Abstract
Neural tissue-like constructs have important application potential in both neural tissue regeneration and individual medical treatment due to the ideal bioenvironment they provide for the growth of primary and stem cells. The biomaterials used in three-dimensional (3D) biomanufacturing techniques play a critical role in bioenvironment fabrication. They help optimize the manufacturing techniques and the long-term environment that supports cell structure and nutrient transmission. This paper reviews the current progress being made in the biomaterials utilized in neural cell cultures for
Keywords
1 Introduction
Over the last two decades, neuroscience attracted attention due to the increasing requirements for the treatment of neural diseases and the development of artificial intelligence (AI). A systematic investigation into the mechanisms behind the signal pathways and the behavior of neural cells revealed several key nodes of the reaction chain, which have become research targets for new medicines to treat neural diseases. Regarding AI technology, biomimetic neural networks are designed and developed to deal with mass data as artificial brains that receive signals and deliver commands. The mechanisms behind the behavior and function of neural cells are vital in the promotion and investigation of biological theory and the construction of artificial information centers. The current methods for studying neural cell behavior and its part in the development of brain diseases and the regeneration of networks mainly rely on two-dimensional (2D)
Neural tissue-like constructs are the ideal 3D samples and have set high standards for artificial tissue fabrication, as is illustrated in Fig. 1. These constructs require long-term
2 Biomaterials and biofabrication for tissue-like constructs
The biological microenvironment contributes to the development of cells by supporting their structure and nutrient transmission. The materials used in the fabrication of the environment must meet four key characteristics: biocompatibility, porosity, supportability, and permeability. Bio-compatibility allows the auxiliary system of cells to grow, and porosity provides a better and more ideal graft for proliferation and differentiation. Sustainability promotes a long-term stable structure to support the system, and permeability enhances the communication between the system and the surrounding environment for functional regeneration. The development of biomaterials plays an important role in brain tissue engineering. Natural materials have led to a revolution in regeneration materials. Spider silk has the advantage of biocompatibility, strongly demonstrated in supporting the growth of Schwann cells for nerve reconstruction [3]. Hyaluronic acid is widely applied in plastic surgery for tissue recovery and healing, and its derivatives can be mixed with stem cells [4]. The cell-laden structure is customized for various kinds of tissue progenitor and mature cell constructs. Although natural materials are beneficial for biocompatibility, their lack of engineering capability impedes their application in medicine. Combining natural and industrial materials might solve this dilemma. Based on double-coaxial laminar flow microfluidic techniques, engineered meter-long core-shell hydrogel microfibers are fabricated to encapsulate stem and primary cells for 3D culturing [5]. In addition, 3D printable graphene composites, with a majority of graphene and a minority of polylactide-co-glycolide particles, greatly support the growth of stem cells as they are ideal extracellular supporting graft [6]. Ionically cross-linkable hydrogels with filaments are fabricated for neurite growth guidance by combining the following techniques: pultrusion and wet-spinning [7]. A suitable mechanical modulus is another engineered advantage and contributes to the long-term stability of tissue constructs. The mechanical modulus of implantable microelectronics should match that of the natural tissue during implantation; if a mismatch occurs, harmful damage to the tissue can occur, leading to a severe immune response [8]. An ideal mechanical modulus range for brain tissue implants is around 1 kPa [9, 10]. In addition to the modulus range, a key aspect of regeneration is the regaining of function. Conductive materials added to the biomaterials mixture promote the transmission of electrophysiology signals between neural cells. Polypyrrole is used to enhance the conductivity of the conductive electrospun nanofiber scaffold synthesized by the mixture of polypyrrole, chitosan, collagen, and polyethylene oxide, benefiting the healing of healing damaged tissues [11]. Reduced graphene oxide (rGO) is an ideal extracellular matrix (ECM) material for enhancing neural signals’ transmission, and micro/nano-fibrous scaffolds based on a mixed composite of rGO and silk have great potential in nerve regeneration due to their architectural similarity to the native ECM [12]. Due to the important developments in materials science over the last few decades, multiple advanced biomanufacturing techniques (e.g., 3D bioprinting) have emerged. These techniques demonstrate an efficient capability in reconstructing artificial tissue-like structures and are the focus of several recent studies.
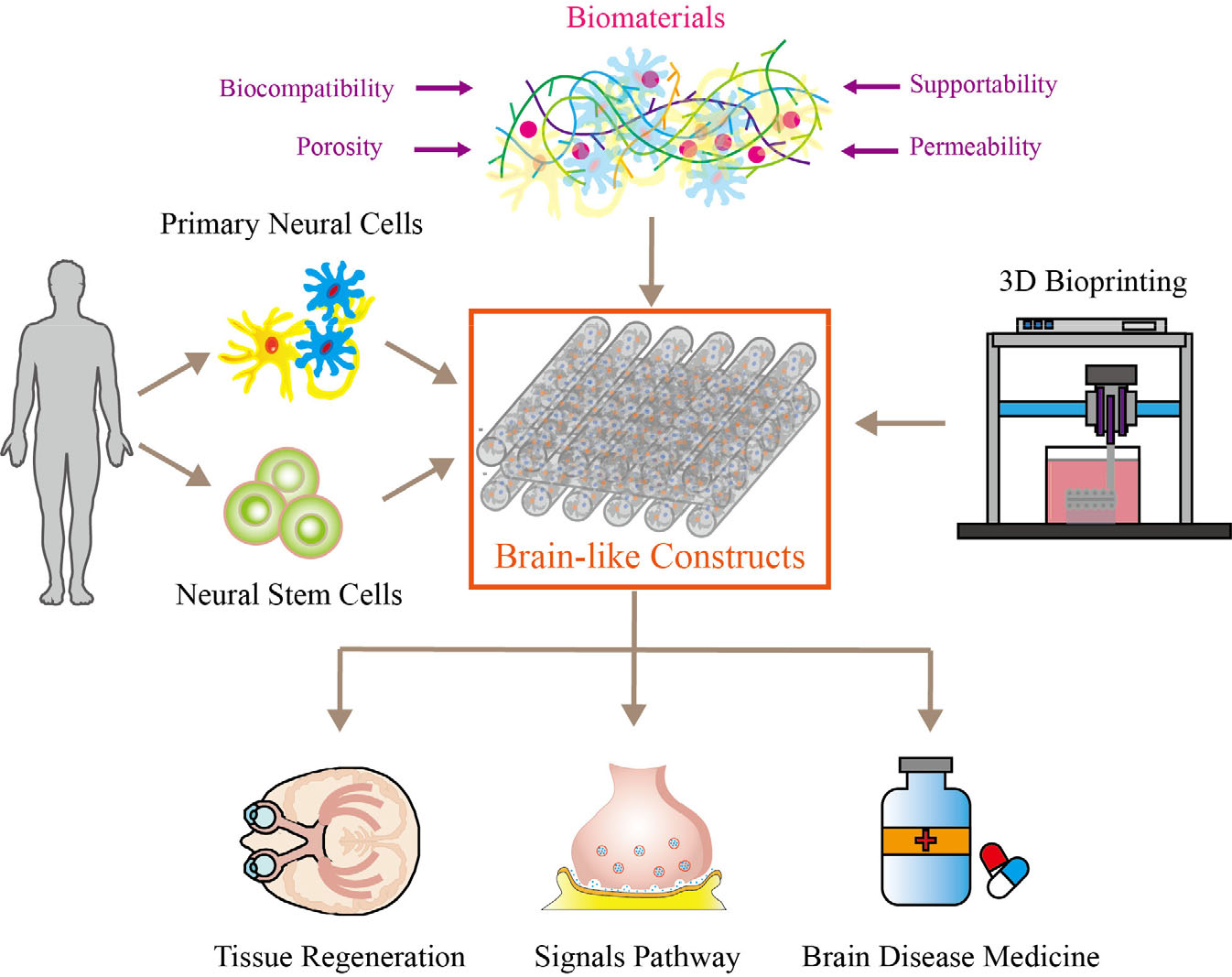
Illustration of the components of neural tissue-like constructs, consisting of primary neural cells, neural stem cells (NSCs), biomaterials, and biomanufacturing methods (i.e., 3D bioprinting). Biomaterials promote long-term culturing by providing an environment with a suitable modulus. Neural tissue-like constructs are widely used in tissue regeneration, neural signal pathways, and brain disease medicine screening models.
3D bioprinting focuses on artificial tissue construction by utilizing the biomimetic concept. The cells encapsulated in bioink materials are regularly patterned, just as they are in natural tissue. This spatial regulation is at the cutting edge of tissue engineering. First, cell-encapsulated tissue constructs with gelatin/alginate were proposed for the therapeutic and nontherapeutic applications of tissue sample replacement [13]. Compared with primary cells, stem cells mixed with bioink pushed the direction of cell therapy forward. Next, the HEK 293FT human embryonic cell was printed to form cell-laden constructs [14]. The parameters of the printing techniques were systematically investigated to obtain the optimized data. The interaction between the biomaterials and the embryonic stem cells (ESCs) was studied in the process of extrusion-based cell printing; the results of this research allowed for the regulation of cell behavior in 3D cultures, not just in 2D cultures [15]. The techniques have been frequently updated to embrace more cell types in 3D printing. High-resolution structures, based on hyaluronic acid hydrogels, promoted natural tissue structures’ spatial manipulation and biomimicking [16].
3 Biomaterials and biofabrication for primary neural cell and brain tumors
Primary neural cells have an important role in neuroscience, particularly in the formulation, differentiation and function of neurons. They affect the entire neurodevelopment process [20]. In addition to brain tissue, retinal and dorsal root ganglia are a source of primary neural cells. Due to the ethical issues surrounding the use of fetal brain cells and the limited quantity of dissected tissue obtained through brain surgery, primary neural cells are often derived from other species (e.g., mice, rats, and chickens). Primary neural cells derived from
As a revolutionary method for depositing cells at a precise location, 3D bioprinting has several potential applications in building bioenvironments similar to microenvironments
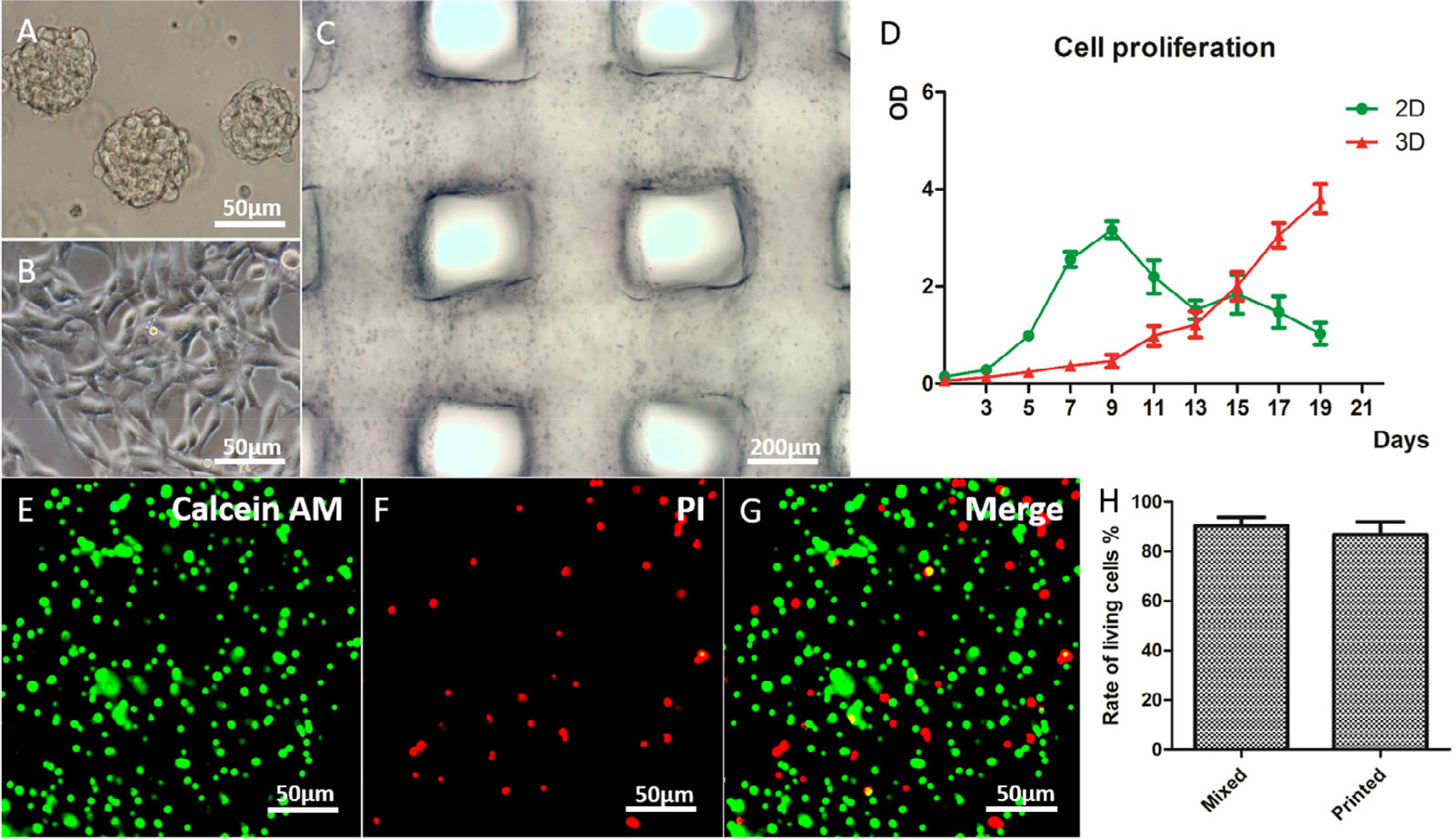
3D-bioprinted gelatin/alginate/fibrinogen (GAF) hydrogels incorporated with glioma stem cells SU3 (unique cells grown in the lab). (A) SU3 cultured in spheres in a stem cell medium. (B) SU3 cultured on a 2D substrate in the complete medium. (C) 3D-bioprinted SU3 at day 1 of culturing. (D) Proliferation of SU3 and 2D substrates and GAF hydrogels (3D), respectively. (E)–(G) Live/dead staining for cell viability after printing. The live and dead cells are stained in green and red, respectively. (H) Cell viability of SU3 after printing. Adapted with permission from Ref. [25].
3D-bioprinted SU3 cultured
Hydrogels play a vital role in bioenvironmental construction around seed cells [26]. High porosity and permeability are necessary for the materials used in 3D bioprinting for ensuring the penetration of nutrients and wastes. Alginate, gelatin, and gelatin methacryloyl (GelMA), noncytotoxic and highly biocompatible materials, are frequently used as scaffolds in 3D bioprinting [27]. Alginate is a natural material derived from algae and is often used in extrusion-based bioprinting. Gelatin consists of peptide sequences derived from collagen, it partially hydrolyzes, and possesses gel–sol properties with an approximate melting point of 30°C. GelMA is a modified form of gelatin and can be cross-linked using UV radiation. Besides these hydrogels, biomacromolecules, such as protein, are important. Fibrinogen, a special natural polymer widely used in bioengineering, is an important biomolecule
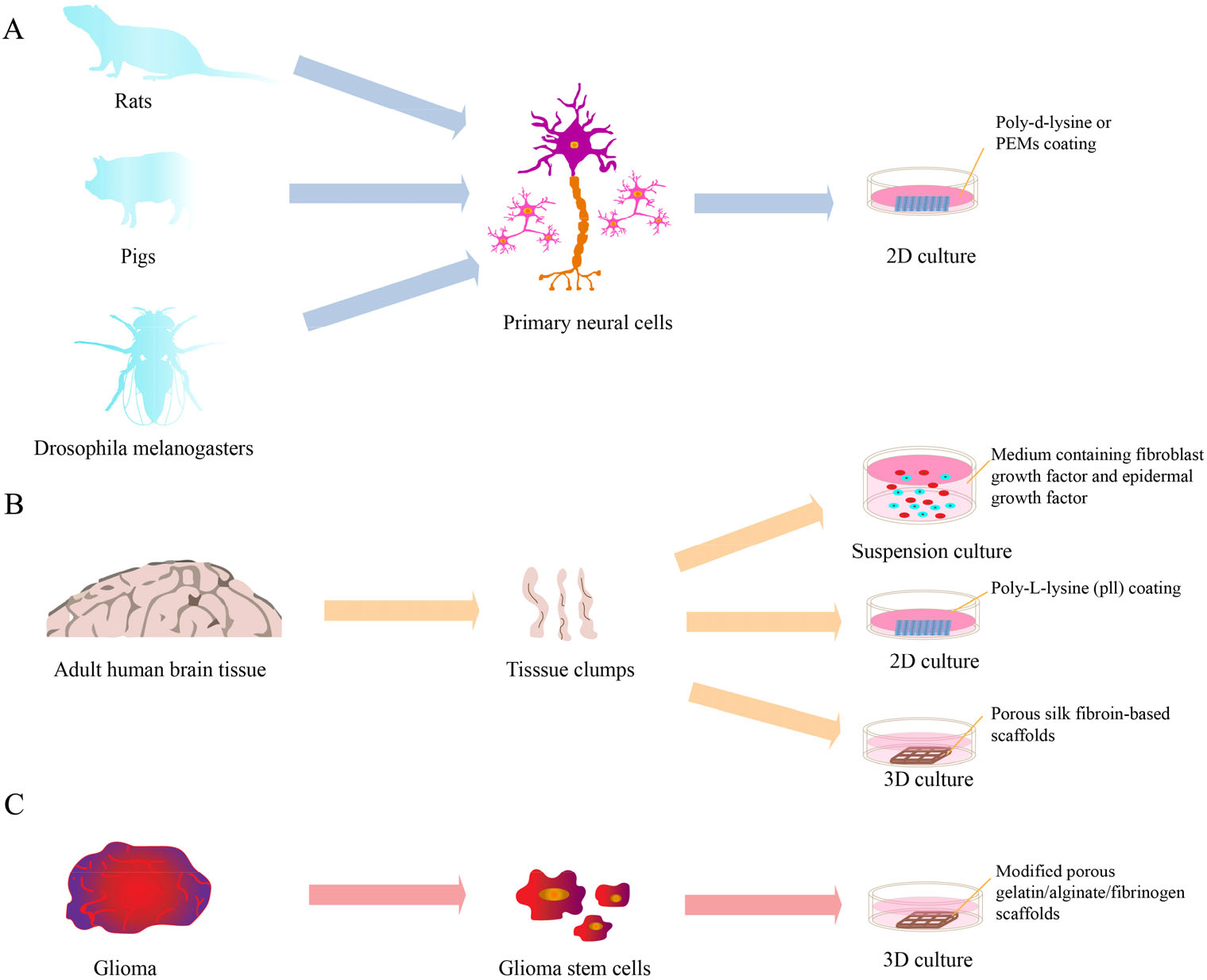
Three types of culture models. (A) Cultural model of primary neural cells. (B) Cultural model of clinical neural tissue. (C) Cultural model of glioma tumors.
3.1 Culture models based on primary neural cells
The primary neural cells culture needs few biomaterials in its culturing system if scaffolds are not required; hence, the modulus is discussed hereinafter. Once the primary cells are dissected or collected from tissue, they are carefully cultured within the medium. Currently, 2D cultures are prevalent although rodent tissue is obtained easily. However, human brain tissue differs from rodent brain tissue at the cellular and functional levels, suggesting that other mammalian species resembling humans should be employed as research models [29]. In previous studies, fetal cell cultures from other species, including chickens, have been performed successfully. However, long-term evolution endows the human brain with special structure and morphology, leading to humans’ excellent capabilities to solve complicated matters over other species [30]. Nevertheless, the differences in brain tissue between rodents and humans appear at the cellular level (i.e., the ratio of gliocytes to neurons and the transcriptomic level including cell phenotype and cell differentiation among the whole neurodevelopmental process). Exploring the mechanism behind these differences is vital in the screening of medication for the treatment of neurodegenerative and neurological disorders (e.g., Alzheimer’s disease and Parkinson’s disease) [31]. A pig’s brain is now considered an ideal replacement research model because of its anatomical similarity to the human brain. For example, the size of a pig’s brain is bigger than a human’s, allowing for high-resolution brain imaging to investigate neural behavior [32]. However, the culture methods of primary porcine neural cells are not sufficiently clear.
Consequently, the development of such methods plays an important role in the neuroscience that determines which mammalian species closely resemble human brains, allowing for the accurate study of the cortex functions and neural mechanisms. For example, primary porcine neural cells from the telencephalon were cultured for seven days
Although few scaffolds are used in primary neural cells culturing, biomaterials for coating are demanded for good
3.2 Cultural models based on clinical neural tissue
Primary neural cells obtained from clinical samples are valuable for investigating brain diseases. For example, human brain tissue dissected during clinical epilepsy surgery is an important bioexperimental source for epilepsy research [39]. However, a limited number of brain tissue experiments based on this source may be unrepeatable. Nevertheless, recently developed biomanufacturing techniques provide solutions for the shortage of valuable samples. 3D cultures perform well in simulated
To support primary neural cell survival
3.3 Cultural models based on brain tumor tissue
Though exciting achievements have been made in treating brain tumors, several issues remain, such as the recurrence of tumors and resistance to anticancer drugs. Patients have no more than a 15-month median survival time after being diagnosed with a high-grade glioma [25]. One of the reasons for this is its rate of recurrence, possibly caused by glioma stem cells. Therefore, treatment targeting the stem cells instead of the glioma itself is likely a better choice, possibly inhibiting the recurrence of the tumor. An ideal tumor model is in increasing demand for studying the physiological characteristics of glioma stem cells and their inherent resistance to anticancer drugs so that a better therapy for treating these tumors can be developed. Previous studies have shown that the modulus of tumor decreases with tumor growth; hence, mechanical properties are key in the model construction [45]. In recent studies on tumor models, 3D bioprinting models perform well in simulating the microenvironment
GAF hydrogels are prevalent in constructing brain tumor models. The mixture is used to mimic the mechanical properties of the tumor cells’ microenvironment and set the modulus < 10 kPa [20]. GAF exhibits a high degree of cell adhesion, migration, and proliferation as well as outstanding crosslinking properties, as shown in Figs. 2 and 3. GAF can stay in the liquid state during printing and maintains a stable solid-state as a scaffold for supporting glioma cells, thus allowing for relatively high resolution and customization. Transglutaminase (TG) is often incorporated as a crosslinker into the system to crosslink gelatin and enhance the stability of the GAF hydrogel system. As a monomeric protein containing active sites, TG can catalyze intramolecular and intermolecular covalent crosslinking of protein polypeptides through acyl transfer reactions. In addition, TG is a nontoxic crosslinker common in mammals [25].
Brain disease models can be efficiently constructed for detailed investigation by collecting lesion cells. However, it is difficult and costly to collect iPSCs for certain classes of diseases. Studies primarily focus on the biocompatibility and porosity of the biomaterials used in primary neural cell cultures. Few works, however, investigate the mechanical properties of the biomaterials. Primary neural cells from lesion parts are more fragile than normal neural cells. Therefore, an environment that does not have specific parameters and one without proper mechanical materials can cause a large amount of apoptosis; this is why it is so difficult to form primary neural cell cultures
4 Biomaterials and biofabrication for neural stem cells
Biologists and therapists have paid increasing attention to NSCs for their unique advantages: their potential to differentiate into various types of neurons and glial cells, their capacity for rapid self-multiplication, and their abundance of sources and types. Due to these same characteristics, NSCs are vital in constructing artificial nervous systems, neuropathological and frug sieve models, and clinical, surgical implants. In native situations, NSCs have dynamic and complex interactions with the ECM. In artificial situations, the materials around the NSCs work as a substitute that aid in adhesion, migration, maintenance of stemness, proliferation, and directional differentiation. In this section, the materials for the simple proliferation and stemness maintenance of NSCs (2D and 3D), for the directionally differentiated multicellular neural constructs, and the tissue-like neural constructs are introduced in sequence, as illustrated in Fig. 5. This provides feasible choices to fulfill the need for the specific applications of NSCs and to enhance comprehension of the cell-material interactions in neural constructs.
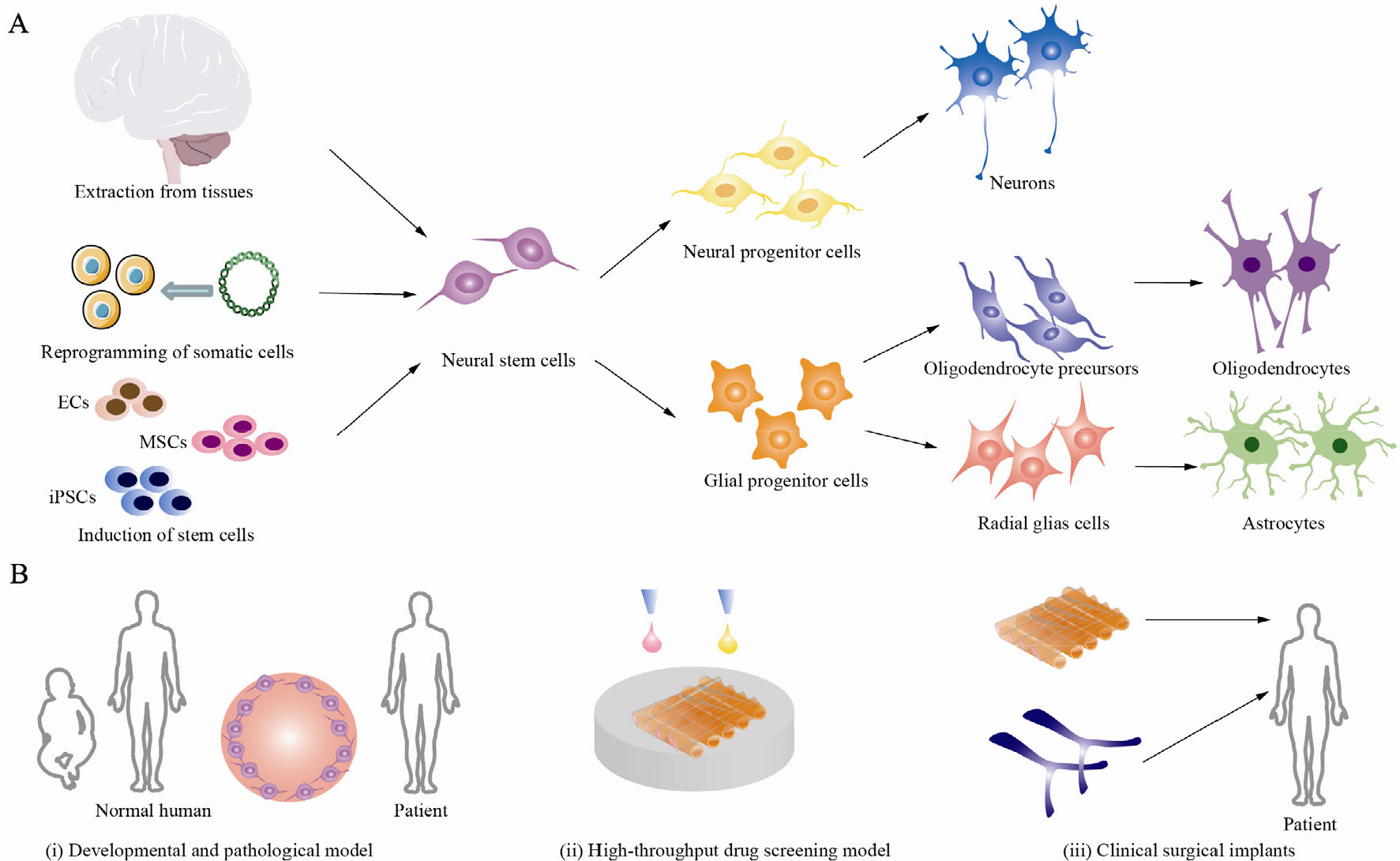
Different sources of NSCs and their downstream differentiation. (A) Three main sources and the downstream differentiation of NSCs. (B) Possible applications for NSCs: (i) the developmental and pathological models; (ii) the high-throughput drug screening model; (iii) and clinical surgical implants including brain patches and nerve scaffolds.
4.1 Culture models based on neural stem cells
Note that NSCs primarily exist in embryos in the early stages of development. Only a small number of neurogenic regions exist, and they have a very limited amount of NSCs in the adult central nervous system (i.e., the dentate gyrus [50] and the subventricular zone [51]). Reprogramming techniques are introduced to transform somatic cells into NSCs to collect sufficient NSCs for research and therapy. In addition, upstream cells such as ESCs, mesenchymal stem cells, and iPSCs are also candidates as sources of NSCs. Multiple materials have been used to maintain the stemness and promote the proliferation of NSCs in both the traditional 2D cultures and the 3D construct cultures.
Both 2D and 3D cultures are implemented in the fabrication of culture models. The 2D cultures are the most mature and common method for the proliferation of NSCs; however, the plane monolayer culture model increases the tendency of cell differentiation [52]. The biomaterials used in 2D cultures mainly promote the adhesion and migration of NSCs with the following optimized functions: accelerating the proliferation and survival rate, maintaining the stemness, and enhancing the differentiation into certain types of neurons and glial cells. In 2D cultures, biomaterials are most commonly used as a coating. Laminin, fibronectin, Matrigel, PDL/PLL, and poly-L-ornithine (PLO) are the most common materials for 2D culture coating [53]. The materials PDL/PLL and PLO are positively charged [54]. They all promote the adhesion of NSCs via electrostatic binding to negative cell membranes and by absorption of some functional proteins like laminin [55, 56]. The material PLL reportedly inhibits the differentiation of NSCs [57], while PLO promotes the differentiation into neurons and oligodendrocytes [58]. Laminin and fibronectin are natural composites in ECM [59], and they both promote the proliferation of NSCs through the integrin receptor [60] as well as encourage the differentiation of NSCs into neurons and oligodendrocytes [61]. Laminin is more powerful than PLO or fibronectin; however, the material mainly originates from animals, and recombinants are expensive [62]. Matrigel is a mixture extracted from mouse sarcomas that can significantly retain the stemness of NSCs [63]. However, the promotion of cell proliferation mainly comes from certain growth factors, so it is a time-limited function [64]. Moreover, Matrigel is not well defined, so the batch differences and biological immune responses are problematic. Due to these reasons, 3D cultures have multiple advantages over 2D cultures, because it offers extra space, expanding the interactions between the cells and the surrounding matter to better mimic the natural cell niche [65] and enhancing cell behaviors’ similarity between
In 3D cultures, the materials interact with the cells on the surface and throughout the entirety of the sample. A dense solid section with little interior space for material transportation and cellular communication suppresses the survival of NSCs [74]. Therefore, materials that can form porous or other similarly spaced structures benefit the proliferation of NSCs. Via freeze-drying, collagen forms porous scaffolds with axially aligned luminal channels; this promotes the extension of the NSCs loaded onto them [75].
For proliferation and stemness maintenance, the materials primarily function chemically and biologically. By offering specific factors that the NSCs require, natural materials such as laminin and Matrigel enhance cell proliferation and reserve pluripotency. With similar principles, certain varieties of engineered peptide materials are designed to interact better with NSCs [76]. As for the physical effects, the most significant one seems to be the stickiness. Better adhesion leads to more efficient proliferation and differentiation, and less adhesion retains the stemness. In addition, the biomaterial works differently in 2D and 3D culturing. In 3D cultures, the materials’ space for the NSCs and the nutrients is important.
4.2 Culture models for directionally differentiated multicellular neural constructs
With stable and sufficient NSCs, oriented induction is possible. An NSC has the potential to differentiate into a neuronal progenitor cell (the term used here is controversial as some researchers use neural progenitor cells to describe all classes of proliferating cells) [77] and a glial progenitor cell. Subsequently, these cells further differentiate into various neurons and glial cells, respectively [78]. However, in most cases, the differentiations happen uncontrollably. As mentioned above, basic coating materials such as laminin, fibronectin, and PLO tend to promote the differentiation of NSCs. Although growth factors and cytokines have a major influence on the manipulation of cell fate, the geometric and mechanical properties also matter.
Surface topography, especially structural orientation, promotes the differentiation into neurons [79]. In natural conditions, NSCs tend to migrate along radial glial cells to form the cerebral cortex [80], implicating that radial and aligned patterns promote the differentiation of NSCs by impacting their distribution and orientation. In addition, the nanosized topological structure can regulate the fate of NSCs through clustering integrin and focal adhesion proteins [81]. For nanometer-sized patterns, rigid materials like silicon and ceramics have advantages in manufacturing but lack the other biocompatible features that NSCs require. Some polymeric materials are flexible at high temperatures or in organic solvents. Therefore, some high-precision manufacturing methods (i.e., electrostatic spinning and near-field electrostatic direct writing) are feasible to build constructs with micrometer or sub-micrometer structures that facilitate and accelerate the neural differentiation of NSCs [82]. Photochromics (e.g., photosensitive resins and polyamides) are formed through photolithography and polydimethylsiloxane (PDMS) and enable soft nano-imprint lithography, which can achieve several micrometers and hundreds of nanometers of accuracy, respectively [83].
Stiffness is a crucial factor affecting the differentiation of NSCs. Neurons are more likely to form in softer gels with a stiffness range of 100–500 Pa, while glial cells form better in harder gels with a stiffness range of 1,000–10,000 Pa [84]. Although low stiffness benefits neural differentiation, it is an obstacle for construct fabrication. The stiffness of hydrogels is often adjustable. The material GelMA with photoinitiators enables photopolymerization [85] and can form hydrogels with a very low range of stiffness (160–900 Pa). It affects the viability of cells both in adherent culture as well as in scaffold culture [86]. Fibrin or fibrinogen is also used to manipulate the stiffness of the structure. The fibrinogen concentration determines the original elastic modulus, which can be adjusted through proteolytic material, aprotinin, or other similar materials during long-term culturing [87, 88]. In addition, by temporarily gelling the constructs with a supporting thermo-reversible gelatin network, several soft materials, (e.g., gelatin, alginate, hyaluronic acid, chondroitin sulfate, dextran, chitosan, heparin, and polyethylene glycol), can be formed in designed structures at low concentration and at low stiffness (thousands of pascals) [89].
For the differentiation of NSCs, materials still work in chemical and biological ways while considering the geometrical and mechanical ways. Here, the contradiction between the machinability and the characteristics of the material conforming to the ECM is highlighted. Materials that are easily fabricated into the required topography and structure are often rigid and lack the biocompatibility and elasticity that NSCs need. Therefore, certain new materials (e.g., PDMS) and innovative forming methods may solve this problem.
4.3 Culture models for tissue-like neural constructs
Building tissue-like constructs through NSCs is a hot topic attracting growing attention for its broad application in the construction of developmental and pathological models and even clinical patches. Organoids and bioprinted models are two main approaches to attain tissue-like constructs.
Organoids are 3D models that start with a stem cell colony in suspension, undergo self-assembly of different cells, and end with tissues that function similarly to organs [90]. Many different organoids, especially brain organoids, have been built with NSCs [91]. Matrigel is the most common biomaterial utilized in the forming and culturing of organoids for its similarity to the ECM
Different from the self-assembly of cells in organoids, bioprinting models enable more designed manipulation. As the brain is organized in six layers of cortical cells [95], layer-by-layer additive manufacturing is inherently suitable for constructing cortical-like models and cerebral functional columns. Moreover, 3D bioprinting meets the requirements of neural implants for its complicated customizable structure [96]. Building constructs with functional neural networks remains an unsolved problem to date. Multicomponent and adjustable material systems are essential to solving this research problem. An alginate/gellan gum/laminin hydrogel is proposed to influence the differentiation of iPSC-derived neural progenitor cells into neurons and astrocytes through stiffness reduction and stress relaxation. When this hydrogel is applied, spontaneous intracellular calcium signals are detected [97]. Conductivity is also an essential characteristic of the material systems, as neurons communicate through electrical signals
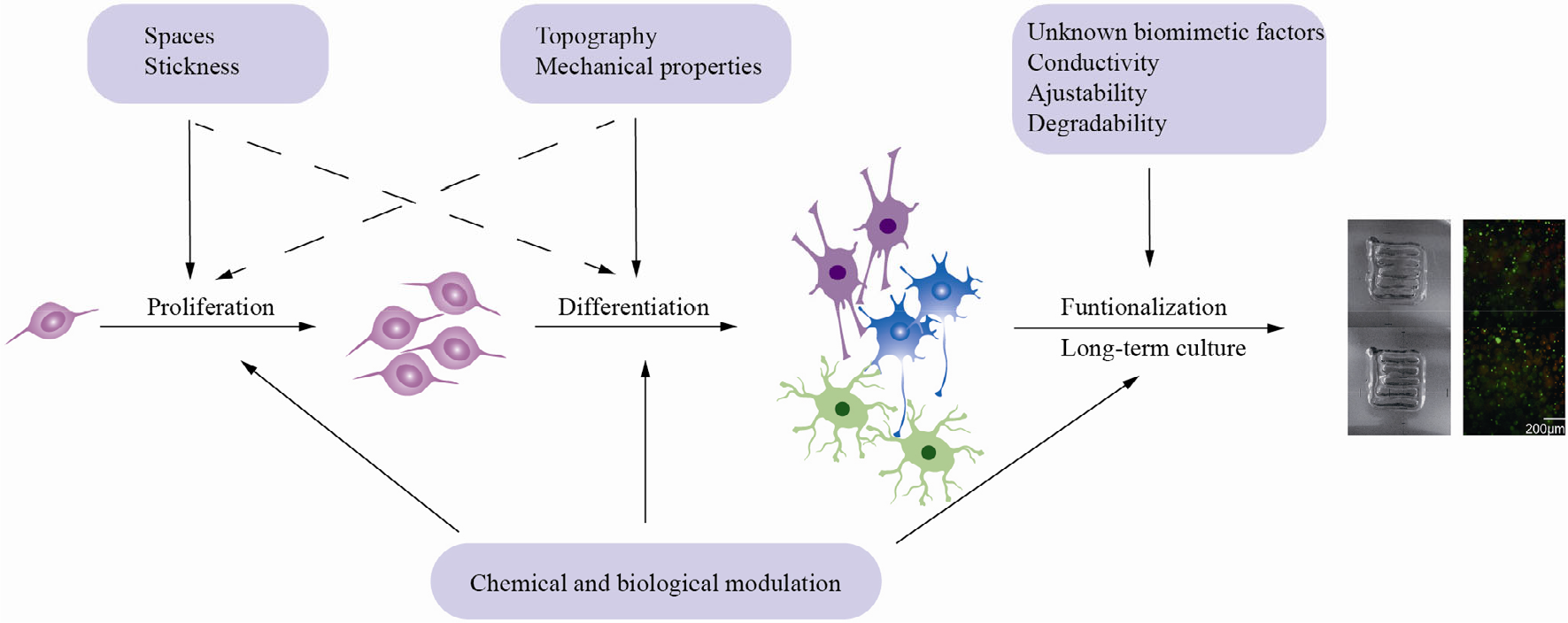
The interaction between materials and NSCs on a space–time scale. Eventually, functional constructs use vascularized brain organoids, bioprinted structures with some neural networks, and transplants into the spines of rats. Adapted with permission from Ref. [97].
In general, most materials used to construct organoids are complex mixtures extracted from natural ECM, which offer relatively comprehensive, with some unknown, supports for NSCs and their cell behaviors. However, a lack of regulation limits the more extensive applications of pure organoids; therefore, bioprinting and other fabrication methods are introduced to build more complex and controllable constructs with or without organoids. Functional properties such as conductivity and degradability play important roles at this level. In addition, the long-term controllability of the combination of multiple components is of great importance.
5 Conclusions
The purpose of summarizing the current culture models based on primary neural cells and NSCs is to determine the common characteristics of the biomaterials utilized. The demand for 3D culture models is higher than ever due to their beneficial potential application in individual medical treatment and the pharmaceutical industry as ideal regeneration models and drug screen models, respectively. However, after a literature review, it is realized that producing efficiently functioning 3D culture is difficult. This may be attributed to the unknown mechanical properties of the biomaterials. The modulus of brain tissue is ˜1 kPa; however, few studies pay attention to this. A failure to match the mechanical properties leads to quick degradation of the 3D constructs during
Footnotes
Conflict of interests
All contributing authors report no conflict of interests in this work.
Funding
This study was supported by 111 Project (Grant No. B17026), the National Nature Science Foundation of China (Grant No. 31700928), and the Chinese Postdoctoral Science Foundation (Grant No. 2017M610881).
