Abstract
Estimates of the numbers of people with dementia in Australia range between 190 000 [1] and 200 000 [2] (around 1% of the population), of whom 64% are female and 81% are aged 75 or over [1]. It is projected that this number will reach 2.8% of the Australian population by 2050 [2]. Alzheimer's disease (AD) is the most common form of dementia accounting for between 50% and 70% of all cases [3]. It is a progressive, degenerative illness affecting brain function. Three anticholinesterases are approved and funded under the Pharmaceutical Benefits Scheme (PBS) Australia for the treatment of mild to moderately severe AD: donepezil hydrochloride, galantamine hydrobromide and rivastigmine hydrogen tartrate. Systematic reviews of randomized controlled trials on the use of anticholinesterases have shown that they have small to modest clinical benefits for patients with mild to moderate AD [4–6]. They may temporarily delay progression and improve symptoms but they do not treat the disease [7]. Guidelines suggest that patients who do not stabilize or improve within the first few months of therapy are unlikely to have any subsequent benefit [8]. The PBS in Australia allows subsidy of an initial treatment with anticholinesterases for up to 6 months in documented mild to moderately severe disease. Continuing the therapy after that time requires demonstrated improvement in cognitive function measured by: (i) an increase from baseline score on the mini-mental state examination or standardized mini-mental state examination; (ii) a decrease on the AD assessment scale, cognitive subscale; (iii) rating of ‘very much improved’ or ‘much improved’ on the clinicians interview based impression of change scale [9]. If the patient does not respond to the treatment or progresses to severe dementia then it is recommended that the therapy should be discontinued as the risk of adverse effects makes it inadvisable to continue [10].
This research aimed to determine the duration of anticholinesterase use for AD among the Australian veteran population. We examined patterns of therapy discontinuation. The National Prescribing Service commenced a program in August 2008 informing prescribers of the small benefits of anticholinesterases and the relatively poor response rates [10]. This study explored methods to evaluate the program and provide baseline rates of use.
Methods
Data source and cohort definition
This was a retrospective cohort study utilizing de-identified pharmacy claims data provided by the Department of Veterans’ Affairs, Australia. The pharmacy claims dataset provided patient-level information on all medicines (subsidized under the PBS/Repatriation Pharmaceutical Benefits Scheme in Australia) dispensed to veterans including the drug dispensed, the date of dispensing, quantity supplied, the dosage form and strength, and patient information (age, gender, date of death, and residential status).
The study population was restricted to veterans aged 65 years and over who were dispensed anticholinesterases (donepezil, galantamine or rivastigmine) during the 4-year index period. The study cohort was defined as veterans with an index (initial) prescription for an anticholinesterase medicine between January 2003 and December 2006 and who had no dispensing of any anticholinesterase medicine in the 12 months prior to their index prescription. The study end point was time to discontinuation. These newly treated patients were followed up until treatment cessation, switching to another therapy, death or 12 months post index prescription. For those with a gap of 99 days, we ascertained the proportion who reinitiated therapy after cessation by determining the date of the next prescription for any anticholinesterase any time in the study period (up until December 2007).
Data analysis
The data extraction and analysis were performed using SAS software (version 9.1; SAS Institute Inc, Cary, NC, USA). Anticholinesterases were identified by Anatomical Therapeutic Chemical (ATC) code N06DA. Treatment cessation was defined as a gap in refill for the index medicine of 99 days or more. This was based on the assumption that each anticholinesterase prescription lasts for 33 days. The time period of 33 days was calculated from the data and represented the number of days in which 75% of anticholinesterase prescriptions were refilled. A change or switch in treatment was defined as a dispensing of an anticholinesterase different from the index treatment. Time to treatment discontinuation was calculated as the number of days on the index treatment to first treatment cessation or to dispensing of a new anticholinesterase, whichever occurred first within the follow-up period.
Statistical analysis
The cohort was divided by the type of the index anticholinesterase. Patients’ characteristics of the three treatment groups (galantamine, donepezil and rivastigmine) were compared using t-tests (for means) and χ2 tests (for frequencies). Kaplan-Meier analysis was used to estimate the cumulative duration rates. Patients were censored at the end of the follow-up period or if they died. The log rank and Wilcoxon tests were used to assess differences between the treatment groups over time. The threshold level for statistical significance was set to p ≤0.05. Cox proportional hazards models were used to compare the risk of treatment discontinuation among the treatment groups and to adjust for the effect of the patient's characteristics at study entry (age, gender and residential status). The proportional hazards assumption was checked by including time-dependent covariates in the Cox model. Results were also stratified by residential status at time of commencement of index therapy. Patients who transitioned between care settings were excluded from this stratification.
Results
The study population comprised 10 088 veterans of which 6705 (66%) were using donepezil, 2989 (30%) galantamine and 394 (4%) rivastigmine. Table 1 provides details about the age, gender and residential status of the treatment groups. The majority of patients were between 75 and 94 years of age, with a similar average age across the three treatment groups. Over half of the patients were males, 87% lived in community and 13% in residential aged care.
Characteristics of study patients
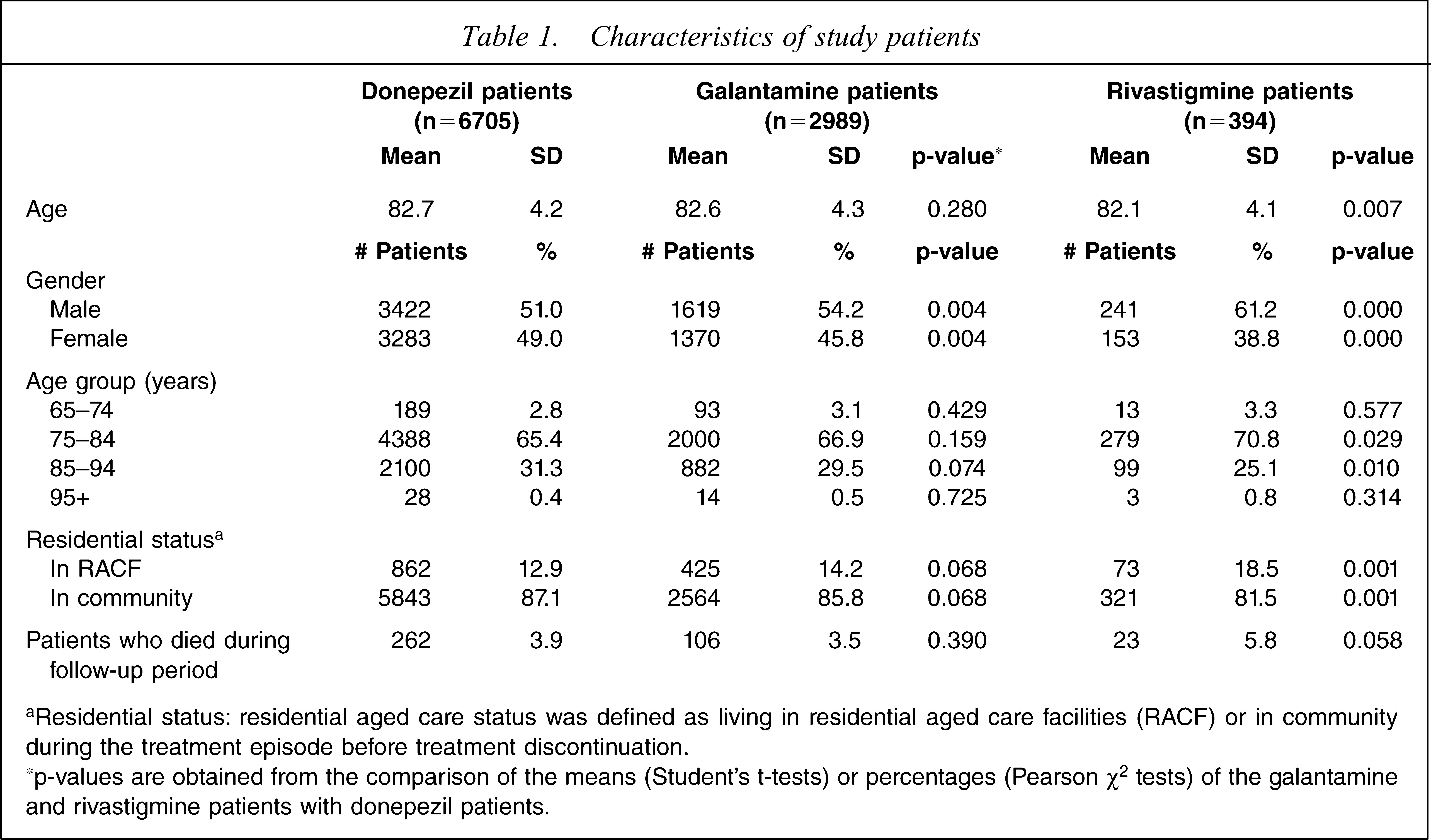
aResidential status: residential aged care status was defined as living in residential aged care facilities (RACF) or in community during the treatment episode before treatment discontinuation.
∗ p-values are obtained from the comparison of the means (Student's t-tests) or percentages (Pearson χ2 tests) of the galantamine and rivastigmine patients with donepezil patients.
The unadjusted Kaplan-Meier curves for treatment duration are presented in Figure 1. Median treatment durations for donepezil, galantamine and rivastigmine were 199 days (95% CI, 182–208), 233 days (95% CI, 212–259) and 219 days (95% CI, 176–260), respectively.
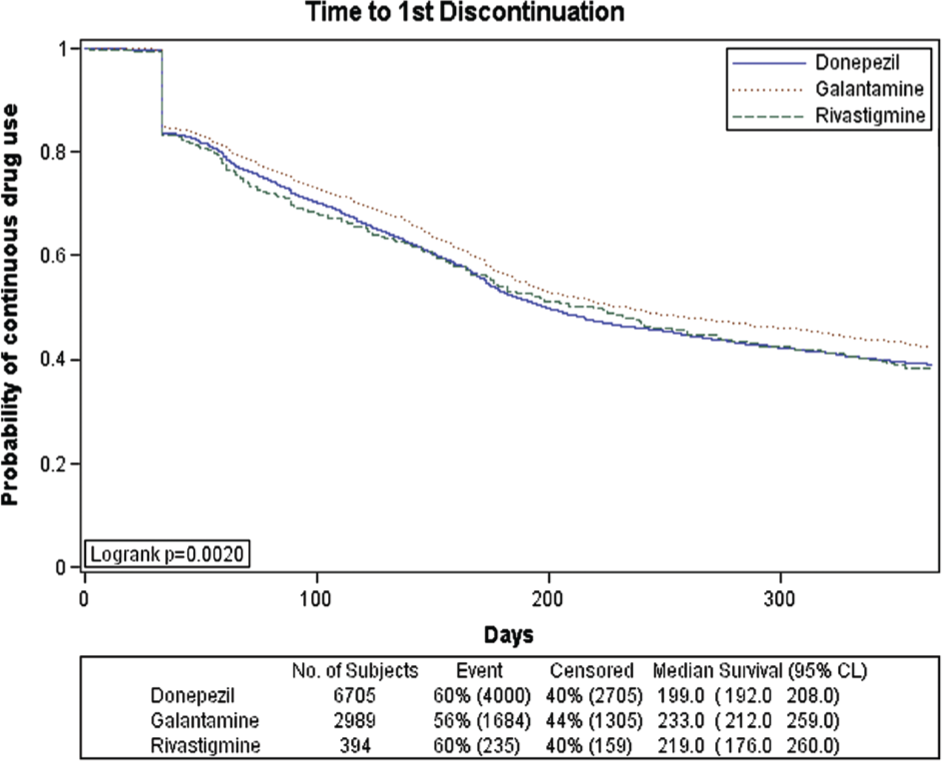
Kaplan-Meier estimates for time to treatment discontinuation of the first episode with anticholinesterases.
For patients who ceased therapy within the follow-up period (n = 5464), the median treatment duration was 104 days for donepezil, 110 for galantamine and 90 for rivastigmine users. Of these 5464 patients, 32% restarted anticholinesterase therapy before the end of study (December 2007); 28% returned to the same index medication after a median time of 138 days (95% CI, 132–144); only 4% restarted a new anticholinesterase after a median time of 194 days (95% CI, 166–219). Of those who restarted therapy, 66% restarted on the same medication within 6 months after ceasing their index medication, 26% returned to the same therapy in 6–12 months, and only 8% in over 12 months. Little switching was observed despite approximately 78% of patients having more than 2 years of follow-up time after cessation in which switching could occur.
For patients who switched therapy within the follow up (n = 455), the median time to the switch was 97 days (95% CI, 84–111).
There was a statistically significant difference in the length of duration for galantamine patients compared to donepezil (p < 0.01), but not between galantamine and rivastigmine, (p = 0.10), nor rivastigmine and donepezil patients (p = 0.66) (Table 2). Adjusting for age, gender and residential status did not significantly change the relative risk for treatment discontinuation.
Relative risk of discontinuation of initial therapy during 1-year follow-upa
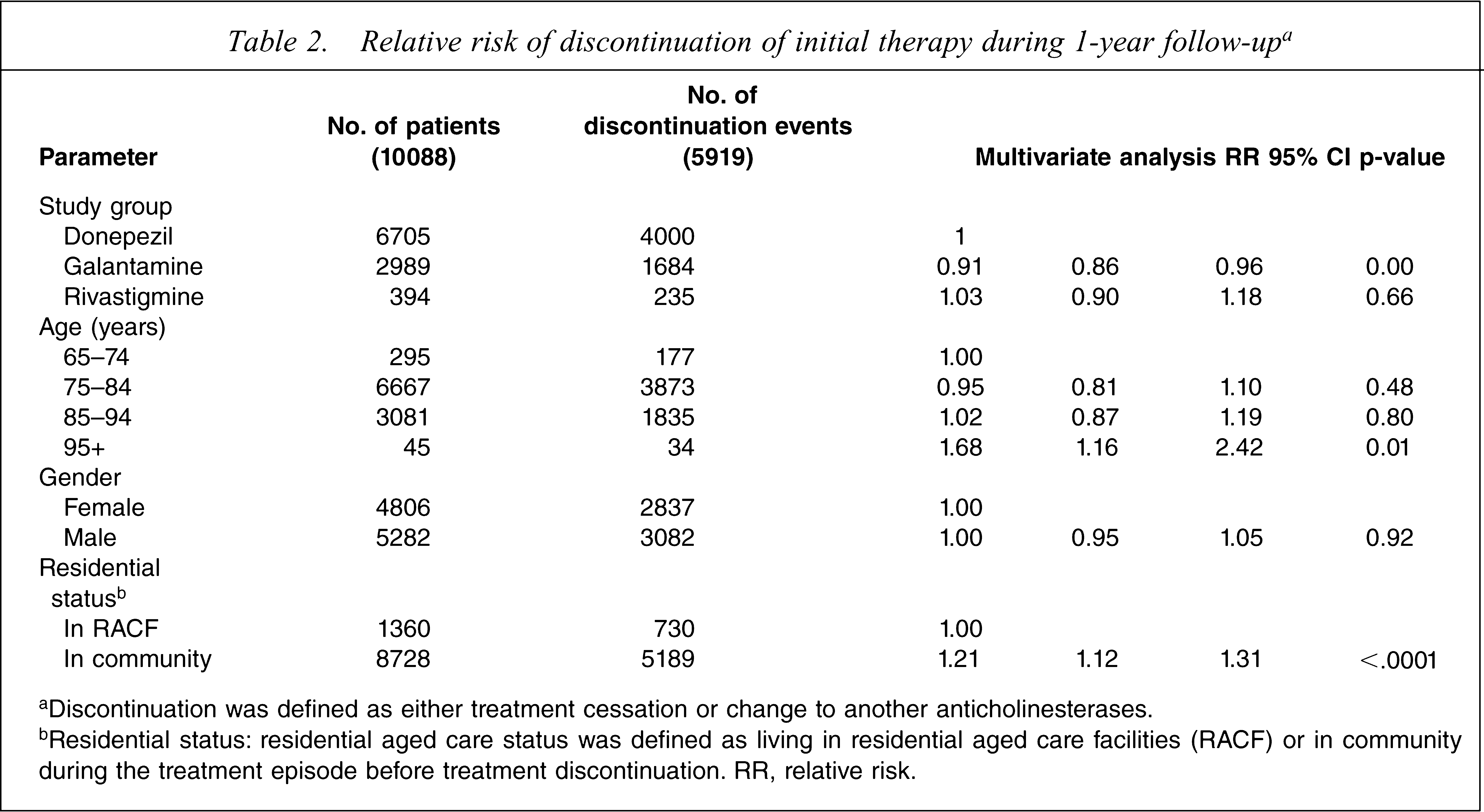
aDiscontinuation was defined as either treatment cessation or change to another anticholinesterases.
bResidential status: residential aged care status was defined as living in residential aged care facilities (RACF) or in community during the treatment episode before treatment discontinuation. RR, relative risk.
Table 3 represents Kaplan-Meier estimates percentiles for patients who discontinued anticholinesterase treatment at days 60, 120, 180 and 365. The results demonstrate that of the newly treated patients, almost half discontinued their initial therapy within 6 months, and around 60% discontinued it at 1 year.
Kaplan-Meier estimates of percentages of patients discontinuing anticholinesterase treatment
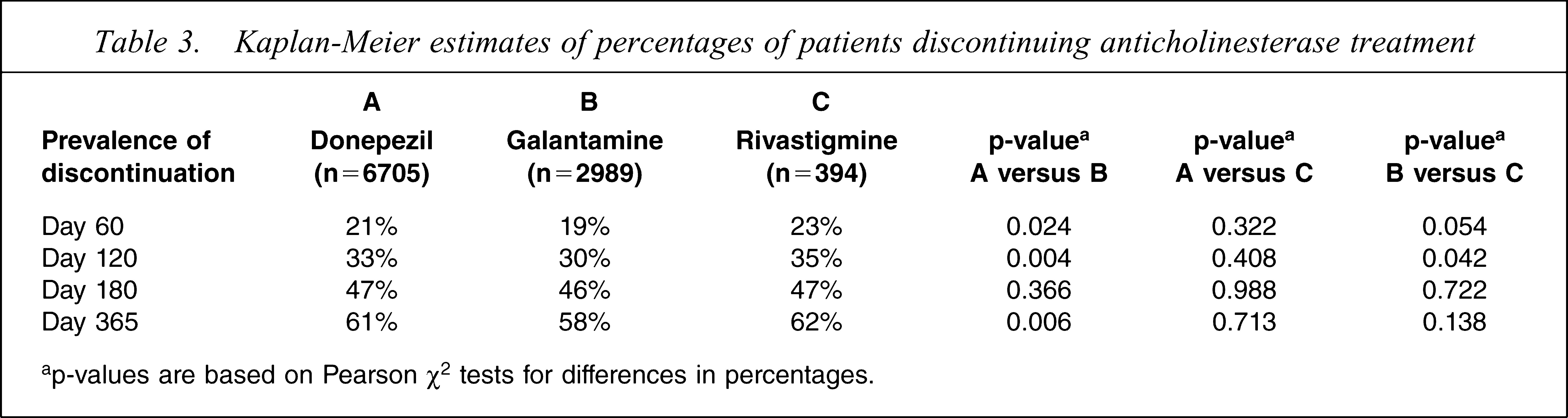
a p-values are based on Pearson χ2 tests for differences in percentages.
The unadjusted Kaplan-Meier curves for treatment duration stratified by residential status are presented in Figure 2. Median treatment duration for patients living in community is 199 days (95% CI, 192–208) compared to 273 days (95% CI, 238–312) for patients in residential aged care facilities (log-rank test, p < 0.0001).
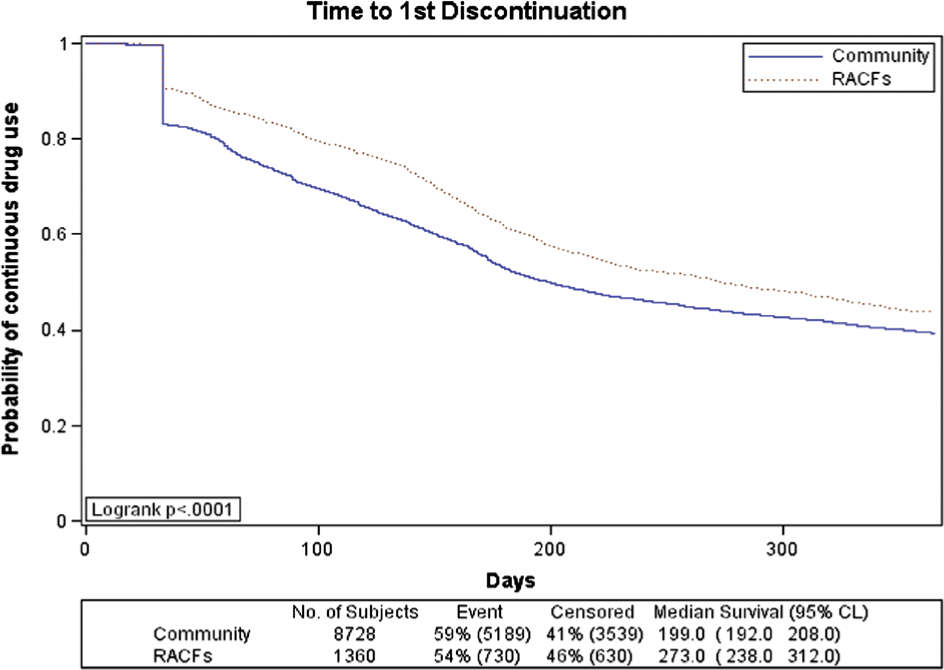
Kaplan-Meier estimates for time to treatment discontinuation of the first episode with anticholinesterases stratified by residential status during the first episode.
Discussion
This study found that almost half of the veteran patients in Australia discontinued their anticholinesterase therapy within 6 months of initiation and approximately 60% discontinued it at 1 year. However, 32% of those who ceased, restarted anticholinsterase therapy and the majority of those who restarted, did so on the same therapy. The study suggests duration rates were similar between the three medicines. While statistically there were slightly higher continuation rates with galantamine, the difference was small and not observed at 180 days post initiation. Patients in community settings were more likely to discontinue treatment earlier than patients living at residential aged care facilities. Clinical trials have shown that patients respond differently to anticholinesterase treatment and only about one in four patients benefit in any ‘clinically meaningful’ way from the therapy [4–6,11]. Our results suggest that more than one in four ceased therapy, but due to the high re-initiation rate on the same therapy within a short time (less than 6 months), this cessation may be due to non-persistence rather than therapeutic failure. Furthermore, it appeared that community-dwelling residents discontinued their initial therapy earlier than those in residential aged care facilities. This suggests that quality use of medicine strategies for those living in the community with dementia should focus on encouraging persistence where treatment is effective. Of those who ceased therapy, 70% did not reinitiate it during the study period, which may indicate that the PBS criteria do facilitate cessation where therapeutic failure occurs.
Our results of 45% discontinuation rates at 6 months for all three anticholinesterase inhibitors are similar to another Australian study in the elderly (including non-veterans) that found cumulative discontinuation rates of 52% for donepezil, 45% for galantamine, and 56% for rivastigmine at 6 months after initial drug supply [12].
Our study is comparable in terms of patient selection and the definition of treatment discontinuation with a US study [13] involving only 783 patients with Alzheimer's diagnosis. Our results suggest discontinuation 3–4 months earlier for the Australian veteran population compared to the US patient sample [13]. The median duration for donepezil in our study was 199 days compared to 315 in the US study, and 219 days for rivastigmine compared to 312. While 47% of Australian veterans on donepezil discontinued treatment at 180 days, only 37% of similar new donepezil users in the US discontinued it at day 180 according to another study [14]. The longer duration of treatment in US might be due to the absence of an explicit improvement requirement in their guidelines; the decision on whether to continue therapy in the US studied population was based on patient satisfaction, which can include maintenance of current mental status and prevention of mental decline [15].
A limitation of our study is that the results were based on prescription claims data which did not provide: (i) direct diagnostic information to confirm the AD diagnoses; (ii) clinical information on treatment side effects or failure to improve, which could explain early treatment discontinuation in some cases. The Department of Veterans’ Affairs authority system for prescribing anticholinesterases requires direct diagnostic information and it can be assumed that the dispensings were most likely for AD, but we cannot rule out that people did not have other diagnoses.
The veteran population have slightly more general practice visits (rate ratio 1.17; p < 0.05) and hospitalizations (rate ratio 1.21; p < 0.05) per year than other Australians aged 40 years and over [16]. However, veterans with no service-related disability have similar levels of use to the Australian population [16]. Similar numbers of prescriptions per general practitioner visit are observed between the veteran population and the Australian population; however, because of the higher rate of general practitioner visits, veterans receive slightly more prescriptions annually than other Australians (rate ratio 1.13; p < 0.05) [16]. This suggests that our study results are likely to be similar to what occurred in the Australian population, but may slightly overestimate the effect.
Conclusions
The results of this study demonstrate that about half of new veteran users of AD medicines ceased or switched their initial therapy within 6 months. Of those who ceased therapy within the 1 year follow up, 28% returned to the same index medication and 4% restarted on a different anticholinesterase before the end of study. Patients on anticholinesterases living in the community discontinue their therapy approximately 2.5 months earlier than patients living in residential aged care facilities.
Footnotes
Acknowledgements
This study was funded by National Prescribing Service Ltd, Australia. We acknowledge the support of the Department of Veterans' Affairs which provided data for the conduct of these analyses.
