Abstract
Pediatric patients are more likely to suffer from brain tumors. Surgical resection is often the optimal treatment. Perioperative management of pediatric brain tumor resection brings great challenges to anesthesiologists, especially for fluid therapy. In this case, the infant-patient was only 69-day-old, weighed 6 kg,but she was facing a gaint brain tumor (7.9 cm × 8.1 cm × 6.7 cm) excision. The infant was at great risks such as hemorrhagic shock, cerebral edema, pulmonary edema, congestive heart failure, coagulation dysfunction, etc. However, we tried to use the parameters obtained by bioreactance-based NICOM® device (Cheetah Medical) to guide the infant’s intraoperative fluid therapy, and successfully avoided these complications and achieved a good prognosis.
1 Introduction
Pediatric brain tumors are considered the second most common pediatric neoplasm, and about 50% of them are nonmalignant [1]. Currently, the best strategy for most of these tumors is radical resection as soon as possible [2], due to the intracranial pressure elevation, brain nerve damage, and even life-threatening conditions resulting from intracranial tumors (> 5 cm in diameter) in children. However, the intraoperative mortality rate remains high [3], especially in children with low weight and life-threatening conditions such as blood loss and coagulopathy, which made it a great challenge for anesthesiologists. Notably, the traditional static parameters such as heart rate (HR), arterial blood pressure (BP), and central venous pressure (CVP) are unreliable to predict fluid responsiveness [4]. Goal-directed fluid therapy (GDFT) based on the functional hemodynamic parameters provided by the cardiac output (CO) monitor has been proven to reduce the incidence of postoperative complications in adults [5, 6]. However, because of technical constraints of age and weight, few hemodynamic monitoring devices could be used during surgery for children [7]. The traditional hemodynamic monitoring equipment, which can be used in children, such as pulmonary artery floating catheters and esophageal ultrasound, is difficult to be used during neurosurgery due to the limitation of patients’ position. Recently, NICOM® device (Cheetah Medical) as a simple, safe, and completely noninvasive equipment has received the attention and application of many clinicians. Studies have proved that the hemodynamic parameters provided by NICOM based on bioreactance technology have acceptable reliability [8, 9]. Thus, we used NICOM in our case to give a successful and precise control of fluid input.
2 Case presentation
A 69-day-old infant who weighed 6 kg was scheduled for an excision of a giant brain tumor (7.9 cm × 8.1 cm × 6.7 cm) adjacent to the third ventricle of cerebrum, which was confirmed by magnetic resonance imaging (MRI) (Fig. 1). After her birth, the tumor grew rapidly; thus, the neurosurgeons decided to remove the tumor by surgery after 17 days of chemotherapy.
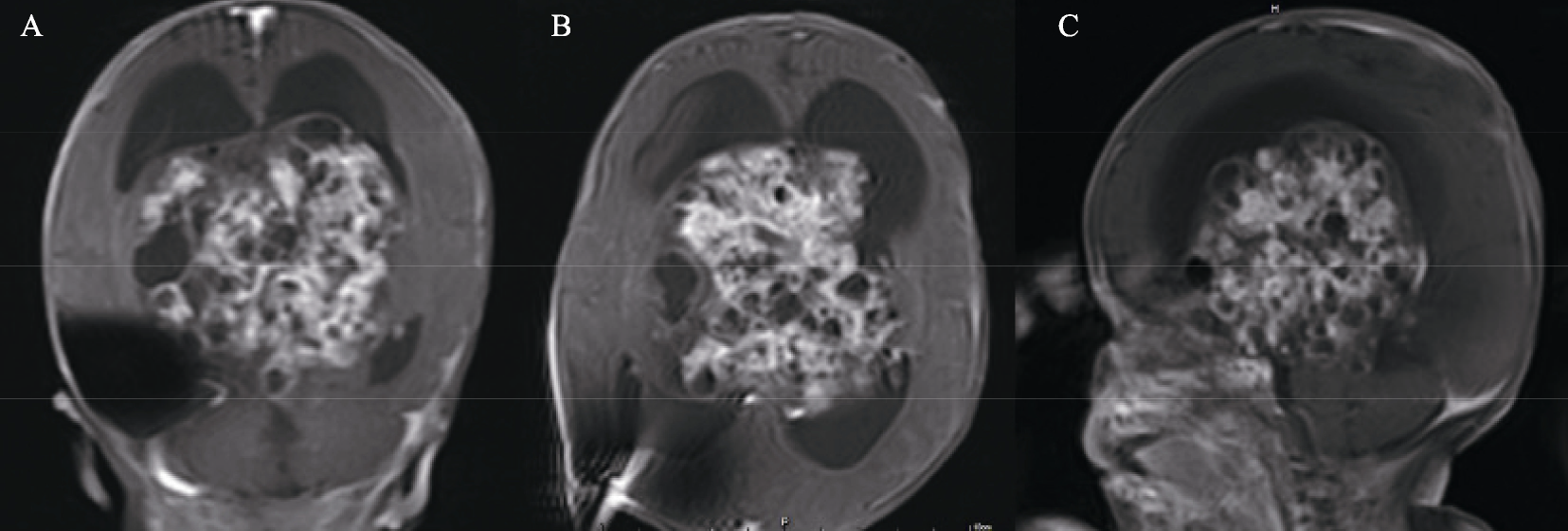
The MRI in the coronal plane (A), axial view (B) and sagittal plane (C) of the patient before operation, confirming the occurrence and size of the tumor.
The results of tests including preoperative electrocardiograph, complete blood count, and checks of serum creatinine, liver functions, and serum electrolytes were normal. 6 h before her arrival at the operating room (OR), the baby was fed with breastmilk. After her entry, electro-cardiogram, pulse oximeter, bispectral index monitor, and a NICOM® system (Cheetah Medical, UK) were set up. Her baseline vital signs included BP of 118/85 mmHg, HR of 144 beats/min, and pulse oxygen saturation (SpO2) of 99%.
After sevoflurane anesthesia, venous and radial artery catheters were applied, then general anesthesia was induced with intravenous administration of 0.3 μg/kg sufentanil, 0.2 mg/kg cisatracurium, and 5 mg/kg propofol, and tracheal intubation was conducted afterward without difficulty. After anesthesia induction, two central venous catheters were inserted into the left and right femoral veins guided by ultrasound, and the general anesthesia was maintained with sevoflurane inhalation, continuous infusion of propofol, remifentanil, and dexmedetomidine, and intermittent administration of sufentanil and cisatracurium.
The rectal temperatures were monitored throughout the surgery. Intraoperative blood salvage was used to collect autologous blood from the surgical site. At 10:45 am, the surgery began, and the procedure of tumor removal lasted 4 h 35 min.
Fluid therapy was guided based on hemodynamic parameters obtained from NICOM. The baseline parameters included a cardiac index (CI) of 2.3 L/(min·m2), total peripheral resistance index of 3258 dynes/(s·m2), stroke volume index (SVI) of 16 L/(min·m2), thoracic fluid content (TFC) of 23.5 kΩ−1, and stroke volume variation (SVV) of 15%. Preoperatively, a “passive leg raising (PLR) test” was performed, and the ΔSVI measured was 12%. Therefore, the basal volume of the patient was considered sufficient, and the hemodynamic parameters in this state were directly used as the baseline values for fluid management. Therefore, 20 mL/kg fluid in total was given during the operation as a basic fluid supplement, while the SVV and SVI were maintained under 20% float to optimize the intravascular volume. A volume expansion of 10 mL/kg crystalloid in 5 min was applied to the patient on the condition that the parameters fell below expectations. For instance, rapid bleeding happened at the third hour of tumor removal. HR rose to 135 beats/min, mean artery pressure (MAP) dropped to 44 mmHg, SVI dropped below 10 L/(min·m2), and SVV rose to 18, all of which indicated abnormal condition. Therefore, 60 mL suspended red blood cells were infused in 5 min. Furthermore, the volume status was evaluated every 5 min until the bleeding stopped and the parameters reached the expected range. Additionally, to evaluate the blood product or blood coagulation substances before transfusion, thromboelastographic (TEG) analysis, blood gas analysis, and coagulation tests were performed. During the operation, SVI was used to monitor her cardiac function and TFC was used to monitor the extravascular lung water content. Meanwhile, TEG analysis was used to guide the supplement of clotting factors or continuous administration of the tranexamic acid. Table 1 presents the detailed treatments applied. Figure 2 shows that the trends of hemodynamic parameters during the whole procedure was relatively stable. At the end of the operation, the MAP was significantly increased after the anesthesia was stopped, and the hemodynamic parameters indicated sufficient volume.
Detailed lists of treatments and vital signs monitored by NICOM.
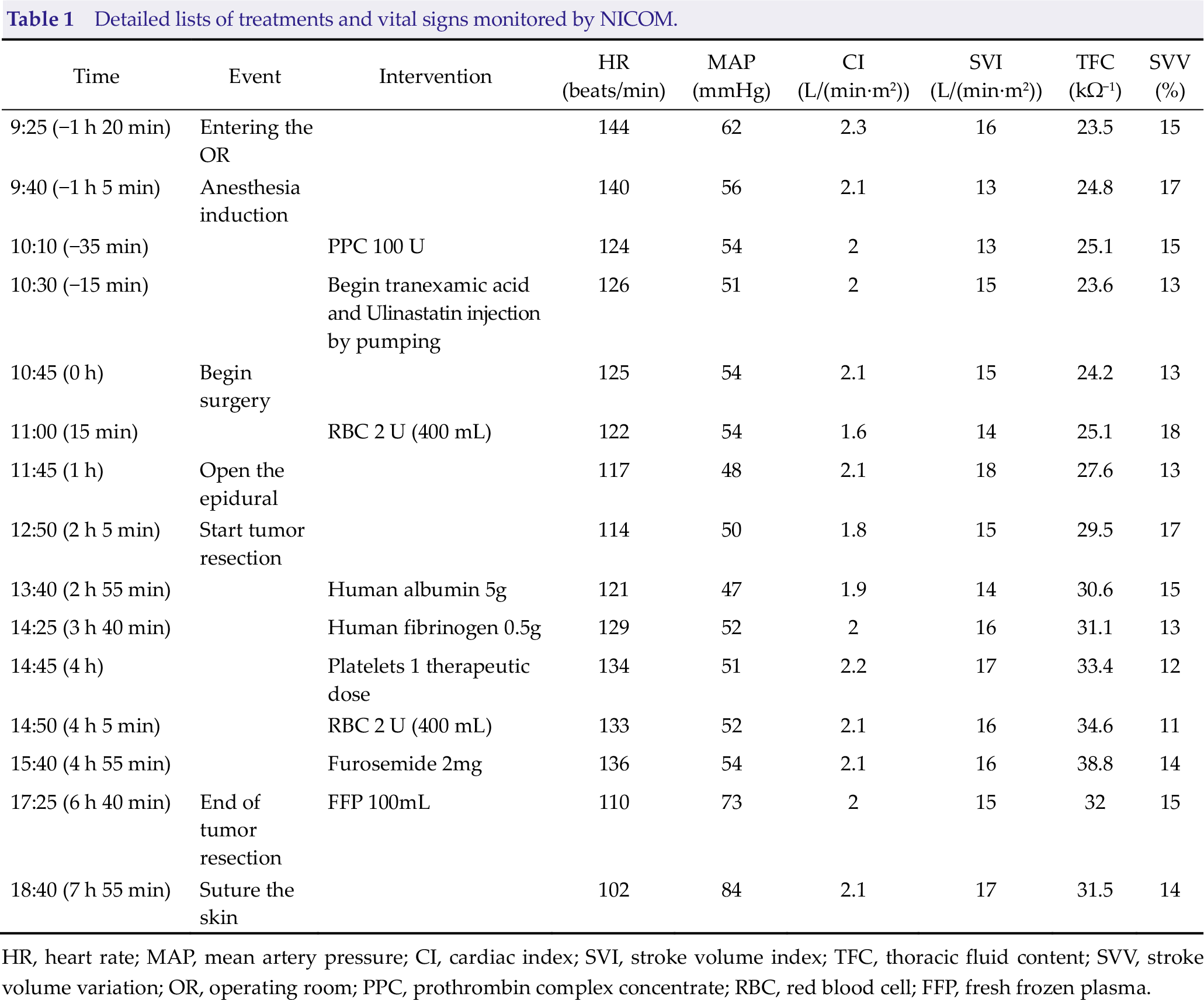
HR, heart rate; MAP, mean artery pressure; CI, cardiac index; SVI, stroke volume index; TFC, thoracic fluid content; SVV, stroke volume variation; OR, operating room; PPC, prothrombin complex concentrate; RBC, red blood cell; FFP, fresh frozen plasma.
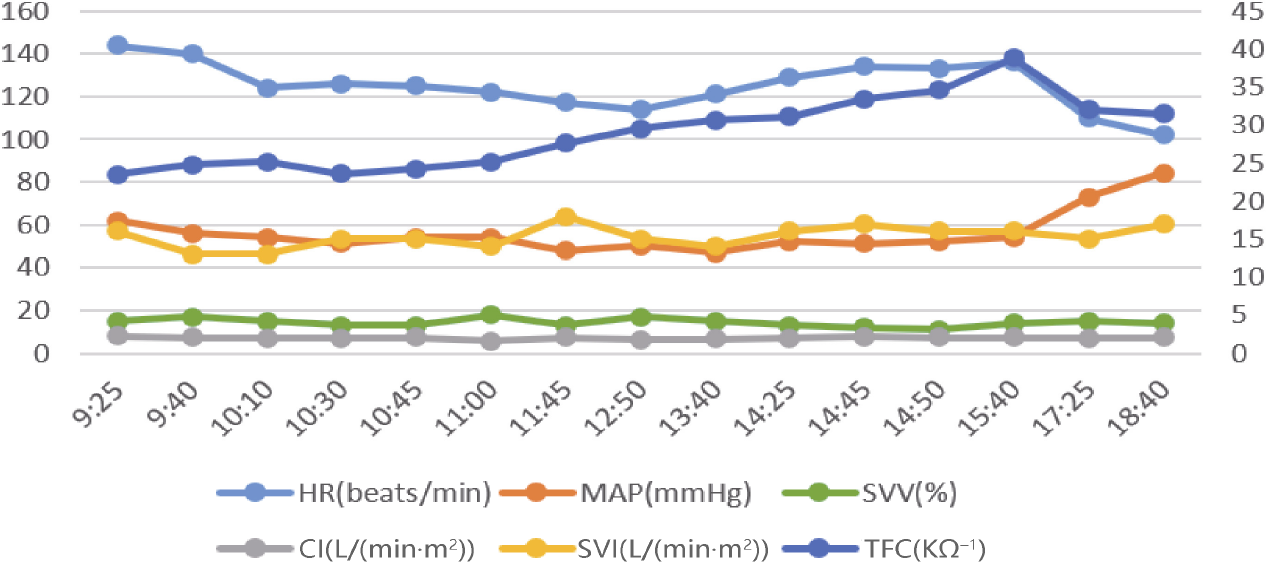
Trends of hemodynamic parameters monitored by NICOM during the surgery. HR, heart rate; MAP, mean artery pressure; SVV, stroke volume variation; CI, cardiac index; SVI, stroke volume index; TFC, thoracic fluid content.
During the 9.5-h procedure, 2600 mL fluid, which was approximately 5 times her blood volume, was infused. Meanwhile, about 700 mL of bleeding and 1200 mL of urine were recorded during the entire operation. The patient remained hemodynamically stable throughout the procedure due to the fluid treatment. The internal environment of the patient remained stable as well as presented in Table 2, suggested by the intermittent blood gas analysis during the operation. Furthermore, the postoperative blood and coagulation tests also indicated a stable internal environment as follows: hemoglobin level of 12 g/dL, platelet count of 361 μL−1, activeated partial thromboplasting time of 38.9 s, international normalized ratio of 1.11, and fibrinogen of 1.78 g/L.
Arterial blood gas analysis results.
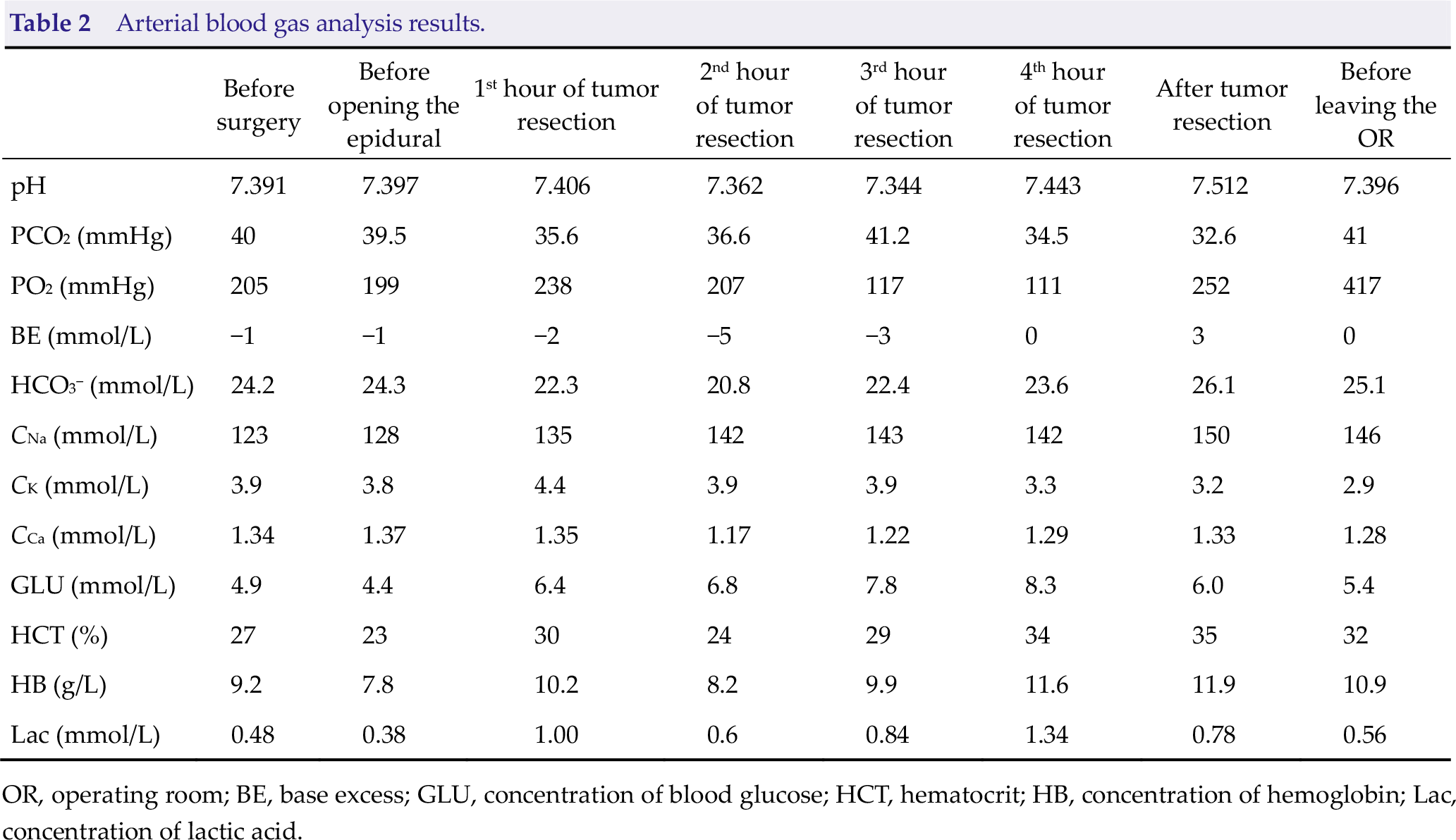
OR, operating room; BE, base excess; GLU, concentration of blood glucose; HCT, hematocrit; HB, concentration of hemoglobin; Lac, concentration of lactic acid.
The trachea was extubated 5 h after the surgery. The patient’s postoperative course was uneventful. The postoperative analysis, including electrocardiography, complete blood count, serum creatinine, liver function tests, and serum electrolyte, was at normal levels. After 6 months, no recurrence or extension of the tumor was observed in the computed tomography (Fig. 3). Further pathological analysis after the operation revealed that the tumor was a mature teratoma.
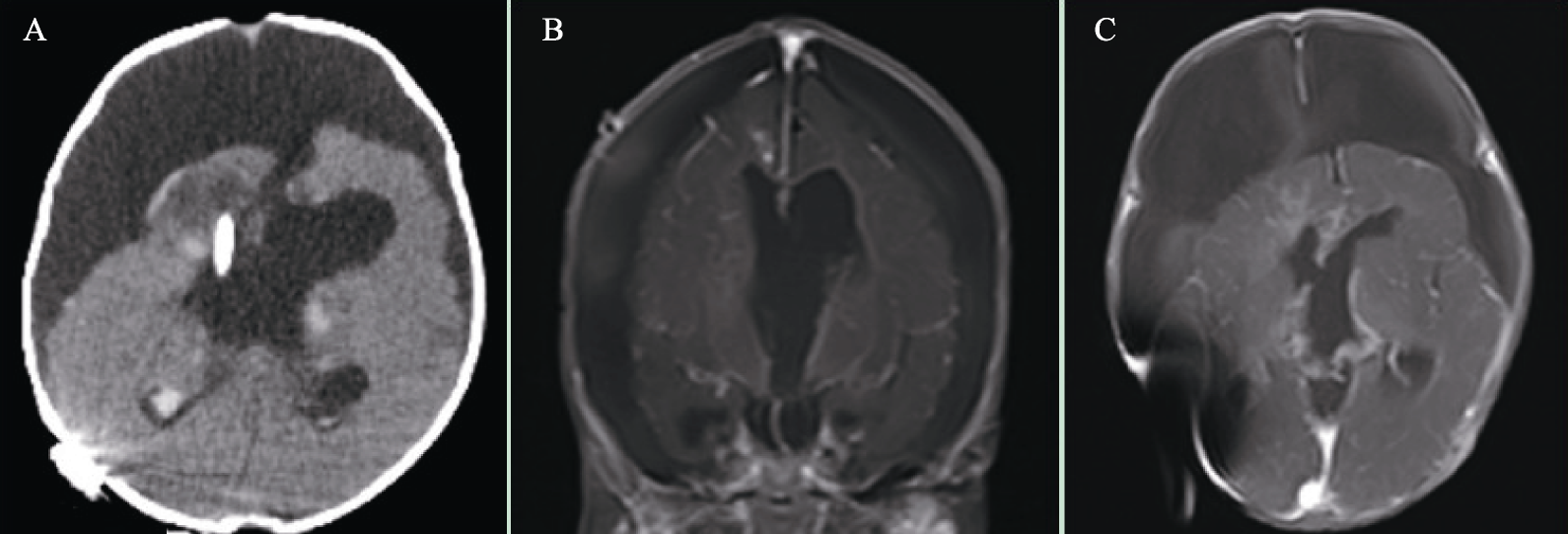
The CT image in the axial view (A), coronal plane (B) and sagittal plane (C) taken at six months after operation.
3 Discussion
The incidence of brain tumors in children is high, ranking first in the solid tumors of children, and about 50% of them are nonmalignant [10]. Brain tumors of children are usually large and more likely to arise in the posterior fossa [11], causing hydrocephalus and high intracranial pressure. Furthermore, emergency surgery is often required, and radical tumor resection is normally quite beneficial to the patients’ prognosis [12, 13]. However, infant patients with low body weight would confront a huge risk when undergoing giant intracranial tumors (> 5 cm in diameter) resection, due to the hypovolemia resulting from larger surface-weight ratio, higher total water content, limited renal ability to concentrate, and greater water loss from thin skin. Alternatively, conventional emergency fluid therapy (including blood transfusion) strategy of excessive supplement would cause other risks such as pulmonary edema, cerebral edema, and even cerebral hernia.
In adults, optimal perioperative fluid management with GDFT has been proven to be an important component of enhanced recovery after surgery pathways [14]. However, investigations demonstrated that static parameters such as HR, arterial BP, and CVP could not properly predict fluid responsiveness [4]. Multiple dynamic indices such as SVV, pulse pressure variation (PPV), and systolic pressure variation were used as the key indicators to predict whether an adult patient was fluid responsive. PPV or SVV over 13% was considered reliable to predict fluid responsiveness [15]. However, it is still unclear if it is also reliable for pediatric patients. Meanwhile, the volume responsiveness predictors could be affected by many factors in both adult and pediatric patients [16]. More importantly, many minimally invasive hemodynamic monitoring devices for adults, especially those calibrated ones, might not be suitable for children. Thus, anesthesiologists are looking for suitable hemodynamic monitoring equipment for children during surgery, and noninvasive equipment may be the ideal choice.
Transthoracic bioreactance is a new technique based on the analysis of the frequency variations of a delivered oscillating current traversing the thoracic cavity [17]. Different from the bio-impedance method, the bioreactance technique analyzes the change in the spectrum of the transmitted oscillating currents through the chest cavity [18]. Furthermore, the stroke volume (SV) is determined by measuring the phase shifts continuously. Therefore, this method has great potential against infection and can be used together with other types of monitoring equipment in the OR. Studies confirmed that the device using the bioreactance technique had an equivalent monitoring effect compared with thermodilution, pulse contour analysis, and ultrasonic detection [8, 19, 20]. This monitoring method is simple and easy to apply to children, avoiding the difficulty of puncturing the artery. The SVI and SVV based on bioreactance measurement had been found to effectively predict fluid responsiveness in children after craniosynostosis repair [22]. The SVV measured using the bioreactance method had also been found to commendably predict fluid responsiveness during mechanical ventilation of children after ventricular septal defect repair [23]. However, this device still has some limitations. Patients with pulmonary edema, pacemaker, aortic stenosis, and so on may get inaccurate parameters when using this device. Li Huang used NICOM, suprasternal USCOM, and esophageal CardioQ to monitor CO, and found that CO based on NICOM is inconsistent with those based on suprasternal USCOM and esophageal CardioQ during the upper abdominal laparotomy (using a surgical retractor) and laparoscopic surgery [21].
Currently, in the literature, there is few reported clinical research on the application of this noninvasive technique for the resection of giant brain tumors in pediatric patients, especially for infant patients with low weight and appropriate SVI. The parameters of children should be quite different from adults due to the physiologic and anatomic characteristics of the pediatric population. Therefore, adult hemodynamic management goals could not appropriately guide children’s fluid therapy. Luckily, a research showed that the ΔCI and ΔSV induced by PLR could be used to predict fluid responsiveness in children (95% confidence interval lower limits are 0.55 and 0.59, respectively) [24]. Thus, relative values were used in this study to guide fluid management instead of referring to absolute values. The baseline value was used as a reference and a 20% float was accepted. When SVV or SVI fluctuated for more than 20%, rapid infusion was given based on the results of the TEG and coagulation test. In this case, the operation was completed without using inotropes or vasopressors because of the chief surgeon’s precise and gentle operation and the baby’s health condition. The hemodynamic parameters monitored by NICOM were used to dynamically guide the fluid treatment of this child, so she got sufficient fluid while not encountering the complications of tissue edema caused by excessive fluid. Finally, she successfully went through the tough situation.
4 Conclusion
Infants with low body weight usually possessed low blood volume and hence high risk of hemorrhage during brain surgery. Therefore, intraoperative anesthesia monitoring of the changes of blood volume could be very important but usually difficult. In this study, a successful case using noninvasive evaluation and applying prompt fluid adjustment was reported, which could elicit the potential of bioreactance-guided fluid therapy in making anesthesia and surgery safe for future pediatric patients.
Footnotes
Ethical approval
This work is approved by the Ethics Committee of Tsinghua University Yuquan Hospital. The guardians of the infant agreed to publish her medical records anonymously.
Consent
The guardians of the infant were informed and have signed a written consent.
Conflict of interests
All contributing authors report no conflict of interests in this work.
