Abstract
Background:
Vagus nerve stimulation (VNS) is a recognized palliative surgical therapy for drug-resistant epilepsy (DRE). However, optimal postoperative parameter adjustment strategies to maximize efficacy remain an area of active investigation, particularly concerning different etiologies and seizure types. This study was designed to explore the therapeutic efficacy of VNS and postoperative parameter adjustment in patients with DRE.
Methods:
Eighteen patients with DRE who underwent VNS implantation were retrospectively analyzed. We included patients with focal DRE unsuitable for craniotomy, encephalitis-related DRE, and idiopathic DRE with no identifiable cause. Postoperative stimulation parameters were precisely individualized and adjusted. The efficacy of VNS in controlling seizures was evaluated using the McHugh classification.
Results:
Eight patients achieved McHugh class IA efficacy (seizure frequency reduction > 80%), seven achieved class IIA efficacy (> 50% reduction), and three achieved class III efficacy (< 50% reduction; IIIA in two and IIIB in one).
Conclusion:
VNS demonstrated significant therapeutic efficacy in patients with DRE, particularly those with a structural etiology and generalized tonic–clonic seizures. The effect was especially notable in post-traumatic DRE. Postoperative current adjustment proved more complex and influential compared with pulse width and frequency modifications and had a greater impact on seizure control.
1 Introduction
Epilepsy is a chronic brain disorder with multiple etiologies. It is characterized by recurrent, paroxysmal, and transient disturbances of the central nervous system resulting from excessive discharges of cerebral neurons [1, 2 ], and is one of the most common neurological diseases. In China, the prevalence of epilepsy is approximately 0.7%, with a male-to-female ratio of approximately 1.15–1.7 to 1. Currently, there are approximately 10 million patients with epilepsy in China [3].

Most epileptic seizures can be controlled using a single antiepileptic drug (AED). However, between 20% and 30% of patients require two or more AEDs for seizure control. In some patients, seizures cannot be effectively controlled even with multiple drug combinations. This condition is referred to as drug-resistant epilepsy (DRE) [4]. For these patients, surgical intervention has become the ultimate treatment option.
Among surgical treatments, vagus nerve stimulation (VNS) is a commonly used approach. The method aims to reduce seizures and treat comorbidities by stimulating the vagus nerve in the neck. Approximately 50% of DRE patients achieve a > 50% reduction in seizures with VNS therapy, and complete seizure freedom is achieved in up to 9% [5]. Owing to its definite efficacy and relatively few adverse reactions, VNS has been increasingly applied in clinical practice.
This study retrospectively analyzed 18 patients with DRE who underwent VNS treatment in our hospital from August 2019 to April 2023 and were clinically followed through August 2025. We examined the efficacy of VNS and the postoperative adjustment of stimulation parameters. Our findings suggest that setting appropriate stimulation parameters for DRE patients based on seizure etiology, disease duration, and medication status can effectively control seizures.
2 Materials and Methods
2.1 General Information
This study included 18 patients with DRE: 12 male and six female. Fifteen were adults and three were children. Mean age was 29.22 ± 12.24 years (range, 7–55 years). Mean disease duration was 12.50 ± 6.80 years (range, 3–26 years).
Among these patients, six had a history of traumatic brain injury, all with varying degrees of cerebral contusion and laceration. Four had undergone craniotomy. Seizure type included 4 cases starting as partial seizures evolving into generalized tonic-clonic seizures (GTCS) and 2 cases with initial GTCS onset. Two patients had a history of encephalitis, one of whom presented with partial seizures evolving into GTCS and the other with GTCS as the initial seizure type. Ten patients had no obvious cause of epilepsy, and all presented initially with GTCS.
All patients had received standardized medical treatment for more than 3 years, with two to four types of AEDs. Seizure frequency ranged from at least once per week to more than six times weekly.All patients and their family provided written informed consent at the time of entering this study.The study was approved by the Medical Ethics Committee of The Third People’s Hospital of Bengbu(Ethics Approval Number: Lunke PI Zi [2020] No. 40)
2.2 Case Selection
Inclusion criteria: Patients who met the diagnostic criteria for DRE published by the International League Against Epilepsy in 2017, including (1) Partial seizures with or without secondary generalization; (2) Inadequate seizure control after adequate dosage and duration of at least two appropriately chosen and tolerated AEDs (monotherapy or polytherapy); (3) No surgically remediable etiology identified, or etiological treatment failed; (4) Multiple epileptogenic foci or poorly localized lesions not suitable for craniotomy, or failed resective surgery [6, 7 ]; (5) Previous craniotomy but persistent seizures; (6) Age ≥ 7 years.
Exclusion criteria: (1) Epilepsy caused by an intracranial space-occupying lesion as indicated by magnetic resonance imaging; (2) Progressive neurological disorder; (3) History of bilateral vagus nerve injury or transection; (4) Active infection at the implantation site; (5) High risk of implant rejection; (6) Poor general condition, unable to tolerate surgery; (7) Psychiatric disorder; (8) Cardiac arrhythmia; (9) Peptic ulcer; (10) Brain tumor or other systemic condition compromising overall health.
2.3 Surgical Methods
Twelve patients received a VNS device manufactured by Pins Medical (Beijing, China), and six received a system manufactured by Cyberonics (Houston, USA).
Procedure: Under general anesthesia, patients were placed supine with the left shoulder elevated and the head turned right and slightly extended to fully expose the surgical site. A transverse incision was made between the left clavicle and mastoid process along the anterior border of the sternocleidomastoid muscle. The carotid sheath was dissected, exposing approximately 3 cm of the vagus nerve between the carotid artery and internal jugular vein.
A transverse subclavicular incision was then made, and a subcutaneous pocket was created for the pulse generator. A tunneling rod was used to pass the lead subcutaneously from the cervical incision to the subclavicular pocket. The helical electrode contacts were anchored onto the exposed vagus nerve, with the negative contact positioned proximally, and connected to the pulse generator. Device testing confirmed normal parameters and lead function. The wounds were closed in layers and dressing applied.
Perioperative outcomes: No infections, hemorrhage, or neurovascular injury occurred. Two patients experienced transient hoarseness, which resolved within 2 months. No other complications were observed.
2.4 Device Activation and Parameter Adjustment
Stimulation was initiated 2 to 3 weeks after surgery. Parameters were adjusted as follows: (1) Initial adjustment was performed every 2 to 3 weeks, with current increased by 0.1 to 0.25 mA per adjustment; (2) Subsequently, adjustments were performed every 1 to 3 months depending on the patient’s condition; (3) The average number of adjustments was 9.22 ± 4.05.
Default stimulation cycle: 30 seconds ON, 5 minutes OFF. Frequency: 20 to 30 Hz. Pulse width: 125 to 500 μs. Current started at 0.20 mA and was gradually increased, not exceeding 3.0 mA.
Magnet-triggered stimulation was set one step higher than cyclic stimulation, lasting 30 to 60 seconds, with frequency and pulse width unchanged.
2.5 Follow-up
All 18 patients were followed for 30 to 72 months. During the initial adjustment phase, AED regimens were not modified. As seizure reduction was achieved through VNS, the number and type of AEDs were gradually adjusted.
2.6 Efficacy Evaluation
Therapeutic efficacy was evaluated using the McHugh classification system (Table 1) [8].
Evaluation criteria for efficacy of vagal nerve stimulation (McHugh, 2007)

3 Results
3.1 Clinical Data and Postoperative Parameter Adjustment
The clinical data and postoperative parameter adjustment outcomes of 18 patients with DRE are shown in Table 2.
Clinical data and postoperative parameter adjustment
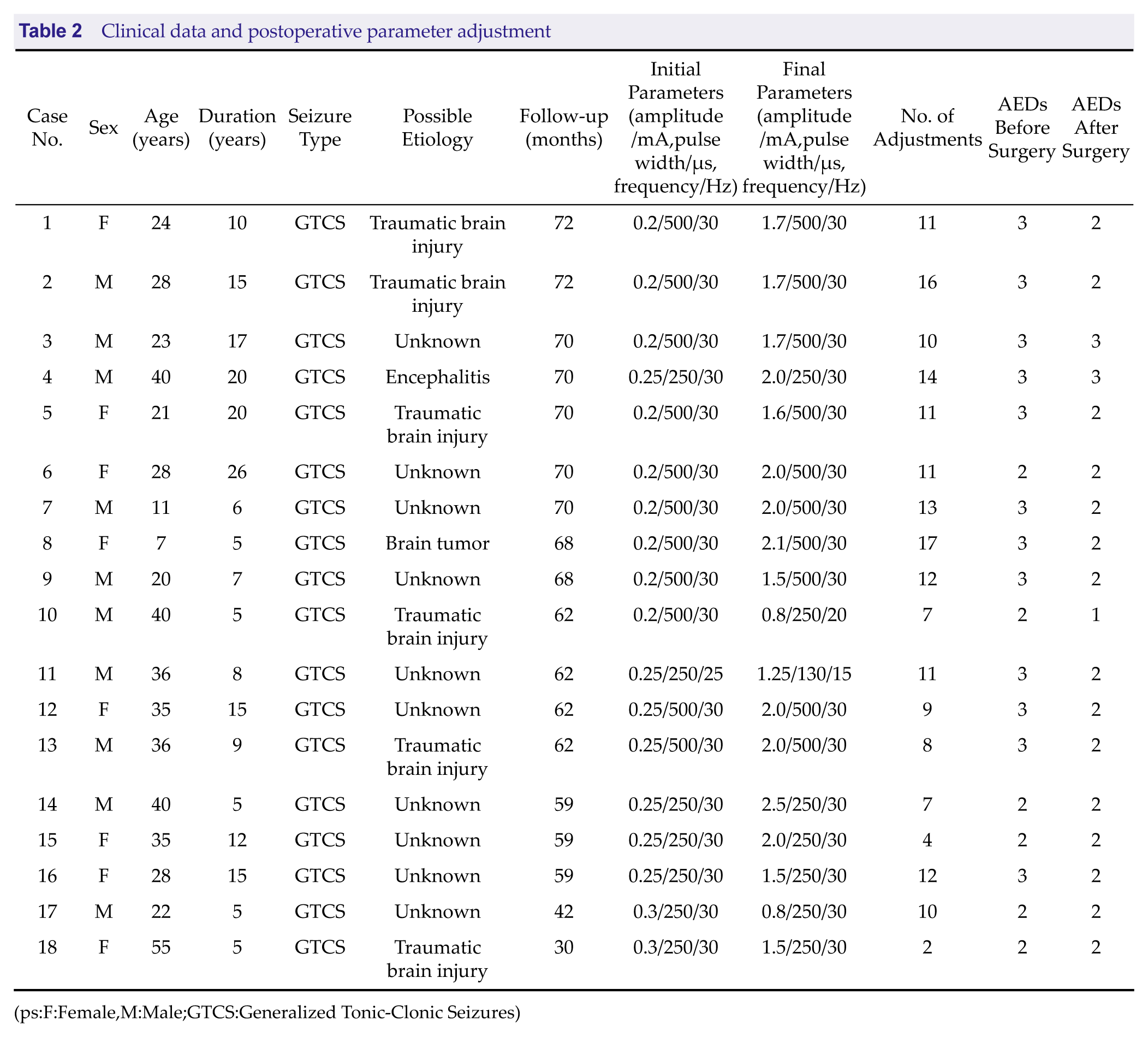
(ps:F:Female,M:Male;GTCS:Generalized Tonic-Clonic Seizures)
3.2 Clinical Efficacy and Adverse Reactions
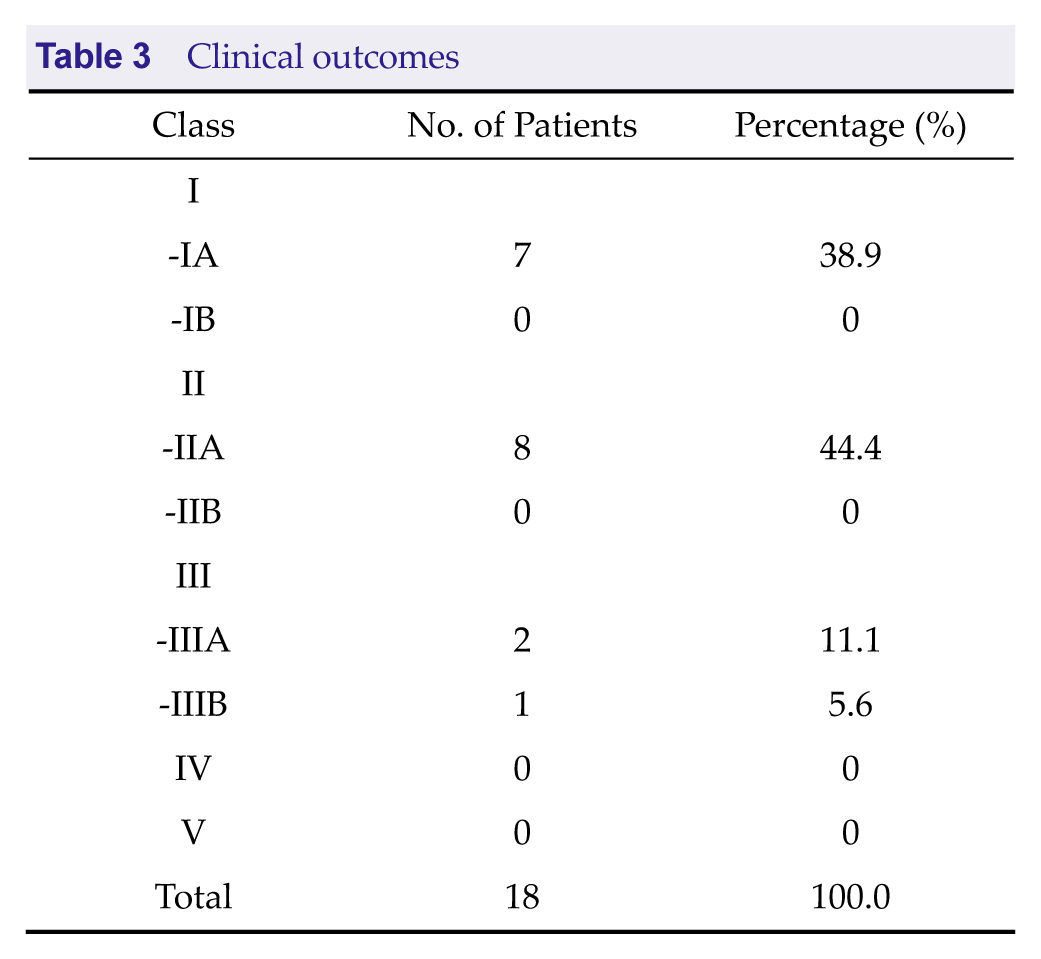
As shown in Table 3, clinical efficacy was classified as class I/II in 15 patients (83.3%) and class III in the remaining 3 (16.7%). An adverse reaction occurred in two patients (11.1%), hoarseness in both.
Clinical outcomes
4 Discussion
Patients with DRE experience long-term seizures, which not only impose a heavy financial burden, but also cause severe impairment of cognitive function, and negatively affect daily life and work. Common surgical treatments for DRE include resection of epileptogenic foci, corpus callosotomy, functional hemispherectomy, multiple subpial transections, and cortical thermocoagulation. However, because these procedures require craniotomy, patients are often unwilling or unable to accept them. VNS is a minimally invasive neuromodulation procedure that delivers intermittent, low-intensity electrical pulses to the vagus nerve in the neck, thereby suppressing abnormal neuronal discharges and controlling seizures.
In recent years, numerous clinical studies have confirmed that VNS, as an adjunctive therapy for DRE patients, can effectively reduce seizure frequency and severity, and can also improve cognitive function [9-13 ]. With further progress in epilepsy research, it has been found that if the epileptogenic network (i.e., seizure onset zone, origin, and abnormal hub nodes) is not simultaneously removed, network connections cannot be altered and prognosis remains poor [14]. VNS, through the pulse generator, stimulates afferent impulses to the cervical vagus nerve, inhibits abnormal synchronized discharges of neurons, and prevents the spread of epileptic networks, thereby reducing or terminating seizures [15]. Lanzone et al. [16] studied differences in brain networks of patients with DRE before, during, and after VNS using electroencephalography, and the results indicated that VNS acts by reducing network synchrony but has no significant effect on network spectral power. Other studies have found that VNS achieves higher seizure reduction rates in children compared with other populations; moreover, VNS can reduce brain function in patients with epilepsy, decrease the vulnerability of functional brain networks during seizures, and promote reconstruction of brain network structures [17–18 ]. Although VNS may not be the ideal choice for all patients, it nevertheless provides a useful therapeutic approach for treating DRE.
This study retrospectively analyzed the clinical efficacy of VNS in 18 patients with DRE treated at our hospital. Of these, 10 (55.6%) had idiopathic DRE, six (33.3%) had structural lesions where resection of epileptogenic foci would damage eloquent brain regions, and two (11.1%) had infectious etiologies without obvious structural abnormalities. Following multidisciplinary evaluation, all patients underwent VNS treatment. Among them, 15 were adults and 3 were children. After 30 to 72 months of follow-up, favorable outcomes were obtained. Among six patients with post-traumatic DRE due to brain tissue injury, postoperative individualized adjustment of stimulation parameters resulted in four cases of class IA efficacy and two of class IIA efficacy, with one patient achieving complete seizure freedom. Two patients with encephalitis- related DRE were followed for 60 to 70 months, and each experienced a suboptimal outcome (class IIA and class IIIA efficacy, respectively). Ten patients with idiopathic DRE were followed for 42 to 70 months, and their outcomes were class IA in four, class IIA in five, and class IIIA in one; one patient achieved complete seizure freedom, indicating good efficacy.
Eight patients (44.4%) in our cohort had > 80% seizure reduction, seven (38.9%) had > 50% reduction, and three (16.7%) had < 50% reduction. Among the six patients with post-traumatic DRE and 10 patients with idiopathic DRE, two (11.1%) achieved complete seizure freedom, which is similar to the 7% to 9% seizure-free rate reported in the literature [5]. Given its definite efficacy and relatively few adverse effects, VNS has been increasingly applied in clinical practice. Of the eight patients in whom class IA efficacy was achieved, four had post-traumatic DRE, suggesting that VNS is particularly effective in this subgroup. Emilie et al. [19] suggested that patients with structural brain lesions respond better to VNS compared with those with genetic epilepsy or idiopathic epilepsy. Englot et al. [20] conducted a meta-analysis and found that patients with post-traumatic epilepsy or tuberous sclerosis respond well to VNS. Among the 10 patients with idiopathic DRE in our study, four achieved class IA efficacy, five achieved class IIA, and only one was class IIIA, also indicating good efficacy. By contrast, none of the two encephalitis-related cases achieved class IA efficacy, suggesting that VNS may be less effective in this subgroup.
This study also found that patients with longer follow-up duration had better outcomes. This is consistent with a retrospective study of 436 cases of VNS for epilepsy, in which 255 cases (63.75%) achieved >50% seizure reduction, and seizure frequency gradually decreased with prolonged stimulation [21]. Other reports have indicated that within 1 to 5 years after VNS, the proportion of patients achieving ≥50% improvement increases annually, and the efficacy in responders tends to further enhance over time [22–24 ].
Seizure type is also an important factor influencing VNS efficacy [25]. Studies have shown that different seizure types respond differently to VNS. Compared with focal seizures, patients with generalized seizures achieve better outcomes after VNS [26]. In our study, all patients presented with GTCS, and the outcomes were consistent with previous reports.
Adverse effects of VNS may occur early or late after surgery. The most common complications are hoarseness, cough, and throat discomfort [27]. Early-onset hoarseness may persist and become a difficult-to-control long-term complication. In fact, nearly 20% of patients report it 5 years after surgery [28]. In our study, two patients developed hoarseness, but both recovered within 2 months after adjusting stimulation parameters. The incidence of adverse reactions in this study was lower than that reported in the literature, possibly owing to the small sample size.
Individualized adjustment of stimulation parameters is a key factor in the success of VNS therapy. The number of adjustments per patient in our study ranged from two to 17. Current intensity was the most frequently modified parameter, while pulse width and frequency adjustments were less common. Among the 18 patients, pulse width was 500 μs in nine, 250 μs in seven, and 130 μs in two; frequency was 30 Hz in 15 patients, 25 Hz in two, and 20 Hz in one. Current was adjusted every 1 to 2 months, in increments of 0.1 to 0.25 mA; however, the maximum did not exceed 3.0 mA. Typically, therapeutic efficacy is achieved when current reaches ≥ 1.0 mA. Most patients in this study fit this pattern; however, three responded well at relatively low current (0.8, 0.8, and 0.5 mA, respectively), with seizure reduction > 80% in two patients and > 50% in one.
This study has several limitations, including small sample size, insufficient analysis of structural lesions and GTCS-related factors, lack of inclusion of electroencephalographic characteristics, and potential unrecognized factors influencing the formation and progression of DRE. The relatively short follow-up period may also have affected our assessment of long-term efficacy. Further studies are needed for confirmation.
5 Conclusion
VNS treatment for patients with DRE is minimally invasive and reversible, and has relatively broad surgical indications with few complications and significant efficacy. Compared with surgeries that involve resection of cerebral tissue, VNS does not cause iatrogenic injury to the brain parenchyma. This provides many DRE patients who are unsuitable for epileptogenic focus resection with a favorable surgical treatment option. VNS demonstrated significant clinical efficacy in patients with a structural etiology and GTCS, particularly those with post-traumatic DRE. In some patients, clinical seizures even disappeared completely.
Footnotes
Acknowledgements
Special thanks to the Xinyang Wang team, the Nina Zhang team, and all the engineers and technical staff for their valuable contributions.
Funding Information
This work was supported by National Key Research and Development Program (Grant Number: 2021YFC2401204).
Author Contribution
Xinyang Wang:data collection and curation; follow-up;writing.Nina Zhang: investigation and evaluation;writing.Yujie Li:surgery.Pengcheng Gao:surgery.Zhongyang Zhou:surgery.Weiqiang Chen:surgery.
Declaration of Conflicting Interests
The authors have no conflicts of interest to declare.
Data Availability Statement
Authors elects to not share data.
Ethics Statement
The study was approved by the Medical Ethics Committee of The Third People’s Hospital of Bengbu (Ethics Approval Number: Lunke PI Zi [2020] No. 40).
Informed Consent
All patients provided written informed consent at the time of entering this study.
