Abstract
Epileptic spasm (ES) is one of the most common types of seizures in children. It is primarily characterized by brief axial contractions lasting less than 2 s and recurring in short clusters. It usually occurs in children of 3 to 12 months of age, although it can also occur after the age of 1 year. In general, children with ES develop other symptoms of epilepsy, such as tonic, tonic‐clonic, or focal seizures, after 3 to 5 years of age. ES in children is often damaging and usually results in developmental regression. First‐line treatments for spasm seizures include adrenocorticotropic hormone (ACTH) and vigabatrin. However, many patients fail to respond to these medications, and continued to have spasms associated with progressive neurodevelopmental degeneration. Therefore, it is important to consider whether children with drug resistance meet surgical indications to consider surgical treatment in such conditions. In this study, we reviewed and summarized the importance of preoperative evaluation in order to provide surgical options for treatment of children with ES.
1 Introduction
For seizures that start from the focal area or hemisphere, surgical resection can achieve similar ideal curative effect as that accomplished in other focal intractable epilepsy. However, because of the uncertainty of semeiology, diffuse interictal epileptic discharges (IEDs), and widespread electroencephalography (EEG) onset, localization of the epileptogenic zone (EZ) is challenging to determine in children. A large proportion of children with seizures do not have a clear lesion on magnetic resonance imaging (MRI), and therefore, other imaging techniques such as fluorodeoxyglucose‐positron emission tomography (FDG‐PET) or magnetoencephalography (MEG) are required to identify epileptic foci. Nevertheless, many children still need subdural electrodes or stereoelectroencephalography (SEEG) to help identify the EZ. Compared to other focal epilepsy, epileptic spasm (ES) has been only recently treated with surgery. Infantile spasms and adult ESs are both considered generalized seizures and are rarely recommended for surgery. In 1979, Branch and Dyken described a 7‐month‐old baby with seizures who underwent resection of choroid plexus papilloma and subsequently became seizure free. A large number of subsequent reports have confirmed the efficacy of lesion resection for intractable ES [1, 2].
With the development of imaging and intracranial electrode technology, clinicians are trying various techniques to locate potential lesions in patients of seizures with negative MRI. In the 1990s, FDG‐PET was first reported for identifying EZ [ 1 ]. The minimally invasive technique of subdural electroencephalography also provided a surgical option for drugresistant patients with negative imaging results. In this article, we review the current status of the surgical treatment of ES to further demonstrate the feasibility of surgical resection in the treatment of ES.
2 Selection of surgical patients
ES is considered to be resistant even when patients are treated with two appropriate antiepileptic drugs, including first‐line drugs such as vigabatrin or adrenocorticotropic hormone (ACTH). Nevertheless, epileptic surgery should be considered if a focal origin is suspected.
Whether ES is of focal origin is usually assessed by noninvasive methods such as MRI, PET, and EEG. Focal epilepsy is characterized by a focal lesion on imaging, focal discharges, focal seizure onset, focal hypometabolism of PET, and asymmetric symptomatology. Surgical outcomes may be highly correlated with these characteristics. In addition, surgical outcomes may be influenced by the etiology and surgical site. In our epilepsy center, we enrolled 64 pediatric patients with ES who had undergone surgery, and found that approximately 83% of the patients had good surgical outcomes, which was considerably higher than estimated. The success rate was also higher than the average success rate reported in recent studies at other centers [ 2 ]. Approximately 50% of the children with ES have identifiable lesions on MRI. A population‐based study in Finland found that 26 of 42 (62%) children with ES had visible lesions on imaging [ 3 ].
We did not find any differences between surgical outcomes and gender, age at first seizure, age at surgery, or duration of seizures. Previous studies reported that children with intractable ES who underwent surgery within 3 years after the first seizure tended to have better outcomes [ 4 ]. The reason for better outcomes at a younger age is unclear. A possible explanation is that ES is more active in young children. It is easier to establish a large epileptic network and form secondary epileptic foci in infants. Early surgical treatment can produce early remission and promote brain development. At the same time, in these patients, early resection can prevent neurological impairment because of early functional compensation. A study in the United States reported 65 cases of medically refractory spasm in children undergoing surgical treatment [ 5 ]. It was found that patients who underwent surgical treatment earlier, and whose imaging lesions were consistent with abnormal discharge of EEG tended to achieve better surgical results.
Even if no lesions are identified on imaging, PET with focal hypometabolism is an important prognostic indicator. Another study showed that lesions identified on MRI were highly correlated with epileptic foci after complete resection and surgical outcomes [ 1 ]. These studies provide an important theoretical basis for the selection of surgical patients.
3 Preoperative evaluation
All patients with ES with suspected focal onset should be evaluated for epilepsy surgery by a multidisciplinary epilepsy surgery team. They have to undergo a comprehensive preoperative examination including MRI, interictal and ictal video‐EEG, PET, and neuropsychological testing. If there is a suspected specific gene‐related cause, such as tuberous sclerosis complex (TSC), patients routinely undergo genetic testing. However, previous studies have reported that there is a broad spectrum of genes associated with ES without specificity, and currently most genetic findings have no immediate influence on patient management [ 6 ].
3.1 Video‐EEG monitoring
At least three habitual seizures should be captured during long‐term video‐EEG (VEEG) monitoring. An experienced EEG specialist will observe the IEDs and the pattern of seizure onset. Ictal onset models may be localized or diffuse (including voltage attenuation). The primary value of VEEG is its ability to identify ES. In addition, the asymmetry of bilateral jerking during ES can be detected by video monitoring, which is of great value for lateralization. Unlike other focal epileptic seizures, ES are usually transient and subtle, and thus IEDs are highly significant in preoperative evaluation (Fig. 1).
3.2 Multimodal imaging examination
Whether MRI lesions are consistent with IEDs can determine the surgical procedure. However, in patients with focal discharges but without imaging lesion, it cannot be concluded that there is no surgical option.
The importance of PET has been reported in some studies. When the EZ is not identified through noninvasive examination including MRI, PET, VEEG, and MEG; or when the boundary between EZ and the functional cortex is unclear, the patient may undergo additional invasive procedures such as subdural electroencephalography or SEEG [ 7 –9 ]. The placement of intracranial electrodes is usually guided by multimodal information such as PET and MEG findings. If MRI, EEG, and PET results are consistent, additional intracranial EEG monitoring is not required [10, 11]. It is evident from reports of a recent series of surgeries that FDG‐PET is often used for preoperative evaluation of ES. Indeed, some experienced groups consider FDG‐PET to be an essential component of the preoperative evaluation of children with ES (Fig. 2). A considerable number of studies have confirmed the role of FDG‐PET in the preoperative evaluation of patients with ES, especially in MRI‐negative patients. It can identify focal metabolic abnormalities and assist in localization. In a recent series of studies on the surgical treatment of ES, 12 of 14 (85%) MRI‐negative patients with ES showed unilateral or focal hypometabolism on PET imaging [ 12 ]. Previous studies have also indicated that if focal hypometabolism of PET is consistent with focal IEDs in MRI‐negative patients, then focal resection can control the seizures and improve the patient’s developmental level [ 13 ].
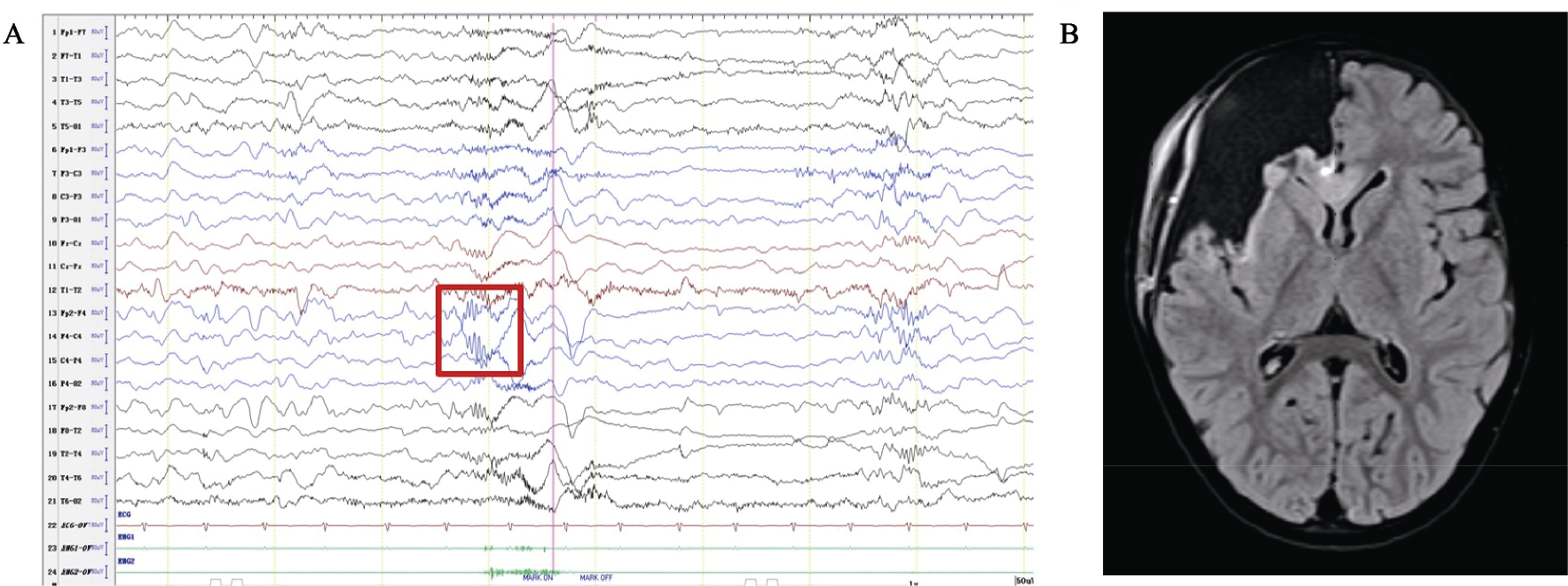
The VEEG and MRI of a 2‐year‐old female patient who had spasm for 7 months. (A) VEEG showed widespread slow wave discharge when seizure occurs. Rhythmic rapid activities appeared in right frontal lobe, which had localization value (red frame). (B)Preoperative MRI showed focal cortical dysplasia of right frontal lobe. The patient had no seizure 1 year after the right frontal lobe resection.
In addition, FDG‐PET can play an important role in identifying the EZ range and determining whether invasive electrodes are required to define the EZ. In patients with focal lesions, electrode implantation can be avoided if PET and MRI results are consistent. If the hypometabolism range of PET exceeds the imaging abnormality, it suggests that we may have underestimated the range of visible abnormality in MRI imaging, which would then require additional examination, including invasive electrodes, to accurately identify the EZ. PET can also predict whether patients can benefit from surgery. It often indicates a good postoperative prognosis for localized hypometabolism, whereas it may indicate a poor prognosis for generalized hypometabolism.
Magnetoencephalography (MEG) may also play a role in lateralization and localization in patients with focal lesions. In a recent report, 9 of 10 patients with ES showed focal IEDs on MEG imaging. Among 10 patients, 4 showed negative MRI results. In these negative cases, PET showed no focal hypometabolism, which further indicated that MEG could identify focal abnormality compared with MRI and PET [ 14 ].
3.3 Intracranial electrode monitoring
In MRI‐negative patients, although PET and MEG can provide useful data on localization, a significant proportion of patients require invasive monitoring to determine the EZ range. Neither PET nor MEG can replace intracranial electrode monitoring, in part because these imaging patterns generally reflect areas of abnormal IEDs, and the true EZ for ES is not well defined. The percentage of previous ES surgeries using invasive intracranial electrode monitoring ranged from 30% to 65%. SEEG was preferred in France and Italy, whereas subdural EEG was preferred by the North American epilepsy team. An Engel Epilepsy Surgery Outcome Scale class I score was achieved in 61% to 83% of patients who underwent both types of surgery. It should be noted that for infants with ES, a thin skull is a relative contraindication to SEEG implantation. Therefore, epilepsy centers that perform ES should also be familiar with subdural electroencephalography techniques, although SEEG is often used as a monitoring tool (Fig. 3).
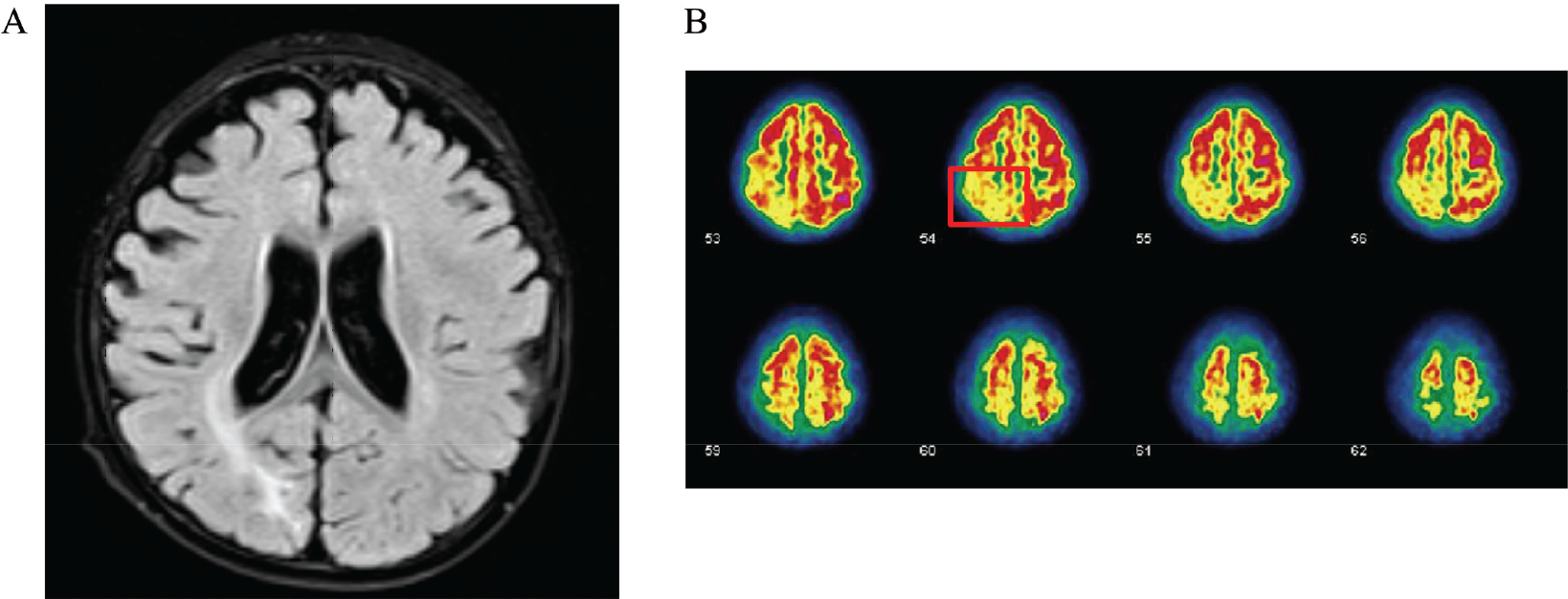
MRI and PET imaging of a 2‐year‐old male patient who had spasm for more than 1 year and was born with hypoxia. (A) Preoperative MRI demonstrated lesions in the right occipital lobe. (B) Preoperative PET showed low metabolism in the corresponding position (red square). The patient had no seizure for 3 years after right patieto-occipital disconnection.
Evidence shows that ES originated in the local cerebral cortex and rapidly spread to the premotor and motor cortex [15, 16]. The ES onset is usually characterized by an extensive slow wave outbreak, accompanied by low voltage rapid activity, which can be superimposed before or immediately after the slow wave and is often more focal. This model and the IEDs can be combined to define EZ. In addition, before fast activities, widespread slow waves are often accompanied by a focal “leading” discharge, which also conducts rapidly to the motor or premotor area within 100 ms. Asano and colleagues confirmed that if the cortex corresponding to these leading spikes were not removed during surgery, the surgical effects would be poor [ 17 ]. As the sampling rate of the EEG monitoring system increased, Nariai and colleagues found that both the leading discharge and the fast bursts during ES were in the high‐frequency oscillation range of 80 Hz to 200 Hz. Thus, the transmission of highfrequency oscillations to the Rolandic cortex is highly correlated with convulsions in the body.
4 Surgical strategies and postoperative outcomes
Abel et al. reported that surgical approaches to ES are varied and tailored to the patient’s EZ location [ 18 ]. Lesionectomy may be considered when noninvasive preoperative evaluation reveals clear lesion and no other metabolic diseases or specific gene‐related epilepsy that would preclude surgery. There is no standard model for surgery, and in focal cortical dysplasia (FCD), lesionectomy plus corticoectomy may be considered. With large lesions, anatomic lobectomy can be performed or even multiple lobectomies and hemisectomies. No known studies have shown an association between surgical site and surgical outcome. In our study, the most common surgical site was the frontal lobe, even after SEEG implantation. This is consistent with previous findings that the frontal lobe is prone to forming these complex clusters of axial movements. The exact mechanism should be determined through additional study of brain networks. For the two most common causes of ES, FCD, and TSC, we found that the consistency among MRI, PET, and scalp EEG results was an important factor in determining surgical strategy. Therefore, preoperative accurate interpretation of highresolution MRI, EEG, and fusion techniques provides an important basis for determining surgical approach. In addition, the SEEG application rate in patients with TSC abnormalities was much higher compared with patient with FCD abnormalities, suggesting that although the lesions in patients with TSC were clear on imaging, intracranial electrodes were often needed to help locate the true epileptogenic nodules due to the number of lesions [ 19 ].
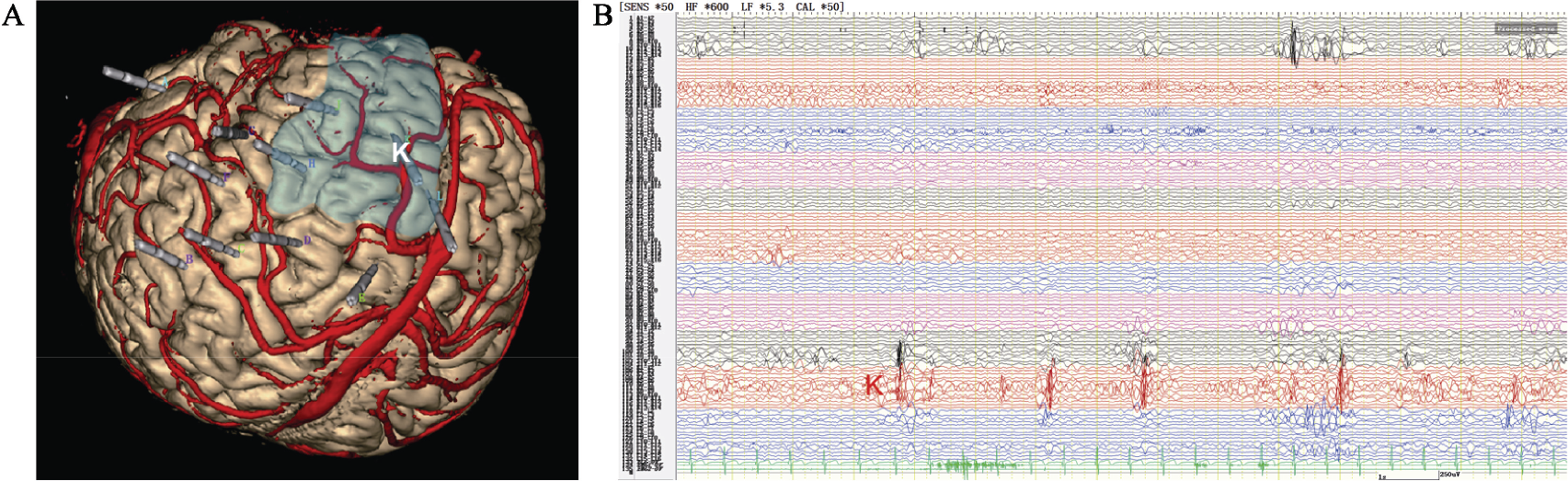
3‐year‐old male patient had drug resistant epileptic spasm for 21 months, he had language and mental retardation. Preoperative assessment could not help finding the epileptogenic zone, the patient was performed SEEG. (A) A 3D MRI image showing the actualplacement of all electrodes. The entry point of the electrode K was in the middle frontal gyrus, and the target was the anterior orbital frontal gyri. (B) SEEG showed that the onset of seizure began in the middle frontal gyrus (K electrode).
For patients with hemispheric electrical or imaging abnormalities, hemispherectomy or hemispherotomy should be considered early. Patients with ES secondary to metabolic disorders, chromosomal abnormalities, diffuse and bilateral brain injury, or diffuse and bilateral brain malformations are unlikely candidates for resective epilepsy surgery [ 19 ]. In patients with functional preservation of this area, the advantages and disadvantages of surgery should be weighed. Invasive intracranial electroencephalography is important in limiting EZ range during this process. Furthermore, intraoperative electrocorticography (ECoG) also play an important role in determining the range of EZ and protecting functional areas. For example, when the scope of intraoperative discharge monitoring exceeds the scope of the lesion, resection of the peripheral cortex may also be considered. According to our data, the proportion of patients with sustained discharges during ECoG was relatively high in the patients with FCD abnormalities. Intraoperative ECoG can be an important indicator of surgical resection range for FCD. It is worth noting that despite the challenges of locating the EZ in patients with ES, the seizure‐free rate of ES after resection was similar to that in other focal epilepsy. Recent studies have shown that the Engel Scale and International League Against Epilepsy (ILAE) class I outcomes after surgery ranges from 61% to 83%. We also suggest that children with intractable ES who underwent surgery within 3 years of their first seizure tend to have better outcomes. The reason is unclear, although one possible explanation is that ES is more active in young children. Large epileptic networks and secondary epileptic foci are more likely to be established in infants than in young children. Surgical treatment should be performed as early as possible to provide early remission and promote brain development.
It has also been reported that complete resection of the identifiable lesions on MRI and the determination of EZ range are the most important factors in determining surgical prognosis [ 20 ]. From our study, agreement between MRI and IEDs and between PET and IEDs, interictal high gamma rhythm, and continuous discharges of ECoG are associated with favorable surgical outcomes. This finding was supported by multivariate regression analysis that confirmed the importance of combining imaging and EEG data in the preoperative assessment. In addition, interictal high gamma rhythm (30–70 Hz) was found to be correlated with favorable surgical outcomes (Fig. 4).
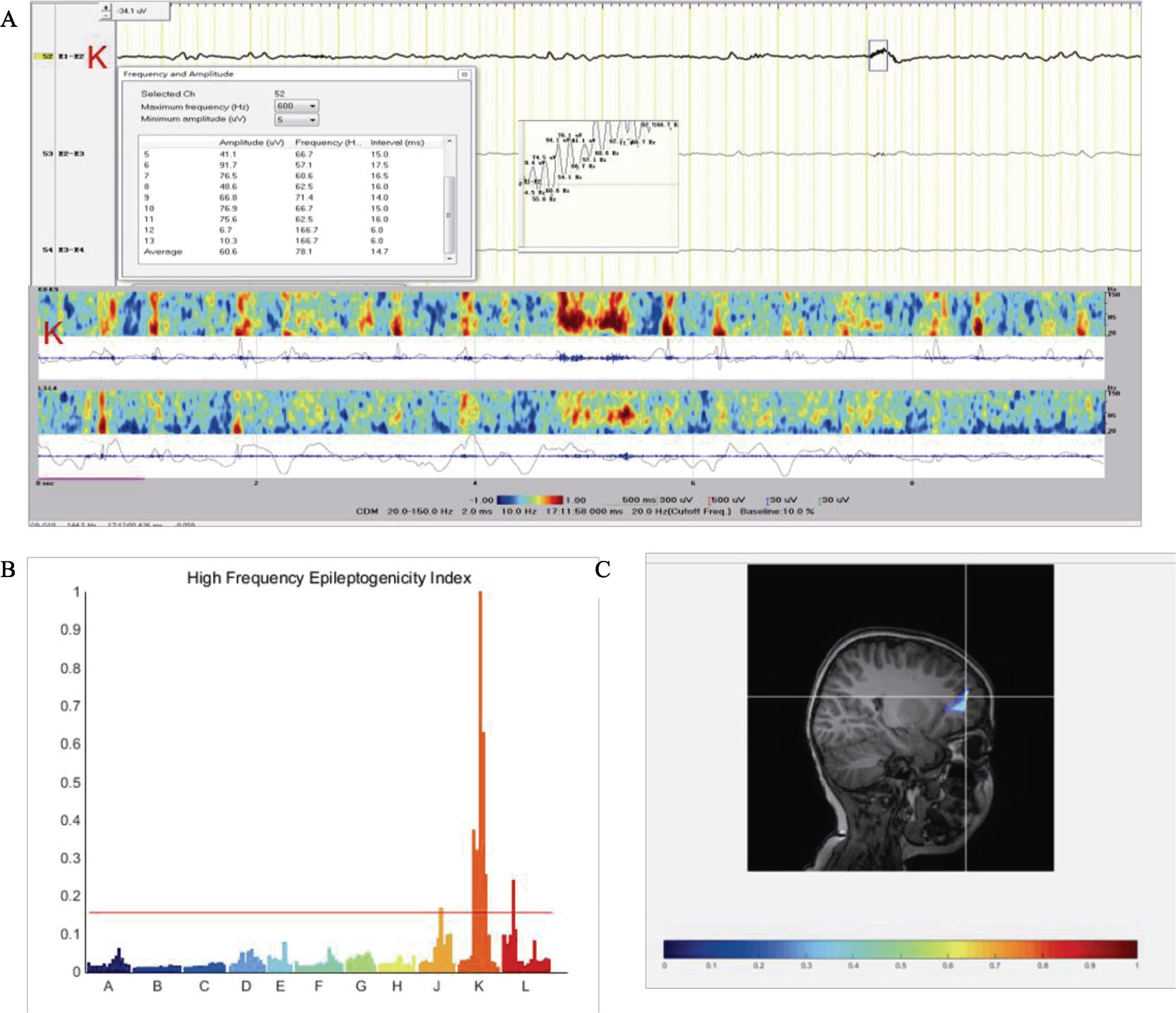
High frequency signal analysis. (A) Seizures started with low‐voltage fast activities in the middle frontal gyrus (electrode K). High frequency signal amplification found the frequency in the high gamma range (upper), SEEG power in the time–frequency plane (lower): seizure onset was characterized by early high‐frequency energy in middle frontal gyrus (electrode K). (B) High frequency quoted on the chart demonstrating maximal epileptogenicity in the electrode K. (C) Epileptogenicity index values overlay MRI image (color scale: from red to blue, epileptogenicity index value decreased), seizure onset was confined to middle frontal gyrus.
5 Conclusion
Surgical resection is effective in children with drug‐resistant ES if certain indications are met. Detailed medical history, comprehensive preoperative examination, and multidisciplinary preoperative evaluation are prerequisites that indicate whether surgery can be performed and to develop the surgical plan. A workflow for presurgical evaluation is also needed to simplify the process and to develop a reasonable surgical plan for the surgical treatment of children with ES (Fig. 5).
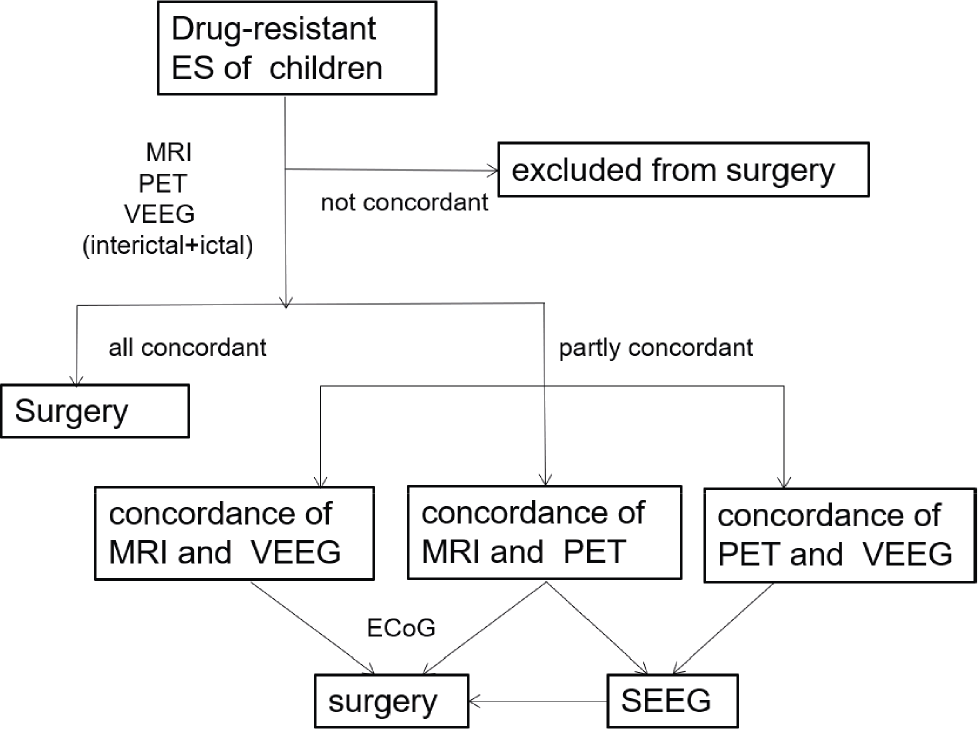
A proposed workflow for preoperative evaluation in children with ES.
Footnotes
Conflict of interests
The authors declare that they have no conflict of interests.
