Abstract
Excitatory-inhibitory (E/I) balance is essential for normal neural development, behavior and cognition. E/I imbalance leads to a variety of neurological disorders, such as autism and schizophrenia. NMDA receptors (NMDARs) regulate AMPAR-mediated excitatory and GABAAR-mediated inhibitory synaptic transmission, suggesting that NMDARs play an important role in the establishment and maintenance of the E/I balance. In this review, we briefly introduced NMDARs, AMPARs and GABAARs, summarized the current studies on E/I balance mediated by NMDARs, and discussed the current advances in NMDAR-mediated AMPAR and GABAAR development. Specifically, we analyzed the role of NMDAR subunits in the establishment and maintenance of E/I balance, which may provide new therapeutic strategies for the recovery of E/I imbalance in neurological disorders.
1 Introduction
There are two types of synaptic transmission in our brain, namely, excitatory synaptic transmission and inhibitory synaptic transmission [1]. The balance between excitatory and inhibitory synaptic transmission (E/I balance) is critical for maintaining the normal functions of the nervous system [2]. Whereas, an E/I imbalance leads to neurological disorders, such as autism and schizophrenia [3, 4]. α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors (AMPARs) and γ-aminobutyric acid type A receptors (GABAARs) are ionotropic receptors and the major receptors on the postsynaptic membrane, which mediate the neuronal excitation and inhibition in the brain, respectively [5]. Investigating how AMPARs and GABAARs develop into functional receptors will unveil the fundamental molecular mechanism of E/I balance establishment [6]. It has been reported that the absence of N-methyl-D-aspartate receptors (NMDARs) leads to enhancement of AMPAR-mediated synaptic transmission, which indicates that NMDARs regulate the development of AMPAR [7, 8]. Additionally, it has been shown that activation of NMDARs enhances GABAAR- mediated synaptic transmission [9, 10], indicating that NMDARs also regulate GABAAR development [11]. Therefore, we conclude that NMDARs play a crucial role in the establishment and maintenance of the E/I balance. Furthermore, mutations in NMDARs, GABAARs and AMPARs cause neurological disorders, suggesting that these ionotropic receptors make important contributions to the pathogenesis of these neurological disorders [12–17]. In this review, we introduce the ionotropic glutamate receptors NMDARs and AMPARs, and GABAARs. Then, we summarize the molecular mechanisms underlying E/I balance establishment and maintenance mediated by NMDARs. Finally, we survey the possible role of E/I imbalance in neurological disorders.
2 NMDA, AMPA and GABAA receptors
NMDARs play a crucial role in the development of GABAergic and glutamatergic synapses by regulating the number, distribution and subtype of AMPA and GABAA receptors on the excitatory and inhibitory neurons [18].
2.1 NMDARs
Typically, NMDARs are di-heteromeric complexes, which are composed of two GluN1 and two identical GluN2 (GluN2A–D) or GluN3 (GluN3A–B) subunits [19, 20]. NMDARs could also assemble as tri-heteromeric complexes containing two GluN1 and two different GluN2 or GluN3 [20, 21]. The NMDAR subunits are encoded by seven genes [22]. GluN1, an obligatory subtype is encoded by GRIN1 gene, and there are eight variants for it due to N- and C-terminal alternative splicing [23]. All NMDAR subunits consist of an N-terminal domain (NTD), glutamate or glycine binding domain (ABD), transmembrane domain (TMD) and a C-terminal domain (CTD) [24]. The NTD has a clamshell-like structure and mediates allosteric changes in response to ligand binding, whereas ABD binds to glycine, D-serine (in GluN1) and glutamate (in GluN2). The TMD forms an ion pore for the NMDAR, whereas the CTD stabilizes the NMDAR and mediates signaling transduction [25]. A study reported that GluN2B-containing NMDARs are replaced by GluN2A-containing NMDARs during development, which suggests that the subunit composition of NMDARs could change during development [20, 26]. NMDAR is an ionotropic channel gated by glutamate and glycine [27]. Glycine binds to GluN1 or GluN3 subunit, whereas glutamate binds to GluN2 subunit [28–30]. However, the glutamate and glycine released into the synaptic cleft from the presynaptic membrane cannot open NMDAR channel since magnesium ion (Mg2+) blocks the NMDAR pore [31]. Once the action potential generates postsynaptic depolarization and removes the Mg2+ block, the NMDARs open, and lead to Ca2+ influx through NMDARs. This activates the downstream Ca2+ signaling pathways [32–35], which are required for learning and memory.
2.2 AMPARs
AMPARs are heterotetrameric ion channels composed of GluA1–4 (GluR1–4) subunits [36, 37], and mediate fast excitatory synaptic transmission. The subunit composition of AMPARs in mouse hippocampal neurons has been determined by single-cell genetic and electrophysiological methods [38]. The results showed that approximately 80% of synaptic AMPARs and over 95% extra-synaptic AMPARs were composed of GluA1–A2 heteromers in native environment [38]. It is worth noting that the subunit composition of AMPARs changes during development, and also in neurological diseases, such as autism and schizophrenia [37, 39–41]. Furthermore, defects in GluA2 subunit might cause neurological diseases, such as schizophrenia and autism [14], which suggests that subunit composition of AMPARs changes in these diseases.
2.3 GABAARs
GABAARs are heteropentameric ion channels, and are composed of 19 subunits, namely, α1–6, β1–3, γ1–3, δ, ε, θ, π and ρ1–3 [42, 43]. They are typically composed of two α subunits, two β subunits and one other subunit [44, 45]. The β subunit is essential for GABAAR inhibitory synapse formation [46, 47]. In addition, compared with β2 subunit, the expression level of β3 subunit was higher in the hippocampal inhibitory synapses [48], indicating that the inhibitory synaptic transmission was primarily mediated by β3 in the hippocampus [46]. Given the critical role of hippocampus in learning and memory [49], we presume that the β3 subunit might make major contributions to learning and memory. However, this has not been investigated yet.
Taken together, alternative splicing and subunit composition confer structural, functional and pharmacological features to NMDAR, AMPAR and GABAAR, which could dramatically enhance the complexity of these receptors [50–52].
3 Excitatory-inhibitory (E/I) balance
When excitation increases, inhibition also increases within a week, which is referred to as the excitatory-inhibitory (E/I) balance [53–55]. E/I balance is established and maintained in a cellular autonomous manner, and has been observed even in single excitatory and inhibitory neurons [56, 57]. Notably, the E/I balance is multi-dimensional and is maintained at both, single-cell and global circuit levels [58]. Spontaneous or evoked synaptic events, such as AMPAR-mediated miniature excitatory postsynaptic current (mEPSC) and GABAAR-mediated miniature inhibitory postsynaptic current (mIPSC), or AMPAR-mediated evoked EPSC and GABAAR-mediated evoked IPSC have been assessed to uncover the E/I balance [56, 59]. It has been reported that a decrease in AMPAR-mediated excitatory input induces a neural autonomic response, and thus reduces the GABAAR-mediated inhibitory input onto the same neuron [56]. Through the autonomic response of neurons, neuronal excitation and inhibition are coincident, and stable activity is produced [54]. However, a decrease in GABAAR-mediated inhibitory input does not reduce excitatory input [60], suggesting that E/I balance is mainly maintained by excitatory input. In line with this, it has been reported that excitatory input controls the development of inhibitory synapses [11, 56]. A disturbance in E/I balance leads to neuronal and circuit plasticity dysfunction [56], thereby causing abnormal visual system and behavior in Xenopus tadpoles [60], which suggests that E/I balance is essential for cognition and behavior. Furthermore, E/I imbalance is observed in multiple neurological disorders [61], indicating the critical role of E/I balance in nervous system development.
3.1 NMDAR regulates E/I balance
Blocking of NMDAR with phencyclidine and ketamine could inhibit neuronal activity, and thus be used as an anesthetic [62–64]. In addition, studies have shown that a subanesthetic dose of ketamine could block NMDAR and enhance neuronal activity, hence can be used as an antidepressant [63, 65–67]. Subsequent studies suggested that GluN2B-containing NMDARs in GABA interneurons are essential for the antidepressant effect of ketamine [67, 68]. Furthermore, ketamine could decrease neuronal PAS domain protein 4 (Npas4) [69], and thereby disrupt the excitatory-inhibitory balance in a cell type-specific activity-dependent manner [70]. Ketamine also shows extensive molecular effects by blocking the HCN1 channel [71–73]. Therefore, NMDAR could regulate both, excitation and inhibition. In line with this, NMDAR regulates AMPAR and GABAAR-mediated excitatory and inhibitory synaptic transmission, respectively (Fig. 1) [65, 74], and thereby controls the establishment and maintenance of E/I balance [75–77].
3.2 NMDARs regulate AMPAR-mediated excitatory synaptic transmission
NMDARs regulate excitatory synaptic transmission by increasing or decreasing AMPAR synaptic localization, and thus modulate synaptic plasticity [78–80]. Under physiological and pathological conditions, NMDARs increase or decrease the synaptic localization of AMPARs through transmembrane AMPAR regulatory proteins (TARPs)/stargazing [81] and proteasome [74], and thus regulate excitatory synaptic transmission (Fig. 1). TARPs/stargazin directly bind to AMPARs, and then stabilize the open state [82]. They also interact with postsynaptic density 95 (PSD-95) and modulate the synaptic localization of AMPARs, and therefore regulate long-term potentiation [83]. Upon activation of NMDARs, PSD-95 is ubiquitinated and subsequently degraded by proteasomes. This results in internalization of AMPARs, and hence long-term depression induction [84]. Furthermore, previous studies have shown that NMDARs might enhance AMPAR-mediated excitatory synaptic transmission through their cleaved GluN1 C-termini and calmodulin (Fig. 1), which indicates that NMDAR GluN1 subunit could play a crucial role in synaptic transmission. However, the molecular mechanisms need to be further investigated [85, 86]. Taken together, these results suggest that NMDARs regulate AMPAR-mediated excitatory synaptic transmission through different signaling pathways, and thus control excitation.
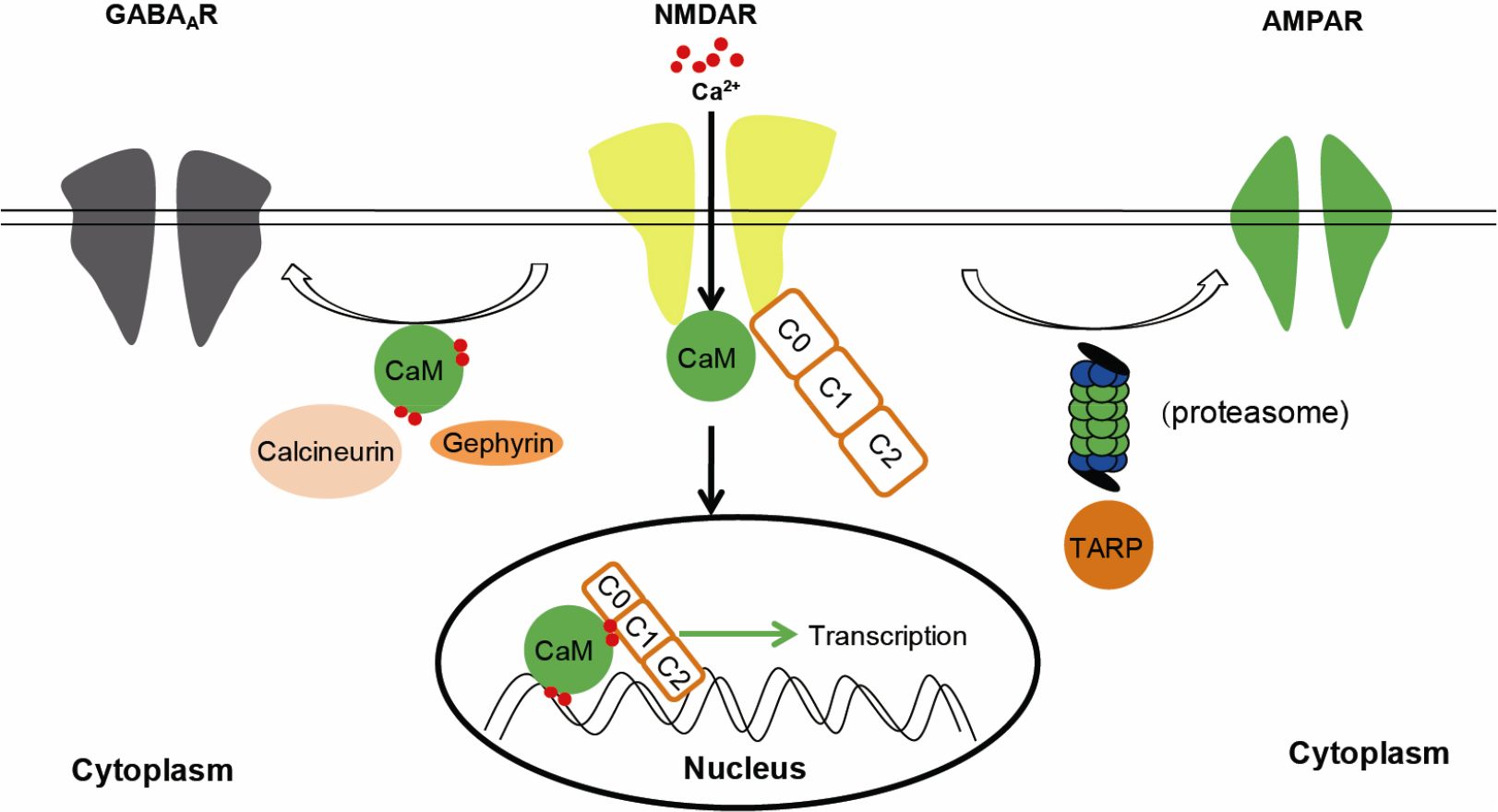
NMDARs regulate GABAergic and AMPAergic synapse formation. NMDARs regulate GABAergic synapse formation by affecting calmodulin (CaM), calcineurin and gephyrin. NMDARs regulate AMPAergic synapse formation by affecting TARP and proteasome.
3.3 NMDAR regulates GABAAR-mediated inhibitory synaptic transmission
GluN1 is the obligatory subunit of NMDARs, and GABAergic synapses are decreased in GluN1-deficient developing neurons, suggesting that NMDARs are required for inhibitory synaptic development [11, 87]. Ca2+ influx through NMDARs activates the phosphatase calcineurin and decreases the phosphorylation of GABAAR γ2 subunit at serine 327, therefore increases the lateral diffusion and clustering of GABAARs [10]. Gephyrin is an inhibitory synaptic scaffold protein, which controls the lateral diffusion and clustering of GABAARs during neuronal excitation [88, 89]. Upon NMDAR activation, CaMKII is activated, which in turn phosphorylates the GABAAR β3 subunit at serine 383, and therefore recruits gephyrin and modulates the surface mobility of GABAARs at inhibitory synapses (Fig. 1) [90]. For further in-depth review of NMDAR-mediated GABAergic synapse development, we recommend to read the relevant review [91], which suggests that NMDARs control the inhibition though binding to calmodulin. Interestingly, GABAergic inhibitory synapses on pyramidal neurons originate from different interneurons and are regulated by distinct mechanisms [59, 92]. NMDAR in somatostatin (SOM) interneurons rather than parvalbumin (PV) interneurons regulates the GABAergic inhibitory synaptic development in hippocampal CA1 pyramidal neurons [92]. Another study reported that the excitatory activity of pyramidal neurons in cerebral cortex were affected by GABAergic inhibitory synaptic development of PV interneurons rather than SOM interneurons [59]. This indicates that the E/I balance in different brain regions is regulated by different mechanisms.
Interestingly, GABAAR mediates inhibitory synaptic transmission in mature neurons, whereas it mediates excitatory synaptic transmission in immature neurons [93]. The synaptic colocalization of GABAARs and NMDARs during early development indicates that there is a functional cross-talk between them [94, 95]. However, if GABAAR regulates excitatory synaptic transmission during early neuronal development, then how is inhibition generated during neuronal development and how can neuronal excitability be balanced? Other glutamate transporters decrease local glutamate concentrations for NMDARs, and therefore prevent their activation [96]. Given the essential role of NMDARs in GABAergic synapse development [11], this might attenuate the GABAergic synapses development. Taken together, these studies indicate that glutamate transporters balance excitability by controlling the NMDAR activation, thereby leading to the establishment of an E/I balance [93, 97].
3.4 NMDAR mutations and E/I imbalance in neurological disorders
Disruption in E/I balance during critical periods of development contributes to autism spectrum disorder (ASD) [98], which is one of the most prevalent neuro-developmental disorders caused by a combination of genetic and environmental factors [99]. Studies have shown that mutations in the GluN2A, GluN2B and GluN2C subunits of NMDARs are associated with ASD and other neurological disorders [76, 100–102]. These mutations in NMDARs affect its receptor functions. For example, the C-terminal S1413L mutation of GluN2B impairs NMDAR-mediated synaptic transmission and reduces spine density [13]. Mutations in GRIN1 gene encoding the NMDAR GluN1 subunit are also associated with neurological disorders, such as intellectual disability [103] and epilepsy [104]. Although major mutations have been proven to attenuate NMDAR function, some mutations have been shown to enhance NMDAR function. It has been reported that GluN2A-P552R increases the glutamate/glycine potency of NMDARs [105]. Taken together, both hyper- and hypoactivation of NMDARs, were proposed to contribute to neurological disorders.
Restoring the E/I balance is a potential therapeutic strategy for neurological disorders [61]. Epilepsy occurred in 1/3 of ASD cases, which indicates that inhibition circuit is dysfunctional in autistic patients [106]. Furthermore, epilepsy caused by inhibitory dysfunction is also common in other neurological disorders, such as tuberous sclerosis [107] and fragile X syndrome [108, 109]. This suggests that restoration of inhibition will ameliorate epilepsy and neurological disorders [58, 110]. PV and SOM interneurons are two major inhibitory interneurons in brain. Disrupted PV- and SOM-expressing interneurons result in autism-related behavioral phenotypes, which further confirms that the inhibition circuit is abnormal in autism [111]. Additionally, knockout of NMDARs in PV interneurons could phenocopy the autism-like phenotypes, indicating that NMDAR deficiency might attenuate the inhibition of interneurons, thereby resulting in autism-like phenotypes [112]. This is consistent with a previous study showing that NMDARs are required and essential for GABAergic synapse development [11]. Excitatory dysfunction was also found in ASD [113], suggesting that E/I imbalance may be the pathophysiological mechanism of these neurological disorders. Mutations in NMDAR [12, 13], AMPAR [14, 15] and GABAAR [16, 17] are observed in the ASD and schizophrenia, which further proves that E/I imbalance may indeed be the neural mechanism of these neurological disorders [61]. Therefore, restoration of E/I balance by targeting these receptors is recommended for treating patients with these neurological disorders.
4 Conclusion and perspective
E/I balance is the key to effective neural coding in the brain. However, the underlying molecular mechanism remains unclear. In the past few decades, studies have shown that the ionotropic functions (ion flux-dependent functions) of NMDARs are closely related to the establishment and maintenance of E/I balance, but the metabotropic functions (ion flux-independent functions) of NMDARs were uncovered recently, which may also affect the E/I balance [114, 115]. The alterations in intrinsic excitability might also affect the NMDAR-mediated E/I balance, and regulate the action potential generation and propagation of neuronal signaling. However, this needs to be further investigated. Further exploration of the functions and molecular mechanisms of NMDARs in E/I balance may unveil the potential therapeutic targets for the treatment of autism, schizophrenia and other neurological disorders, and thus benefit patients with these neurological disorders.
Footnotes
Conflict of interests
All contributing authors have no conflict of interests to declare.
Funding
This work is granted by the National Natural Science Foundation of China (Grant No. 32170975) and Guangdong Nature Science Foundation (Grant No. 2019A1515011309).
Authors’ contribution
LZ wrote the manuscript. LZ, XHS, and JJD revised the manuscript. All the authors proved the manuscript.
Acknowledgments
We thank Bullet Edits Limited for the linguistic editing and proofreading of the manuscript.
