disturbance of a ‘specific circuit’ underpinning its phenomena. The particularity of the electrophysiological disturbance in our ERP data suggests that large P3a amplitudes may be a marker of this circuit. P3a appears in the time window 240−300 ms post stimulus. This is the only time window in which significant differences between components of ERP in BPD and controls were shown in this study. In particular, P3b was unaffected.
Posner and his colleagues concluded that a disturbance in attentional control which they demonstrated in their BPD subjects is related to a failure in a system involved in emotional regulation. A relationship between emotional processing and attentional systems, including novelty detection as reflected by P3a, is evident on consideration of the way in which ‘meaning’ is attributed to novel stimuli.
Such attributions extend beyond the fundamental distinction between ‘familiar/strange’ which is the basis of the orientating/habituation response, to include a wide range of meanings necessary to the experience of ‘self’. They depend upon a complex matching process in which an event in the environment is compared with models of similar events stored in memory, including very recent memory. The hippocampus is a crucial element in this comparison [46,47]. The emotional state which arises from this comparison, and of which the individual may be barely aware, determines the judgement of response. As Damasio and his colleagues have frequently pointed out, evaluation of the significance of stimuli is not simply a matter of cognition but includes emotional processing. They emphasize the significant role of the orbito-frontal cortex in this processing [48]. The orbitofrontal cortex (OFC) is involved in the inhibitory control and modulation of emotion [49].
Different forms of matching and mismatching trigger various emotions. Mismatch might evoke such feeling-states as apprehension, alarm, or mere interest and alertness. A matching response will evoke different emotions, for example familiarity, security, or pleasure.
A gathering body of evidence suggest that the child who is at risk for developing BPD has been exposed to a social environment involving excessive mismatching. This is suggested by studies which show that disorganized attachment in children is an important prediction for future BPD support this inference [50–53]. The mothering figure's responses in the creation of disorganized attachment produce repeated mismatches with her child. The responses are typically unpredictable, frightening, frightened, or unavailable to the child's attachment overtures [54]. The effect upon the child can be seen as one of repeated minor ‘shocks’, or cumulative miniature traumata. In displaying disorganized attachment, the child at times looks as if stunned, akin to the dissociated state of a traumatized adult. This is a form of ‘relational trauma’ [55] additional to any physical or sexual abuse which may have been inflicted upon the child. Abuse is very common in the childhood of those who suffer BPD but is not universal [56]. Nor, of course, is it unique to BPD.
Our study is limited in that it does not allow for a distinction to be made between the effects of matching and mismatching. A recent study suggested that they may be distinct although related processes, with the left side responding to mismatches and the right to matching [57], a suggestion resonant with a similar proposal from Hughlings Jackson, who remarked upon ‘what is most fundamental in metal operations – the double process of tracing relations of likeness and unlikeness’ [58, p.129]. Relations of likeness underlie the use of metaphor which depends upon right brain activity [59].
Our hypotheses imply that matching of a certain kind promotes maturation while mismatching impedes it. The former proposal has gained influence in recent years due to the work of researchers such as Trevarthen [60] and Stern [61]. Their observations suggest that the matching necessary to maturation has characteristic qualities. It involves the mother using her facial expression and tone of voice to resonate with her child's immediate reality. This kind of matching is not imitation. For example, the temporal contour of a maternal non-verbal vocalization might match, in a way that gives a sense of fit, what she sees in her infant's face. This kind of matching is ‘analogical’ [16]. An analogue of a thing is another thing which is like it but not a copy.
Mother–child interplay involving iterative analogical responding goes on only for a time and only during periods of infantile non-distress or positive affect. Trevarthen called it a proto-conversation [15]. It is likely to be crucial to the maturation of ‘self’. Maternal responses during periods of infantile non-distress predict the emergence of symbolic play [62] which is seen as the necessary and immediate precursor of the child's attainment of the reflective awareness of inner events [63] at about four years of age [64]. Maternal responding during periods of non-distress does not predict the later exhibition of symbolic play.
The right hemisphere seems to be peculiarly set up for the proto-conversation. Recognition of faces [65], and especially the emotions they represent [66–69], is largely right-hemispherical, as is the processing of prosody [70] and rhythm [71]. P3a, evoked by auditory stimuli is sensitive to changes in rhythm [71].
Maturation of the right hemisphere may be particularly vulnerable during the first years of life. The right hemisphere is in a growth spurt during the first two years of life, a period of right brain dominance, and so right hemisphere resources are the first to develop [72,73]. This growth is not totally encoded in the genome, but is indelibly shaped by experiences with the environment. The neurobiological maturation of the emotion processing right hemisphere in the early critical period of the first two years of life and is thus ‘experience-dependent’. It is specifically the affect communicating and analogically representing interplay between mother and child involving prosody, facial expressions and rhythm that impact the experience-dependent maturation of prefrontal cortical-limbic circuits of the early developing right cortical hemisphere [74]. The baby's right brain seems to be particularly set up to respond to the mother's face. For example, babies of 4–9 months move their eyes more quickly to the mother's face than a stranger's face when the stimuli are presented in the left visual field. In contrast, simple geometric shapes are discriminated equally well in either field [75, p.1046]. Lyons and colleagues demonstrate that varying maternal behaviours in infancy produce ‘significant differences in right but not left adult prefrontal volumes, with experience-dependent asymmetric variation most clearly expressed in ventral medial cortex measured in vivo by magnetic resonance imaging’ [76, p.51]. A deficit in appropriate responding may produce the structural changes reported by Chanen et al. [77] in a group of first presentation BPD patients in whom secondary effects of the disorder over time were likely to be minimized. They showed right sided OFC loss of grey matter related to controls but no hippocampal or amygdaloid difference.
It seems important that the interplay of the proto-conversation involves neurophysiological activation of a similar kind in both partners. For example, affective prosody is not only processed right hemispherically but is also delivered as a consequence of right brain activation [78]. It might be said then, in a partially figurative way, that the proto-conversation represents an interplay between two right brains. In the case of the mother, at least, OFC activation is prominent. It occurs when mothers view pictures of their babies [79]. The OFC, periaqueductal grey matter, anterior insula, and dorsal and ventrolateral putamen become active when a mother views her baby smiling at the mother while playing with her [80]. Viewing her infant's distress evokes a different neural response. The smile is likely to enhance activation in the medial OFC beyond that evoked by the image of the baby alone [81]. Such observations lead to the speculation that the analogical matching behaviour of the mother during the proto-conversation might evoke a mirrored neurophysiology in her baby and tend to stimulate activation and maturation of circuitry involving the OFC. The observations of a study from Minagawa-Kawai et al. [82] support the possibility of such reciprocity. They showed OFC activation in the brains of mothers and babies when shown a video of the other. In the mother's case, the activation was right-sided.
These observations lead to implications for treatment of BPD designed to facilitate the emergence of reflective awareness of inner events, a state which Bateman and Fonagy [14] call ‘mentalization’. This kind of consciousness does not appear until fairly late in child development, at about age four. It arises from an earlier and more fundamental state of mind which is developing during a period when the right hemisphere is dominant. The data presented in this study suggest this development has been impeded in those who suffer BPD. In order for the reflective capacity to reveal itself the foundation of the earlier kind of consciousness must be made. This suggests that a major role is played, particularly in early phases of treatment, by an engagement between therapist and patient which depends upon a conversation in which the elements of the emotional, non-verbal and analogical ‘language’ of the right hemisphere are prominent. Clinical evidence gathered during studies of the effect of the conversational model of psychotherapy [9,20,83] suggests that this kind of relatedness allows higher order consciousness to come forth in those with BPD in a natural and spontaneous way [84,16].
Finally, although diminished amplitude of P3 is a very uncommon finding in psychiatric illness, this study is limited by the absence of control groups with Axis I disorder. BPD is comorbid with all major Axis I disorders [5]. It is possible that a comorbid condition not identified in our study is the basis of the P3a findings. Subsequent studies are needed on depression and, most particularly, dissociative disorders. Furthermore, future investigation should involve, for each patient, careful and extensive phenomenological examination together with a history of the developmental background, since not all BPD subjects may show the findings which were characteristic of our group of unmedicated patients.
Footnotes
Acknowledgements
