Abstract
The real-time, noninvasive, nonionizing, high spatiotemporal resolution, and flexibility characteristics of molecular fluorescence imaging provide a uniquely powerful approach to imaging and monitoring the physiology and pathophysiology of ischemic stroke. Currently, various fluorescence probes have been synthesized with the aim of improving quantitative and quantitative studies of the pathologic processes of ischemic stroke in living animals. In this review, we present an overview of current activatable fluorescence probes for the imaging and diagnosis of ischemic stroke in animal models. We categorize the probes based on their activatable signals from the biomarkers associated with ischemic stroke, and we present representative examples of their functional mechanisms. Finally, we briefly discuss future perspectives in this field.
1 Introduction
Ischemic stroke, caused mainly by blocked blood flow or stenosis of blood in the brain, is a leading cause of disability and death worldwide [1]. The series of severe sequelae that can occur include ischemia, hypoxia, ionic imbalance, overproduction of reactive oxygen species (ROS), metabolic impairment, increased release of inflammatory cytokines (tumor necrosis factor α [TNFα], interleukin-6 [IL-6], and interleukin-1β [IL-1β], etc.) as well as increased recruitment of neutrophils, and accelerated apoptosis of neurons [2–4]. Accurate, real-time, and noninvasive monitoring of the dynamic progression of ischemic stroke would therefore provide tremendous opportunities for better prediction, diagnosis, and therapeutic interventions. Prediction of the potential for stroke before its onset through acquired images could help in avoiding and reducing loss of, or damage to, neurons. Upon stroke onset, cerebrovascular images could provide clinical information to evaluate severity, which could assist in choosing timely therapeutic interventions. After therapy, imaging could assist in evaluating the progress of recovery.
Currently, anatomic and functional imaging modalities such as computed tomography and magnetic resonance imaging are already in clinical use for assessing stroke and monitoring treatment [5, 6]. However, the ionizing radiation delivered efficacy during computed tomography limits the number of times that that technique can be used within a short period. Magnetic resonance imaging avoids the use of ionizing radiation and offers excellent soft-tissue contrast; however, its temporal resolution and sensitivity are both low.
Different from traditional anatomic imaging, molecular imaging enables visualization of biologic and pathologic processes at the cellular and molecular levels, and therefore, it allows earlier detection of diseases, precise evaluation of disease stages, and theranostic monitoring of disease progression in a quantitative manner. In particular, fluorescence molecular imaging offers real-time monitoring of biologic systems and subtle molecular abnormalities with high sensitivity and selectivity and excellent spatiotemporal resolution. As such, a series of fluorescence probes have been developed. Based on the signals response towards the molecular targets, the available fluorescence probes fall into two main categories: “always on” and “activatable” [5, 7, 8]. “Always on” probes provide constant luminescence signal despite where they distribute. Thus, their signals are read primarily based on the concentration difference of the probes in normal and diseased tissues. By contrast, activatable probes are initially non-luminescent, and they emit signals only in the presence of the target biomarkers or events of interest in normal/diseased tissues. Consequently, they offer concentration-independent signal activation with high SBR and real-time semi-quantification of biomarkers in living organisms.
Considering the latter benefits, our focus here is on activatable fluorescence probes for the detection and diagnosis of ischemic stroke. We categorize the probes based on the biomarkers to which they respond. Recent advances in probe development, with their resulting advantages and limitations, are discussed. Finally, the summary and future perspectives of fluorescent neuroimaging will be discussed.
2 Metal ion-activatable fluorescence probes
Cations and anions act as crucial regulators of various biologic processes [9]. Dysregulation of cellular ionic homeostasis is closely correlated with the onset of various diseases [10]. In particular, iron ions (Fe2+/Fe3+), the most abundant and essential transition metal in humans, have indispensable roles in oxygen generation and DNA/RNA synthesis and repair [11, 12], heme synthesis [13, 14], and neural activity [15]. Aberrant iron metabolism has been implicated in diseases such as anemia, cancer, and Alzheimer’s and Parkinson’s disease, among others [16]. To spatiotemporally track the presence and abundance of Fe2+ after ischemic stroke, Wang et al. designed an “off–on” probe in which N-aryl-O-acylhydroxylamine functioned into naphthalimide-based fluorophore (Fig. 1) [17]. Due to the electron-withdrawing effect of the acylated hydroxylamine on the internal charge transfer system, the probe was initially nonfluorescent. Upon Fe2+-induced N–O cleavage, the generation of an electron-donating amine offered the probe with a desired “off−on” response. The probe responds to Fe2+ quickly (within 15 min) and with good selectivity, enhancing the fluorescence intensity by a factor of more than 27. Moreover, the probe can detect Fe2+ at concentrations as low as 0.5 μM, with a noticeable fluorescence enhancement by a factor of 3. This probe thus has the capacity to detect elevated Fe2+ in ischemic brain tissue.
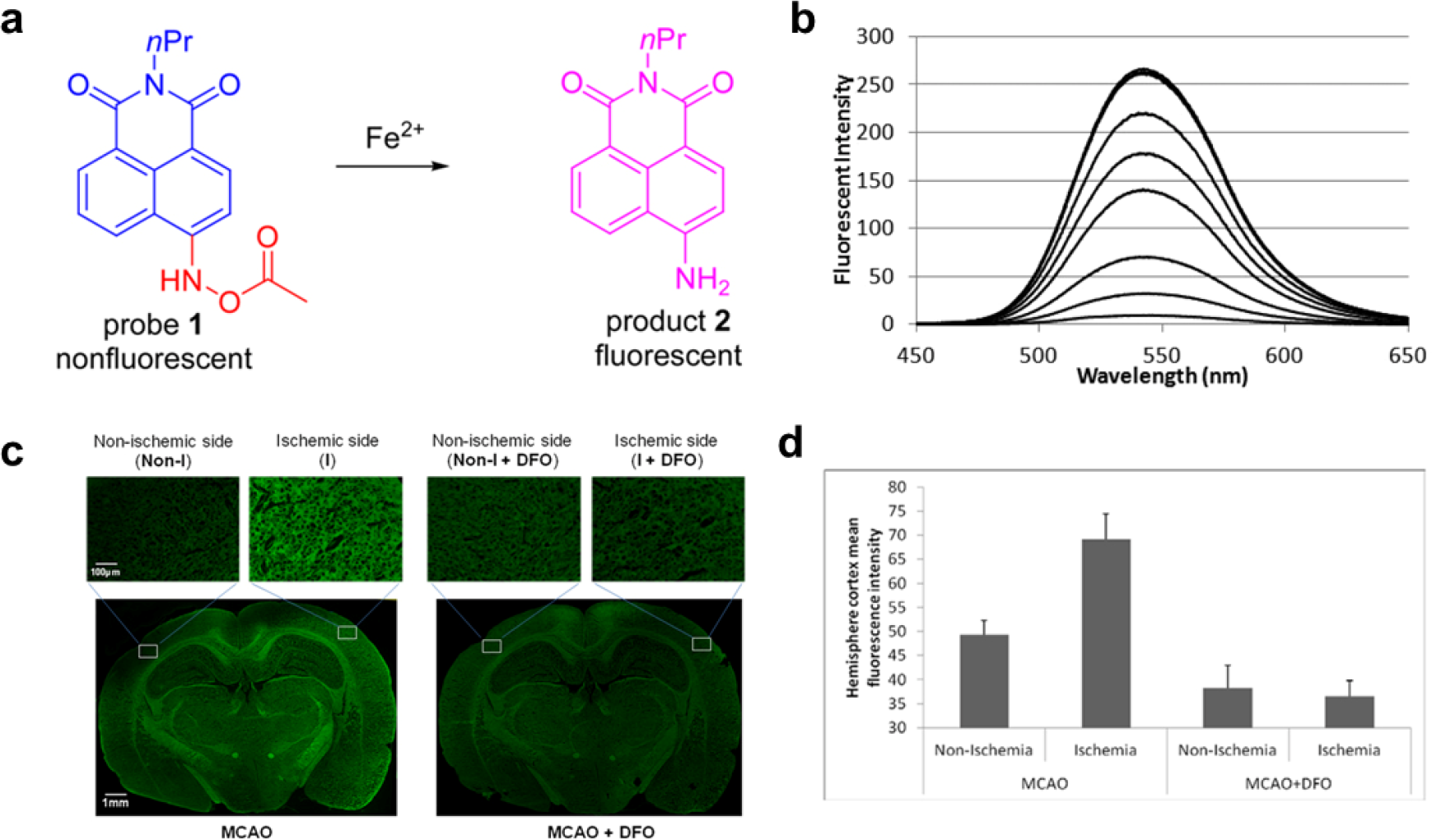
Reaction-based “off–on” fluorescent probe for detecting elevated Fe2+ in ischemic stroke. (a) Design of the probe, based on fluorescence induced by N-aryl-O-acylhydroxylamine in the presence of Fe2+. (b) Sensitivity study of probe 1 in the presence of Fe2+. (c) Images of labile Fe2+ in rat ischemic brain tissue. Tissue sections were incubated with 5.0 μM probe 1 for 15 min at the nonischemic and ischemic sides. (d) Relative mean fluorescence intensity in the brain sections. Reprinted with permission from Ref. [17] ©2016, American Chemical Society.
3 Protease-activatable fluorescence probes
Proteases cleave the peptide bonds in proteins and peptides into polypeptides or amino acids by hydrolysis [18]. They form large family, including metalloproteinases (MMPs), serine, cysteine, threonine, and the aspartic proteases. Specifically, the MMPs are a family of zinc-dependent endopeptidases responsible for tissue homeostasis and remodeling in normal conditions [19, 20]. Aberrant expression of MMPs results in apoptosis, disruption of the blood–brain barrier (BBB), hemorrhage, neuroinflammation, and extracellular matrix turnover [19, 20]. Thus, they can serve as biomarkers of pathologic progression in many conditions. The MMP-activatable probes for imaging ischemic stroke currently include MMPSense680 [21], MMPSense750 [22], MMP2/ 9-activatable cell-penetrating peptides [23, 24], and MMP-P12 [25].
Near-infrared (NIR) fluorescence imaging, which uses wavelengths between 650 nm and 900 nm, offers low autofluorescence, deep tissue penetration, and low phototoxicity, rendering it attractive for in vivo studies [26]. To this end, Ju et al. reported an ultrafast MMP-activatable NIR fluorescence imaging probe for the real-time visualization of MMP activity in a mouse model of ischemic stroke (Fig. 2) [27]. The side-chain-protected MMP substrate GPLGVRGKGG was labeled with Cy5.5 dye and connected to BHQ-3 as a quencher. Because of its optimized stability, blood half-life, and enzyme susceptibility, polyethylene glycol was selected as the backbone of the quenched molecular beacon. The probe remained in a quenched state until activated by MMPs under pathophysiologic conditions, at which point it emitted intense fluorescent signals. Significant signal enhancement was observed in as little as 15 min, with peak signal intensity occurring at 1 h post injection. More importantly, the signals indicated a significant linear correlation between activated p38 MAPK protein expression and MMP-9 activity, which was much higher than the correlation between p38 MAPK and MMP-2 activity. This study highlighted the potential for using optical probes not only to acquire a better understanding and further explore novel MMP-associated pathways, but also to screen potential therapeutics.
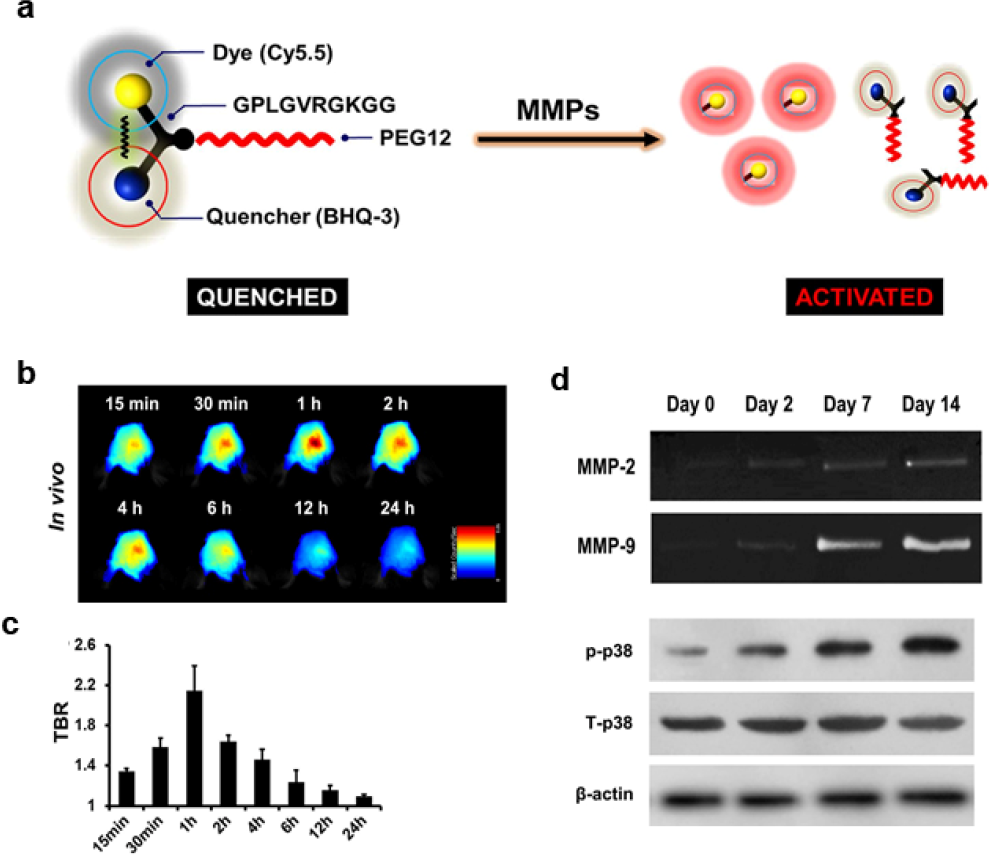
Role of p38 MAPK in MMP activity demonstrated in a photothrombotic mouse model of stroke as measured using an ultrafast MMP-activatable probe. (a) Chemical operation of the MMP-P12 probe. (b) In vivo near-infrared fluorescence images of the ischemic brain from 15 min to 24 h after probe injection on day 7 post stroke. (c) Quantitative target-to-background ratio values (n = 3). (d) Correlation between MMP expression and p38 MAPK activation. Reprinted with permission from Ref. [27] ©2015, Nature Publisher.
4 Reactive oxygen species–activatable fluorescence probes
Lanthanide upconversion nanoparticles (UCNPs) utilize two or more low energy photons, usually at NIR regions, and emit at higher energy photons, typically from UV−vis to NIR regions [28–31]. Their excellent optical properties — including high photostability, lack of photoblinking, and chemical stability, among others — make them useful in diverse fields. Liu and coworkers designed a BBB-permeable and hypochlorous acid (HOCl)–activatable upconversion nanoparticle probe with NIR emission for monitoring the progress of neuroinflammation with cerebral stroke in a mouse model [32]. The nanoprobe was consisted of three moieties: NaYbF4:Gd@NaYF4:Yb/Tm (UCNPs) as the signal reporter, Cy-HOCl dye as the energy acceptor from the UCNPs and recognition unit of the HOCl, and amphiphilic polymers endowing the probe with biocompatibility and BBB permeability. Because the absorption of Cy-HOCl matched well with the emission of UCNPs, a luminescence resonance energy transfer process occurred, and the upconversion luminescence of the UCNPs was quenched. Upon reacting with HOCl, Cy-HOCl absorption decreased, inhibiting the luminescence resonance energy transfer. Consequently, upon intravenous injection, the probe crossed the BBB in the mice via low-density lipoprotein receptor–related protein-mediated transcytosis, and the upconversion luminescence signal from Cy-HOCI–UCNPs was recovered in a HOCl-dependent manner, providing real-time monitoring of neuroinflammation in vivo.
Fluorescence imaging in the NIR-II region (1000–1700 nm) provides deep tissue penetration, high spatial and temporal resolution in vivo due to its minimal photon absorption, suppressed scattering, and low tissue autofluorescence [8, 33]. NIR-II fluorescent probes are now widely used for in vivo NIR-II fluorescence bioimaging, in vivo tracking, image-guided diagnosis, and cancer therapy. To this end, in the study by Wang and coworkers, a targeted ONOO−- responsive NIR-IIb nanoprobe (V&C/PbS@Ag2Se) was designed (Fig. 3) [34]. Reactive oxygen (ROS) and/or nitrogen species (RNS) generates are generated during disturbances of the oxygen supply induced by microvascular occlusions or arterial branch embolisms in the brain. In particular, peroxynitrite (ONOO−) generated during the development of ischemic stroke could be a biomarker for early injury. This probe maintained an “off” state because of competitive absorption of excitation irradiation between Cy7.5 and PbS@Ag2Se quantum dots. Upon intravenous injection, the V&C/PbS@Ag2Se quickly accumulated in lesional regions based on VCAM-1-binding peptide targeting the vascular endothelium inflamed by ischemic stroke. Subsequently, the probe was rapidly activated by ONOO− oxidation of Cy7.5, instantly illuminating the lesional regions. In contrast, a V/PbS@Ag2Se probe (“always-on” because of the lack of Cy7.5) failed to distinguish between normal and lesional regions. Quantitatively, at 60 min, the activatable probe had a signal-to-noise ratio of 11.5 compared with 1.5.
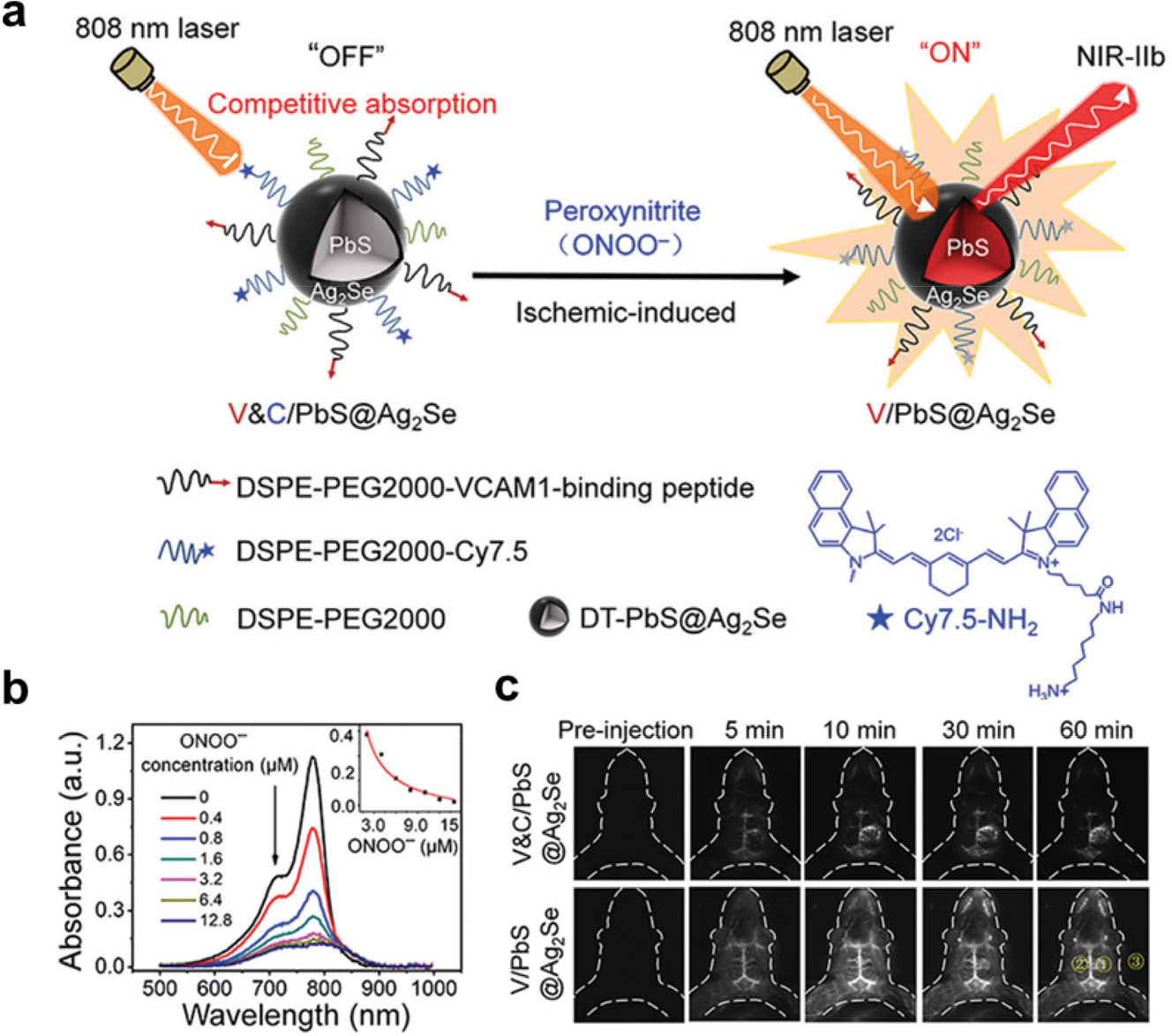
A targeted, highly sensitive near-infrared (NIR)-IIb-activatable probe for the detection of ischemic stroke. (a) Schematic of the construction of V&C/PbS@Ag2Se probe and detection of peroxynitrite (ONOO−) in a mouse model of ischemic stroke. (b) The absorption spectra of Cy7.5 (10 × 10−6 m) with the gradual addition of ONOO− (from 0 to 12.8 × 10−6 μM) (c) Time course of NIR-II fluorescence in early ischemic stroke at various time points after injection of V&C/PbS@Ag2Se and V/PbS@Ag2Se. Reprinted with permission from Ref. [34] ©2021, John Wiley and Sons.
5 Conclusions and perspective
Studies in the past decade have highlighted the potential of fluorescence imaging for the diagnosis and imaging of ischemic stroke. Activatable imaging probes that use favorable chemistry offer the benefits of high spatiotemporal resolution, fast acquisition, and flexible operation, etc. With a set of representative examples, we have highlighted typical probes that have been assessed for applications including identification of ischemic stroke, detection of inflammation post ischemic stroke, possible biomarkers, and screening for therapeutics.
Nevertheless, activatable fluorescence probes remain in the proof-of-concept stage, and challenges such as fluorescence properties (e.g., quantum yields, emission wavelengths, photostability, solubility) and biocompatibility, are needed to be addressed. Moreover, given the current limited penetrance of fluorescence imaging, multimodal activatable imaging that integrates the benefits of multiple modalities offers a more comprehensive view of ischemic stroke detection and pathology, which unfortunately have received little attention to date. More effort and more detailed investigation are urgently needed. Theranostic platforms that combine therapeutics such as tissue plasminogen activator, thrombolytics, and anti-inflammatory agents to dissolve blood clots and mitigate neuroinflammation with activatable imaging systems is a particularly promising research direction.
Many challenges in the development of activatable molecular probes remain. Still, this emerging field holds great potential to improve the imaging and treatment of ischemic stroke.
Footnotes
Conflict of interests
The authors declare no competing interests.
Funding
This work was supported by the National Key Research and Development Program of China (Grant No. 2021YFA1201203).
Authors’ contribution
M. W and Y. Z. conceived the idea and co-wrote the manuscript.
