Abstract
Tobacco smoking is the leading preventable cause of morbidity and mortality worldwide. Although a number of smokers are aware of the adverse outcomes of smoking and express a strong desire to stop smoking, most smoking quit attempts end in relapse within the first few days of abstinence, primarily resulting from the aversive aspects of the nicotine withdrawal syndrome. Therefore, studying the neural mechanisms of smoking abstinence, identifying smokers with heightened relapse vulnerability prior to quit attempts, and developing effective smoking cessation treatments appear to be promising strategies for improving the success of quit attempts. In recent years, with the development of magnetic resonance imaging, the neural substrates of smoking abstinence have become extensively studied. In this review, we first introduce the psychophysiological changes induced by smoking abstinence, including affective, cognitive, and somatic signs. We then provide an overview of the magnetic resonance imaging-based evidence regarding abstinence-related functional changes accompanied by these psychophysiological changes. We conclude with a discussion of the neural markers that could predict relapse during quit attempts and a summary of the psychophysiological interventions that are currently often used to help with smoking cessation. This review extends our understanding of the role of the central nervous system in smoking abstinence.
1 Introduction
As the world’s largest tobacco manufacturing and consuming country, both the prevalence of tobacco use and the associated burden of morbidity and mortality are extremely high in China [1]. Cigarette smoking is linked to many negative health consequences, such as cancer, cardiovascular disease, and pulmonary disease [2]. Although a number of smokers are aware of the adverse outcomes of smoking and express a strong desire to stop smoking, most smoking quit attempts end in relapse within the first few days of abstinence, primarily resulting from the aversive aspects of the nicotine withdrawal syndrome, which remains poorly understood. Smoking withdrawal can lead to many psychophysiological changes, making it difficult for people to quit. Studies have demonstrated that smoking withdrawal-induced psychophysiological changes are key predictors of relapse to smoking [3–5]. As such, understanding the neurobiological mechanisms underlying nicotine withdrawal in the nicotine deprivation state is crucial to the development of effective smoking cessation treatment interventions that aim to improve smoking cessation rates.
Nicotine addiction is a brain disorder. The emergence of neuroimaging techniques, including magnetic resonance imaging (MRI), positron emission tomography, electroencephalography, and magnetoencephalography, has enabled researchers to noninvasively investigate the role of the central nervous system in smoking abstinence [6–9]. Of all these neuroimaging techniques, functional neuroimaging techniques, especially functional MRI (fMRI) based on the contrast of blood oxygen level-dependent (BOLD) signals, allow for the objective examination of brain mechanisms associated with smoking abstinence [10–17]. Moreover, studies that aimed to develop relapse predictors or smoking cessation treatment interventions have also applied neuroimaging techniques to associate brain responses with the changes in smoking withdrawal symptoms [18–21], thus providing a deeper understanding of the mechanisms of relapse and treatment effectiveness.
In this review, we first introduce smoking-abstinence-induced psychophysiological changes, including affective, cognitive, and somatic signs. We then provide an overview of the MRI-based evidence regarding abstinence-related functional changes accompanied by these psychophysiological changes. Finally, we summarize the possible neural markers that could predict relapse as well as the neural substrates of psychophysiological interventions that are often used to help facilitate smoking cessation (Fig. 1).
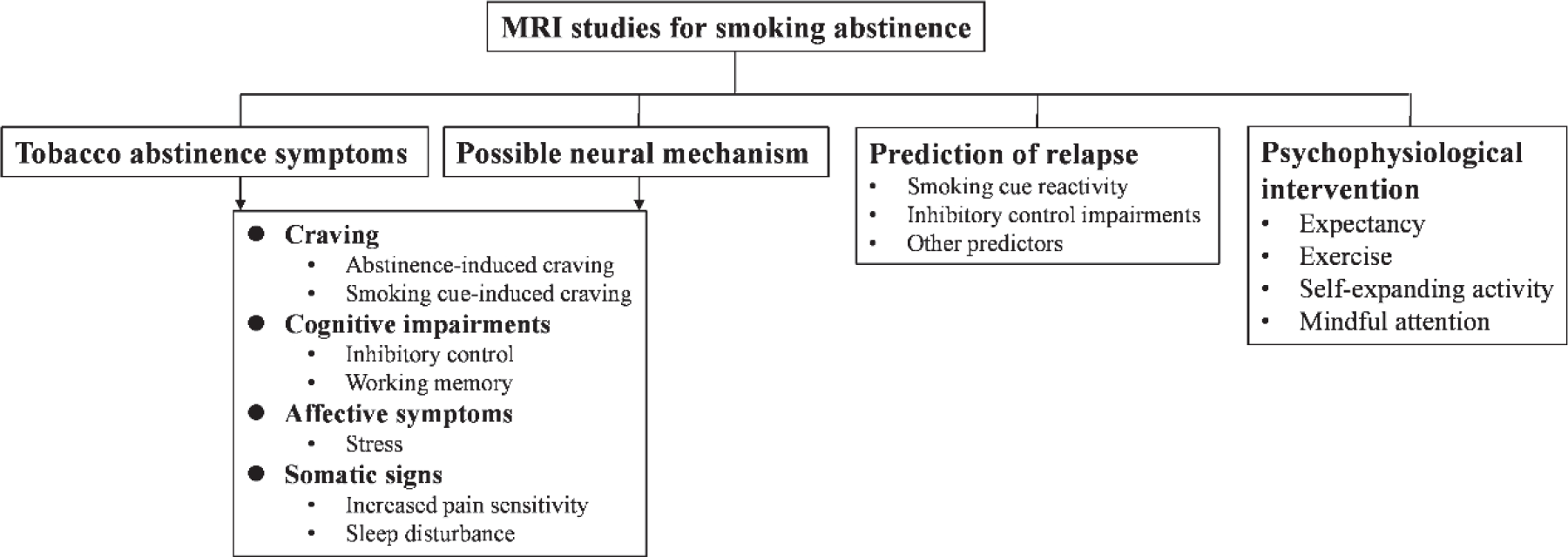
Schematic diagram of the framework of this review. MRI, magnetic resonance imaging.
2 Tobacco abstinence symptoms
Abstinence from chronic tobacco use produces a series of withdrawal symptoms, including affective, cognitive, and physical or somatic components [22]. Affective symptoms include craving and a depressed mood (e.g., depression, anxiety, irritability, restlessness, stress, and difficulty in concentrating). Cognitive components include decreased cognitive performance (e.g., inhibitory control and working memory). Somatic components include increased appetite or weight gain, sleep disturbance, and increased pain sensitivity. According to the duration of nicotine withdrawal, the symptoms of nicotine withdrawal can be divided into acute (7–10 episodes of withdrawal) and chronic (7–10 days after withdrawal) withdrawal symptoms. In general, nicotine withdrawal symptoms begin to appear as early as 4 hours after the last cigarette, peak in ~3 days to 1 week, and last for 2 to 4 weeks [22]. Some withdrawal symptoms, such as craving for cigarettes, are still measurable 1 month after the cessation of tobacco use [23]. Although abstinence-induced somatic signs are unpleasant for smokers, studies have demonstrated that of all withdrawal symptoms, affective and cognitive components play critical roles in the maintenance of nicotine dependence [24, 25].
3 Potential neural mechanisms of withdrawal symptoms
Nicotine, the main addictive substance in cigarettes, can produce profound physiological modulations by binding to nicotinic acetylcholine receptors (nAChRs) [26]. Chronic exposure to nicotine results in the desensitization and upregulation of nAChRs and neuroadaptations throughout the brain [27]. Research has shown that the desensitization and upregulation of nAChRs following chronic nicotine exposure are influential in producing withdrawal symptoms upon cessation of smoking [28–30]. In addition to evaluating the contribution of nAChRs in producing withdrawal symptoms, assessing brain changes may yield a better understanding of the neural substrates of smoking abstinence.
3.1 Craving
Craving is a prominent feature of nicotine dependence and is an important factor that can precipitate relapse during smoking quit attempts [3, 31]. After smoking cessation, craving develops rapidly, which is known as abstinence-induced craving. Abstinence-induced craving is sensitive to the effects of nicotine delivery. In addition to this unprovoked craving, smoking cue-induced craving, which is thought to arise from a behavioral conditioning process, in which stimuli associated with smoking trigger drug-seeking behavior, develops. Numerous studies have examined the neural substrates of cravings elicited by smoking abstinence [17, 32–35] and smoking-related cues during the abstinence state [14, 36–39] (Table 1).
Summary of smoking-abstinence-related magnetic resonance imaging studies.
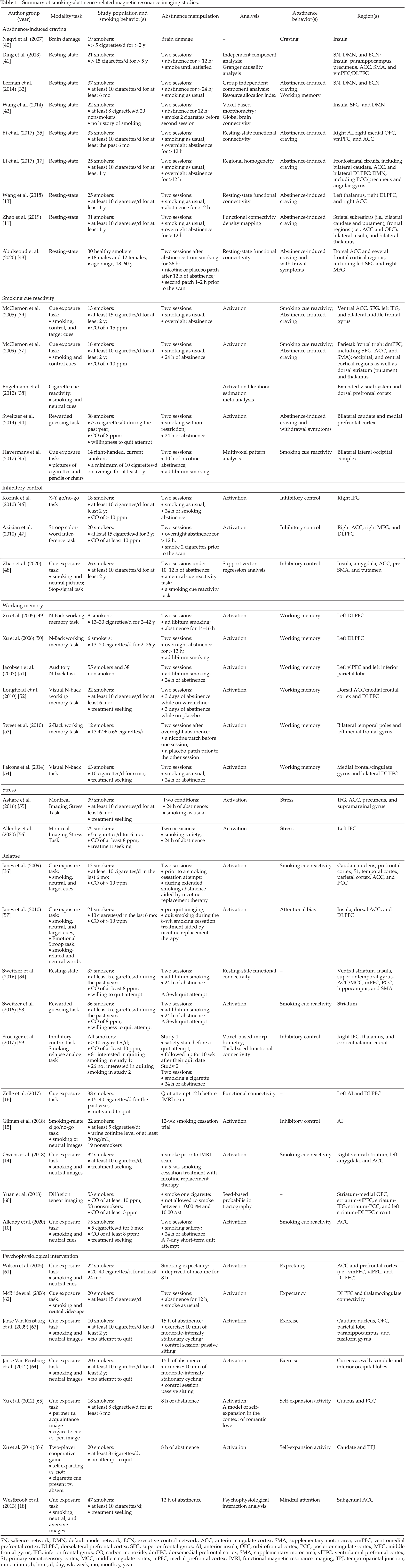
SN, salience network; DMN, default mode network; ECN, executive control network; ACC, anterior cingulate cortex; SMA, supplementary motor area; vmPFC, ventromedial prefrontal cortex; DLPFC, dorsolateral prefrontal cortex; SFG, superior frontal gyrus; AI, anterior insula; OFC, orbitofrontal cortex; PCC, posterior cingulate cortex; MFG, middle frontal gyrus; IFG, inferior frontal gyrus; CO, carbon monoxide; dmPFC, dorsomedial prefrontal cortex; SMA, supplementary motor area; vlPFC, ventrolateral prefrontal cortex; S1, primary somatosensory cortex; MCC, middle cingulate cortex; mPFC, medial prefrontal cortex; fMRI, functional magnetic resonance imaging; TPJ, temporoparietal junction; min, minute; h, hour; d, day; wk, week; mo, month; y, year.
3.1.1 Abstinence-induced craving
Abstinence-induced cigarette craving can promote relapse in abstinent smokers, and smoking a cigarette can rapidly reverse the symptoms that develop. The neural substrates of abstinence-induced craving have been extensively investigated using fMRI techniques [11, 13, 17, 33, 35, 67] [Fig. 2(a)]. The within-subject experimental design has been commonly used to measure abstinence-induced brain functional changes in smokers. During the experiments, smokers were scanned at resting state on two occasions, in a counterbalanced order: (1) smoking satiety, and (2) smoking abstinence (after several hours of smoking deprivation).
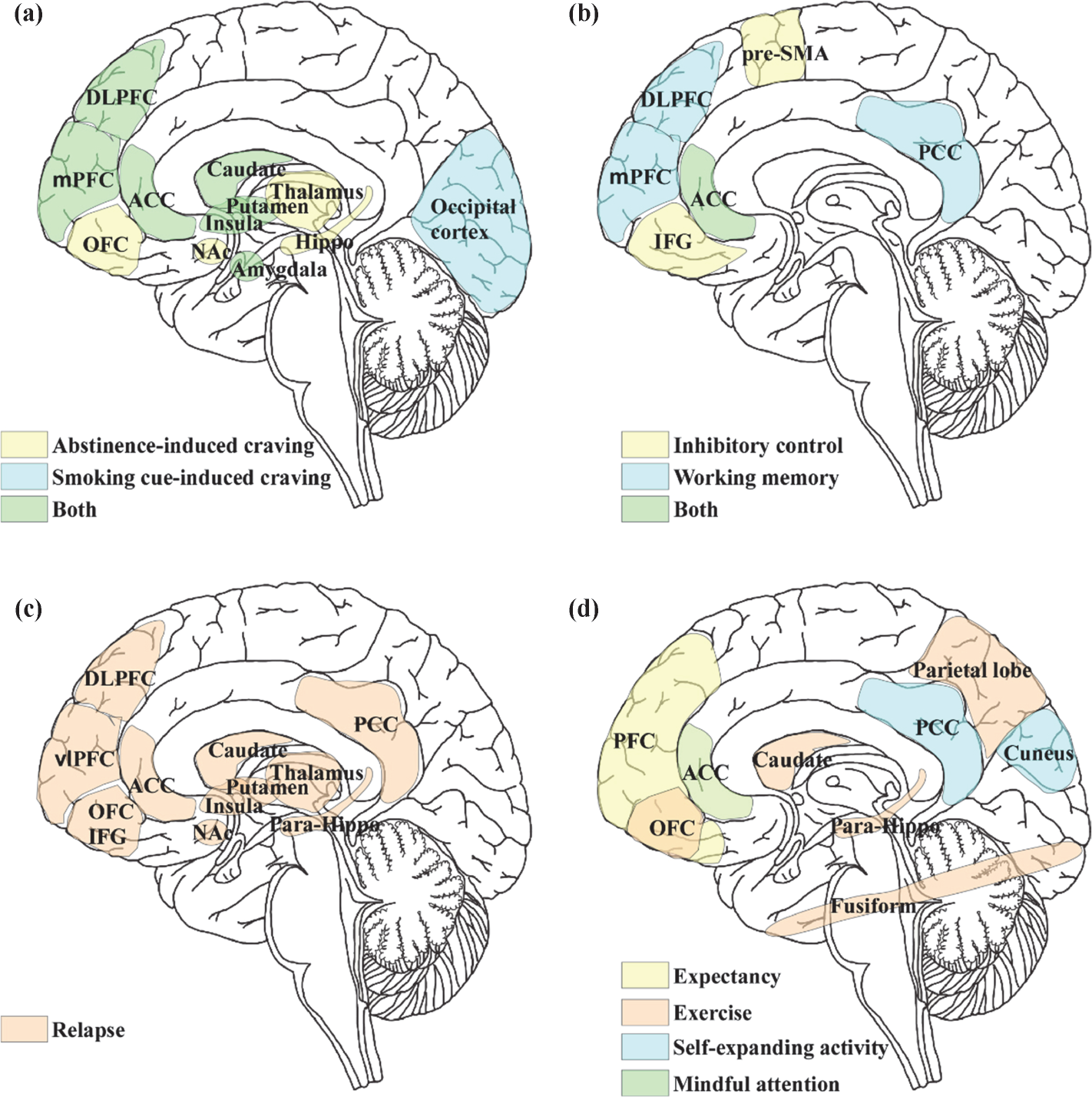
Brain regions implicated in (a) abstinence-induced craving as well as smoking cue-induced craving; (b) cognitive impairments; (c) prediction relapse during quit attempts; and (d) psychophysiological intervention effects. DLPFC, dorsolateral prefrontal cortex; mPFC, medial prefrontal cortex; ACC, anterior cingulate cortex; OFC, orbital frontal cortex; NAc, nucleus accumbens; pre-SMA, pre-supplementary motor area; PCC, posterior cingulate cortex; IFG; inferior frontal gyrus; vlPFC, ventrolateral prefrontal cortex; para-Hippo, parahippocampus.
Resting-state brain functional changes induced by smoking abstinence have been usually examined by measuring spontaneous brain activity, circuitry connectivity, and network connectivity using resting-state fMRI. Zhao et al. [11] used the functional connectivity density mapping method and found that 12-hour abstinence (vs. satiety) induces higher local functional connectivity density and global functional connectivity density in regions commonly implicated in nicotine addiction, which include the striatal subregions (i.e., bilateral caudate and putamen), frontal regions [i.e., anterior cingulate cortex (ACC) and orbital frontal cortex (OFC)], insula, and thalamus. A lesion study demonstrated that damage to the insula disrupts smoking behavior in stroke patients [40], as characterized by their ability to quit smoking easily, immediately, without relapse, and without the persistence of the urge to smoke. This observation emphasizes the critical role of the insula in the maintenance of smoking addiction. Increased circuit connectivity of the insula with the parahippocampus, precuneus, ACC, supplementary motor area, and ventromedial prefrontal cortex (PFC)/dorsolateral PFC (DLPFC) was also detected in the abstinence state relative to the satiety state using seed-based resting-state functional connectivity analysis [35, 41]. In particular, the abstinence-induced resting-state functional connectivity changes between the right anterior insula and right lateral OFC were found to be significantly correlated with craving changes [35], which contributes new insights into the functional role of the insula in abstinence-induced craving.
Cigarette smoking exerts its initial reinforcing effects by activating reward circuits and releasing dopamine in the striatum [68, 69]. Long-term cigarette smoking triggers dopamine system dysfunctions and the neuroplasticity of the frontostriatal circuits associated with craving and cognitive control [17, 60, 70–72]. Converging lines of evidence demonstrated the functional changes in these circuits during withdrawal as well as their relationship with abstinence-induced craving [17, 34, 43]. For example, higher regional homo-geneity in the frontostriatal circuits, including the bilateral caudate, ACC, and bilateral DLPFC, was detected in the abstinence state (> 12 hours) relative to the satiety state. Moreover, the regional homogeneity changes in the ACC and bilateral caudate were found to be positively correlated with abstinence-induced craving changes in young adult smokers [17]. In addition, with the dorsal ACC (dACC) regarded as the seed, Abulseoud et al. [43] found an increase in resting-state functional connectivity between the dACC and several frontal cortical regions, including the left superior frontal gyrus and right middle frontal gyrus, after 36 hours of nicotine abstinence. Moreover, the change in circuit strength between the dACC and left superior frontal gyrus was found to be negatively correlated with the changes in withdrawal symptoms, including craving. The ventral and dorsal striatum are key neural substrates of reward processing and motivation, and they have been repeatedly linked to nicotine dependence [60, 71–73]. Researchers have examined the effects of abstinence on brain connectivity with ventral and dorsal striatum seed regions and observed robust abstinence-induced changes (24 hours of abstinence) in the connectivity of both ventral and dorsal striatum with a network of regions implicated in addictive disorders. These findings suggest the critical role of the frontostriatal circuits in abstinence-induced craving [34].
The human brain is a complex patchwork of interconnected regions. Recent literature has indicated that not only the activity within each functional network but also, and especially, the interaction between these networks is critical for elucidating abstinence-induced craving and cognitive impairments [74]. Three networks have received the most attention in smoking addiction, namely, an executive control network (ECN) implicated in attention to and processing of exogenous stimuli, a default mode network (DMN) involved in stimulus-independent thought processes (e.g., self-referential thinking), as well as a salience network (SN) facilitating orientation to external (ECN) and internal (DMN) information processing and allocating attention. Sutherland et al. [74] proposed that, in the nicotine-deprived state, the SN allocates enhanced attentional resources toward internal symptoms of withdrawal, thereby biasing activity toward the DMN and away from the ECN. In line with this hypothesis, researchers have observed higher brain activity/connectivity within the DMN [17, 42], weaker inhibition between the DMN and SN, as well as lower connectivity between the ECN and SN [41] in the abstinence state (> 12 hours of abstinence) relative to the satiety state. Furthermore, the weaker internetwork connectivity between the DMN and SN could predict abstinence-induced cravings to smoke and less suppression of DMN activity during the performance of a subsequent working memory task [32]. Therefore, alterations in the coupling of the SN and DMN as well as the inability to disengage from the DMN may be critical in cognitive/affective alterations that underlie nicotine dependence.
Using arterial spin labeled perfusion MRI technique, mixed results were reported across studies, possibly due to differences in their sample size, duration of smoking deprivation, methodologies, and individual differences, such as variants in the dopamine D2 receptor [75]. For example, Wang et al. [33] detected increased cerebral blood flow (CBF) in the ACC/medial OFC and left OFC in the abstinence state relative to the satiety state after 12 hours of smoking deprivation. Abstinence-induced cravings were predicted by CBF increases (abstinence minus satiety) in regions commonly implicated in visuospatial and reward processing, such as the right OFC, right DLPFC, occipital cortex, ACC, ventral striatum/nucleus accumbens, thalamus, amygdala, bilateral hippocampus, left caudate, and right insula. On the other hand, another study using a shorter period of abstinence manipulation (after 4 hours of nicotine deprivation) found significantly reduced regional CBF in the hippocampus and ventral striatum. Similar to prior research, the abstinence-induced regional CBF changes in the medial and lateral OFC and anterior ventral insula were also found to be correlated with the magnitude of the change in craving [76], confirming the role of the OFC and insula in abstinence-induced craving. Furthermore, given the role of the ventral striatum, thalamus, and medial frontal cortex in nicotine addiction, a study that focused on regions of interest in these three regions did not find significant CBF changes after smoking abstinence. Despite this negative result, nicotine replacement could significantly increase CBF in the ventral striatum, and a significant correlation between the increase in withdrawal symptoms and a reduction in thalamic CBF was detected [67].
3.1.2 Smoking cue-induced craving
Frequent pairings of the visual, tactile, and olfactory sensations of smoking with the rewarding effects of nicotine result in a classical conditioning effect, such that exposure to smoking cues (e.g., the sight and smell of a burning cigarette, cigarette packs, and lighters and smoking-related pictures and videos) even for several seconds or minutes can elicit a strong urge to smoke and promote tobacco use in addictive smokers, especially when they are abstaining from smoking. Indeed, abstinent smokers experience greater difficulty in suppressing their desire to consume cigarettes when they are exposed to these salient smoking cues [38]. Smoking-related cues may be an important factor that promotes relapse among smokers who are trying to quit.
In typical cue reactivity paradigms, brain responses between conditions using smoking-related and neutral cues are compared. Studies using fMRI have indicated that smoking cues reliably evoke larger brain responses in the extended visual system, precuneus, posterior cingulate gyrus, anterior cingulate gyrus, dmPFC, insula, and dorsal striatum than neutral cues in smokers [38]. Under nicotine deprivation, smoking abstinence significantly potentiates neural responses to smoking-related cues versus neutral cues in brain regions subserving reward processing, visual sensory processing, attention, and action planning [36, 48], including the striatum, occipital cortex [45], insula, amygdala, ACC, and putamen [48] [Fig. 2(a)]. In detail, drug use, including smoking, is associated with dysregulated reward processing, attentional bias to smoking-related cues, as well as dysfunctional inhibitory control and action planning [37, 44, 45, 58, 77]. This dysregulated processing is further exacerbated during deprivation from smoking. For example, an increased BOLD response (e.g., striatum) in anticipation of smoking reward, decreased BOLD activation in anticipation of monetary reward [44, 58], and elevated lateral occipital complex activation in response to smoking-related cues [45] were detected after a period of nicotine deprivation. Furthermore, attenuation of monetary reward-related activation and increased smoking cue activation in the dmPFC, which is involved in action planning and decision making [37], during abstinence were found to be significantly correlated with abstinence-induced increases in craving and withdrawal [37, 44] and to potentially predict smoking lapse during a quit attempt [58]. These findings suggest that smoking abstinence can evoke hyperactive brain reactivity to smoking-related cues and that this heightened brain reactivity is a precipitating factor in smoking relapse. Thus, reducing reactivity to smoking cues appears to be a promising strategy for improving the success of quit attempts.
3.2 Cognitive impairments
Acute nicotine use can produce short-term cognitive enhancement [78, 79]. However, long-term cigarette smoking would induce cognitive impairment and cognitive decline, especially during nicotine withdrawal [80, 81]. Cognitive impairments, including inhibitory control and working memory impairments, are commonly reported in abstinent smokers [49, 50, 52, 53, 82] and contribute to greater relapse vulnerability [5]. Understanding the neurobiology of abstinence-induced cognitive impairments using fMRI paradigms is of great interest to improve tobacco dependence treatment outcomes [Fig. 2(b) and Table 1].
3.2.1 Inhibitory control
Long-term cigarette smoking was found to be associated with decrements in inhibitory control, especially during withdrawal [46]. The attenuated inhibitory control of smokers is a stumbling block for treating nicotine dependence. Response inhibition, an important form of behavioral impulse control, is the ability to inhibit behavioral responses to salient approach cues. The go/no-go task, Stroop color-word task, and stop-signal task are common measures of response inhibition and have highlighted inhibitory control deficits in smokers [46–48, 83]. Using a go/no-go task, Kozink et al. [46] examined neural correlates of response inhibition and error monitoring in abstinent smokers and found that smoking abstinence (after 24 hours of abstinence) robustly increased errors of commission in stop trials and event-related BOLD signals in the right inferior frontal gyrus. Previous fMRI research using a Stroop color-word paradigm has also demonstrated that smokers had significantly higher task-related activity in the right ACC after overnight abstinence from smoking (> 12 hours of abstinence) compared with the satiety state [47]. Two other studies using a stop-signal task detected similar greater neural activation in the inferior frontal gyrus during successful response inhibition in abstinent smokers [83] and a hyperactive right pre-supplementary motor area during reactive inhibition in smoking cue versus neutral cue conditions [48]. All these findings suggest that successful inhibitory control during withdrawal is associated with increased cognitive processing demands on a cortical center associated with attention to inhibitory signals [46].
3.2.2 Working memory
When smokers abstain from smoking, specific deficits in working memory emerge [50, 54, 80] and promote smoking relapse [84]. For example, abstinence (versus smoking) was associated with longer response latencies and reduced accuracy of working memory performance [50, 54]; these performance decrements were more severe with earlier age of onset of smoking and in male smokers than in female smokers [80]. Many investigators have employed fMRI paradigms, such as the visual N-back working memory task, to examine brain functions associated with working memory deficits during the abstinence state. Researchers have observed that these deficits are accompanied by reduced activation in executive control regions (e.g., DLPFC) [54] and less suppression of activation in task-independent regions (e.g., posterior cingulate cortex) [5, 32, 53]. Furthermore, these working memory-related brain activities could predict subsequent smoking relapse [5]. Inconsistent results on whether overnight abstinence (> 14 hours of abstinence) was associated with higher task-related neural activity in the left DLPFC at the 1-back level following abstinence as compared with smoking, which indicates that neural processing related to working memory in the left DLPFC is less efficient during acute abstinence than in the satiety state, have also been reported [49]. In one study, a task with a high working memory load in the context of smoking abstinence required more cognitive processing demands, as reflected by greater activation of the working memory neurocircuit during the task [51]. nAChRs play a key role in cognition. Evidence has shown that cognitive deficits could be reversed by treatment with the α4β2 nicotinic receptor partial agonist varenicline by increasing working memory-related brain activity in the dACC/medial frontal cortex and bilateral DLPFC [52]. This observation provides a method to improve tobacco dependence treatment outcomes.
3.3 Affective symptoms
3.3.1 Stress
Subjective stress is considered to be a well-documented predictor of early smoking relapse. Thus, understanding the neural correlates of stress during acute nicotine withdrawal is important to intervene in stress-induced relapse. The Montreal Imaging Stress Task is a psychosocial stress task that requires solving difficult mental arithmetic problems while receiving negative performance feedback. Researchers have found that taking the Montreal Imaging Stress Task while undergoing fMRI not only increased subjective measures of stress [55] but also induced significantly greater activation in the inferior frontal gyrus, ACC, precuneus, and supramarginal gyrus in deprived smokers (24 hours of abstinence compared with the satiety state) [56] and non-deprived smokers [55] (Table 1). Further correlation results revealed that an abstinence-induced change in the brain response to stress is positively associated with a change in self-reported stress [56]. Therefore, investigating stress-related activation during nicotine withdrawal may identify those who are most vulnerable to relapse and represent a target for novel pharmacological intervention.
3.4 Somatic signs
3.4.1 Increased pain sensitivity
Cigarette smoking serves as a nicotine delivery vehicle in humans. Experimental studies have suggested that nicotine has analgesic properties [85]. Acute nicotine intake can suppress pain among animals and humans [85]. However, chronic nicotine exposure may alter pain processing and contribute to the development of chronic pain [86]. The pain processing alterations are pronounced especially when individuals quit smoking or are abstinent from smoking. Both clinical and experimental studies have consistently found enhanced pain perception in chronic smokers when deprived of smoking [87, 88]. Furthermore, clinical evidence has shown that, in addition to and in line with their increased pain sensitivity, patients deprived of cigarettes require a higher quantity of postoperative analgesics compared with nonsmoking patients [87]. Therefore, revealing the mechanisms underlying the effect of nicotine withdrawal on pain perception not only is of scientific interest but also has clinical relevance in the practice of anesthesiology and pain medicine. Currently, the underlying psychophysiological bases of this hypersensitivity are not completely understood. Future research using fMRI to explore the neural pathways responsible for these effects is needed.
3.4.2 Sleep disturbance
Subjective quality of sleep, which includes less total sleep time, longer sleep onset latency, increased difficulty in falling asleep as well as maintaining sleep, and waking up earlier than desired, is impaired in smokers compared with nonsmokers [89]. Several lines of evidence suggest that smoking abstinence during nicotine withdrawal not only can induce craving, negative affect, cognitive impairments, and increased pain sensitivity but also often worsens sleep [22]. Sleep disturbance may contribute to the risk for depressed mood and decreased smoking cessation outcomes [90]. However, the neural mechanism of abstinence-induced sleep disturbance remains poorly understood. Further research should focus on the neurobiological basis and consequences of sleep disturbance during tobacco cessation.
4 Prediction of relapse
Relapse to smoking after an initial abstinence period is a major challenge during smoking cessation. Identifying smokers with heightened relapse vulnerability during quit attempts could enable treatment personalization and improve smoking cessation treatment outcomes. Relapse-vulnerable smokers can be identified prior to a quit attempt. By measuring pre-quit behavior, brain activity, and connectivity, researchers have found that smokers who relapse have both greater smoking cue reactivity and lower inhibitory control compared with those who remain abstinent [15] [Fig. 2(c) and Table 1].
4.1 Smoking cue reactivity
A 9-week smoking cessation treatment study revealed that the neural response to smoking cues was greater in the insula, DLPFC, posterior cingulate, parahippocampus, putamen, thalamus, and cerebellum in participants who lapsed at any point during the subsequent smoking cessation treatment [57]. Another research compared smoking cue reactivity during acute abstinence with that in the satiety state and found that participants who showed greater smoking cue reactivity in the ACC during acute abstinence were more likely to relapse [10]. Greater abstinence-induced change in ACC activation also predicted fewer days of abstinence. These results suggest that lower smoking cue reactivity prior to smoking cessation or during acute abstinence may be associated with successfully maintaining abstinence during treatment.
4.2 Inhibitory control impairments
Several studies have highlighted the role of impaired inhibitory control in promoting relapse during smoking cessation. Gilman et al. [15] designed a go/no-go inhibitory control task in the presence of smoking-related cues and found that smokers who remained abstinent at the end of a 12-week smoking cessation program had greater neural activation in the anterior insula during no-go trials with smoking-related cues compared with those who relapsed. These findings indicate that decreased inhibitory control activation during exposure to smoking cues may be a marker of difficulty in quitting and relapse vulnerability in cigarette smokers [15]. Two other reports supported this result and observed that smokers who slipped had decreased functional connectivity between the insula and brain regions involved in cognitive control, including the dACC and DLPFC, possibly reflecting reduced top-down control of smoking-related cue-induced emotions [16, 57]. Moreover, an investigation in two cohorts of smokers found that greater activation in the inhibitory control circuitry (e.g., right inferior frontal gyrus) was associated with quicker relapse to smoking [59], indicating that the neural response to inhibition may be a potential marker that determines whether a patient is likely to attain long-term abstinence.
4.3 Other predictors
With the striatal circuits being critical substrates of reward processing and motivation in nicotine dependence, their dysfunction has been implicated in smoking behaviors and lapses during quit attempts [17, 60, 70–72]. Yuan et al. [60] observed that abstinence-induced increases in craving were associated with the tract strength of the left striatum-medial OFC and striatum-ventrolateral PFC in abstinent smokers. Notably, the tract strength of the left striatum-DLPFC predicted lapse in smokers with an accuracy of 68.3% [60]. In addition, a functional study revealed that 24-hour abstinence induced decreased functional connectivity between the ventral striatum and a network of regions implicated in addictive disorders, including the insula, superior temporal gyrus, and anterior/mid-cingulate cortex, among smokers who later lapsed, whereas the opposite pattern was observed among non-lapsers. These findings suggest that modulation of striatal connectivity with the cingulo-insular network during early withdrawal may be associated with smoking cessation outcomes [34].
5 Psychophysiological interventions
Considering the high relapse rate among smokers who attempt quitting without assistance, identifying ways to reduce the adverse effects of smoking abstinence may be important in increasing the success of quit attempts. Currently, numerous therapies have been extensively applied to increase the smoking cessation rate, including pharmacotherapy (i.e., nicotine replacement therapy, bupropion therapy, and varenicline therapy) [9, 20, 21, 91]; neuromodulation (i.e., transcranial magnetic stimulation and transcranial direct current stimulation) [19, 92–94]; and psychophysiological interventions (i.e., expectancy, exercise, self-expanding activity, mindful attention, and emotion regulation strategy) [18, 61–64, 95]. Among these intervention therapies, psycho-physiological interventions for reducing smoking cue reactivity and cravings play an important role in potentially disrupting the maintenance of cigarette smoking owing to their convenience, low cost, and ease of implementation. The neural substrates underlying these psychophysiological interventions have been examined using fMRI [18, 61–64, 95] [Fig. 2(d) and Table 1].
5.1 Expectancy
Cues associated with cigarette smoking can trigger craving, tobacco seeking, and relapse in deprived smokers. Behavioral studies have suggested that expectations regarding the opportunity to smoke a cigarette influence the pattern of neural responses to smoking cues, which ultimately modulates the level of craving to smoke [61, 62]. In people expecting to smoke immediately after an MRI scan, smoking cues activated brain areas implicated in arousal, attention, and cognitive control. However, when subjects knew that they would not be allowed to smoke for 4 hours, almost no brain activation in response to smoking cues was detected, despite equivalent reported levels of craving. In particular, subregions of the PFC (i.e., ventromedial, ventrolateral, and dorsolateral) were found to exhibit cue-elicited activation that was modulated by smoking expectancy [62]. Therefore, smoking expectancy could regulate craving to smoke by modulating smoking cue reactivity. For individuals seeking treatment to quit smoking, decreasing their expectation of an opportunity to smoke a cigarette might be an optional strategy to help them quit smoking.
5.2 Exercise
Exercise has been found to be an effective non-pharmacological method of reducing cigarette cravings and withdrawal symptoms among smokers attempting to quit smoking and among temporarily abstinent smokers [63, 64]. Evidence has demonstrated that the cravings, withdrawal symptoms, and adverse effects in abstinent smokers were significantly reduced during and following exercise [96]. Further post-exercise (i.e., using the Monark cycle ergometer) scanning showed hypoactivation in areas related to reward (caudate nucleus), motivation (OFC), and visuospatial attention (parietal lobe, parahippo-campus, and fusiform gyrus) with a concomitant shift of activation toward areas identified in the default mode network during smoking cue exposure [63]. Rensburg et al. [63] suggested that brief bouts of moderately intense physical activity have the potential to downregulate smoking cue reactivity and thereby possibly reduce cue-elicited cravings.
5.3 Self-expanding activity
With regard to dysfunctional reward processing during abstinence, researchers have found that engaging in novel/exciting (“self-expanding”) activities (e.g., learning a new skill, taking an exciting course, and engaging in romantic love) can potentially substitute for the reward from nicotine by activating the mesolimbic dopamine pathway [65, 66]. This self-regulation strategy effectively attenuates cigarette cue reactivity among nicotine-deprived smokers and appears to be beneficial as an aid in smoking abstinence and cessation [66]. Using a model of self-expansion in the context of romantic love, Xu et al. [65] observed that among nicotine-deprived smokers who were experiencing moderate levels of craving, cigarette cue reactivity regions (e.g., cuneus and posterior cingulate cortex) showed significantly less activation during self-expansion conditions compared with control conditions. These results provide evidence that rewards from self-expansion can act as a substitute for the reward from nicotine to attenuate cigarette cue reactivity. Future research could focus on the parameters of self-expanding activities that produce this effect and test the utility of self-expansion in clinical interventions for smoking cessation.
5.4 Mindful attention
Mindfulness is often defined as attention to a moment-to-moment experience coupled with a nonjudgmental, accepting attitude toward that experience. An emerging body of research suggests that mindfulness-based treatment is beneficial for smoking cessation, possibly by alleviating emotional states, such as distress, and therefore reducing cue-induced cravings [18]. Westbrook et al. [18] confirmed this hypothesis in treatment-seeking smokers (12 hours of abstinence). They found that mindful attention could significantly reduce smokers’ cue-induced cravings as well as neural activation in a craving-related region of the subgenual ACC and its connectivity to other craving-related regions compared with nontreatment conditions, suggesting that mindfulness may decouple the craving neurocircuitry in smokers when viewing cues [18].
6 Prospects and conclusion
Although the smoking relapse biomarkers and strategies of psychophysiological interventions to help smoking cessation reported to date are promising, some issues remain to be determined. First, neuroimaging-based biomarkers for smokers with heightened relapse vulnerability prior to quit attempts have been developed, but their clinical applications have yet to be fully explored. Translating laboratory findings to the clinical setting should be carefully carved in the future. Second, regarding the convenience, low cost, and ease of implementation of psychophysiological interventions, combining these intervention strategies with traditional pharmacotherapy or neuromodulation therapies might improve smoking cessation outcomes. However, psycho-physiological intervention-related studies are limited, and several intervention details or parameters should be investigated in future studies. For example, the extent to which the observed intervention effects may change or evolve over time is unclear. Whether exercise treatments with varying intensity, time, and frequency differentially affect smoking quit attempts remains unknown. Whether less intense but widely experienced self-expansion activities (e.g., interactions with friends and family members) as well as self-expansion at the individual level (e.g., engaging in a new sport or hobby or in spiritual experiences) would be useful to help smokers quit remains to be discovered. Therefore, further research that systematically and conclusively evaluates these questions should be performed to help improve smoking cessation outcomes.
In summary, the rapid development of neuroimaging techniques has significantly extended our understanding of the role of the central nervous system in smoking abstinence. Notably, among the smoking withdrawal symptoms, increased craving to smoke (especially smoking cue-induced craving) and decreased cognitive performance mainly contribute to heightened relapse vulnerability during quit attempts. Identifying smokers with heightened relapse vulnerability prior to quit attempts and using psychophysiological treatments to reduce the neural response to smoking cues appear to be promising strategies for improving the success of quit attempts.
Footnotes
Conflict of interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by the National Natural Science Foundation of China (Nos. 31800926, 32071061, 31822025) and the Scientific Foundation of Institute of Psychology, Chinese Academy of Sciences (No. Y8CX351005).
