Abstract
Motor imagery brain–computer interfaces (MI‐BCIs) have great potential value in prosthetics control, neurorehabilitation, and gaming; however, currently, most such systems only operate in controlled laboratory environments. One of the most important obstacles is the MI‐BCI inefficiency phenomenon. The accuracy of MI‐BCI control varies significantly (from chance level to 100% accuracy) across subjects due to the not easily induced and unstable MI‐related EEG features. An MI‐BCI inefficient subject is defined as a subject who cannot achieve greater than 70% accuracy after sufficient training time, and multiple survey results indicate that inefficient subjects account for 10%–50% of the experimental population. The widespread use of MI‐BCI has been seriously limited due to these large percentages of inefficient subjects. In this review, we summarize recent findings of the cause of MI‐BCI inefficiency from resting‐state brain function, task‐related brain activity, brain structure, and psychological perspectives. These factors help understand the reasons for inter‐subject MI‐BCI control performance variability, and it can be concluded that the lower resting‐state sensorimotor rhythm (SMR) is the key factor in MI‐BCI inefficiency, which has been confirmed by multiple independent laboratories. We then propose to divide MI‐BCI inefficient subjects into three categories according to the resting‐state SMR and offline/online accuracy to apply more accurate approaches to solve the inefficiency problem. The potential solutions include developing transfer learning algorithms, new experimental paradigms, mindfulness meditation practice, novel training strategies, and identifying new motor imagery‐related EEG features. To date, few studies have focused on improving the control accuracy of MI‐BCI inefficient subjects; thus, we appeal to the BCI community to focus more on this research area. Only by reducing the percentage of inefficient subjects can we create the opportunity to expand the value and influence of MI‐BCI.
Keywords
1 Introduction
Brain–computer interfaces (BCIs) record brain activity signals containing human intent, decode specific brain activity patterns using a computer, and translate them into control commands for external devices. The first BCI was proposed by Vidal in 1973 [1], and BCI technology has developed rapidly over the past 20 years due to developments in computer hardware and brain activity signal acquisition equipment [2, 3]. The greatest advantage of BCI is that it can realize direct communication between the brain and the external world without using the brain’s normal output pathways, e.g., the peripheral nervous system and muscle tissue. Thus, it has important application value in communication, motor assistance, neural rehabilitation, and clinical diagnosis [4 –7].
Generally, BCI systems comprise four modules, i.e., the signal acquisition, feature extraction, pattern classification, and stimulation and feedback modules. The main function of the signal acquisition module is to record continuous brain activity signals. The signal reflecting the operator’s intention is typically hidden among a large number of spontaneous brain activity signals, and the main function of the feature extraction module is to extract feature signals related to the operator’s intent from the spontaneous brain activity signals. The pattern classification module discriminates the extracted feature to decode the operator’s intention. Most BCI systems require external stimuli to induce specific brain responses, e.g., visual and auditory stimuli [8]; thus, the stimulation and feedback module presents such stimuli to the operator and displays the decoding results in real time. In some cases, the feature extraction and pattern classification modules can be combined, e.g., deep learning methods [9, 10], by which the operator’s intent can be decoded directly from raw brain activity signals in a single step.
Electroencephalography (EEG) acquisition devices are most commonly used for the signal acquisition module because such devices are non‐invasive, portable, have high time resolution, and relatively low cost [11]. Currently, the following three types of EEG‐based BCIs have developed rapidly: motor imagery BCI (MI‐BCI), steady‐state visual evoked potential BCI (SSVEP‐BCI), and P300‐BCI. SSVEP‐BCI and P300‐BCI can output more commands compared to MI‐BCI; however, they require external stimuli. Thus, the most suitable application for them is target selection, e.g., speller and home appliances control [7, 12]. The greatest advantage of MI‐BCI is that it does not require external stimuli and only depends on the operator’s internal motor imagery tasks to complete the control. In addition, there is large overlap between the brain network involved in motor imagery and motor execution [13]; therefore, impaired motor function may be repaired by motor imagery training, and BCI‐actuated functional electrical stimulation has been proven capable of achieving significant and lasting motor recovery in chronic stroke patients [5].
When imagining limb movements, the power of the EEG μ rhythm (8–13 Hz) and β rhythm (18– 26 Hz) measured by the sensorimotor area of the brain will decrease, and the decreased power will recover after limb imagination. Such phenomena reflect the decrease or increase of synchronization of neuron groups in the corresponding brain area. The power decrease is referred to as event‐related desynchronization (ERD), and power increase is referred to as event‐related synchronization (ERS) [14]. In addition, the spatial distribution of ERD/ERS observed from EEG signals differs when imagining different parts of the body. For example, when imagining left‐hand movement, the ERD/ERS phenomenon of the right sensorimotor area is more significant. In contrast, when imagining right‐hand movement, the significant area is in the left hemisphere. MI‐BCIs primarily use this difference to decode motor imagery tasks. The changes in μ (8–13 Hz) and β (13–30 Hz) rhythm mainly occur in the sensorimotor cortex, and they are commonly referred to as sensorimotor rhythms (SMR). Therefore, MI‐BCI is also referred to as SMR‐BCI [15].
Motor imagery‐related EEG features are not induced by external stimuli; thus, the feature distribution between subjects is very different in the frequency band and spatial position. Therefore, feature parameters must be customized for each participant [16]. In addition, EEG signals are nonstationary physiological signals [17], which frequently causes covariate shift in motor imagery‐derived EEG feature distributions [18], and transfer learning techniques are required to compensate such inter‐session variability of feature distributions [19 –21]. Even if many feature extraction and pattern classification algorithms have been proposed to increase the accuracy and robustness of MI‐BCI control [19, 22], the average accuracy of MI‐BCI is still less than that of P300‐BCI and SSVEP‐BCI [23, 24]. One important reason for this difference is that inter‐subject variability is very serious in MI‐BCI systems.
The remainder of this review is organized as follows. Section 2 defines MI‐BCI inefficiency and the percentage of inefficient subjects in the population. Then, factors that influence MI‐BCI control performance are categorized into four types in Section 3. In Section 4, we discuss our proposal, i.e., dividing inefficient subjects into three categories, and summarize five potential solutions to improve MI‐BCI control performance for a specific category of inefficient subjects.
2 MI‐BCI inefficiency
MI‐BCI control performance varies significantly across subjects. MI‐BCI inefficiency refers to subjects exhibiting poor MI‐BCI control performance after being well trained [25]. Typically, accuracy is used to measure the control performance of a subject, and Kübler et al. suggested that the minimum accuracy required by the subjects is 70% to achieve successful control of the BCI system [26]; thus, MI‐BCI inefficient subjects could be defined as those who cannot achieve accuracy greater than 70% [27]. Note that such subjects have been widely referred to as “MI‐BCI illiterates”. However, this name was criticized by Thompson in 2019 [28] because it may blame poor control performance on the subject rather than the BCI system [29], which is inadequate for BCI researchers.
The primary question about this phenomenon is how many people are MI‐BCI inefficient. In 2003, Guger et al. recruited 99 subjects in an MI‐BCI experiment with two sessions of data recorded for each subject. The classification results demonstrated that 48.7% of the subjects obtained accuracy less than 70%, 32.1% demonstrated accuracy between 70% and 80%, and the remaining 19.2% achieved accuracy greater than 80% [30]. According to Blankertz et al., 30 of 80 subjects (37.5%) could not achieve MI‐BCI control accuracy greater than 70% [31]. Based on a study including 40 subjects, Zhang et al. found that 45% of subjects could not achieve accuracy greater than 70% when performing motor imagery tasks for the first time [32]. A large survey involving 151 participants was conducted by Acqualagna et al. in 2016, where the recruited subjects were from two different universities and naïve to BCI. The online feedback experiment demonstrated that 40.4% of subjects reached average accuracy that was less than 70% [33]. The dataset released by Lee et al. in 2019 showed that 53.7% of 45 subjects are BCI inefficiency [24]. These studies highlight that MI‐BCI inefficiency should not be neglected.
To date, we have not found any survey about the percentage of MI‐BCI inefficiency in the motor function impaired patient group. Previous studies have shown that such patients have distinctive activation patterns and reduced ERD during motor imagery compared to healthy controls [34, 35], and the average accuracy of MI‐BCI is reduced in this patient group [35, 36]. Based on these results, we infer that the percentage of MI‐BCI inefficiency in this patient group is even higher than that of the healthy group. Healthy people may use other modal BCI systems if the MI‐BCI control performance is poor. However, for motor function impaired patients who require a BCI, they do not always have a choice. For example, MI‐BCI is the only choice when constructing a BCI‐based motor function rehabilitation system for stroke patients [37]. Currently, the MI‐BCI inefficiency is one of the factors that limit the widespread practical use of MI‐BCIs. It is the responsibility of the MI‐BCI researchers to explain the reasons for inefficient control and continuously refine MI‐BCI systems to allow more users to achieve efficient control.
According to Sannelli et al., MI‐BCI inefficiency could be further subdivided into two categories [29]. One category is assigned to subjects who achieve good offline training performance but poor online test performance, which has been found to account for 43% of MI‐BCI inefficient subjects [29]. The following factors might be the cause underlying the reduction in online control performance. First, MI‐related EEG feature distributions may change from offline to online sessions [18]. Second, online feedback may interfere with the motor imagery process. Third, the classifier model may be overfitted during training. The other category is assigned to subjects who achieve poor MI‐BCI control performance in both offline training and online feedback sessions, which is found to account for 57% of MI‐BCI inefficient subjects [29]. The main reason is that subjects with poor control performances in both sessions do not exhibit discriminative task‐related SMR changes during motor imagery.
3 Causes of MI‐BCI inefficiency
In this section, we review recent findings about MI‐BCI inefficiency relative to resting‐state brain function, task‐related brain activity, brain structure, and psychological factors. Brief summaries of these factors are given in Tables 1, 2, 3, and 4, respectively.
Summary of the resting‐state brain function factors.
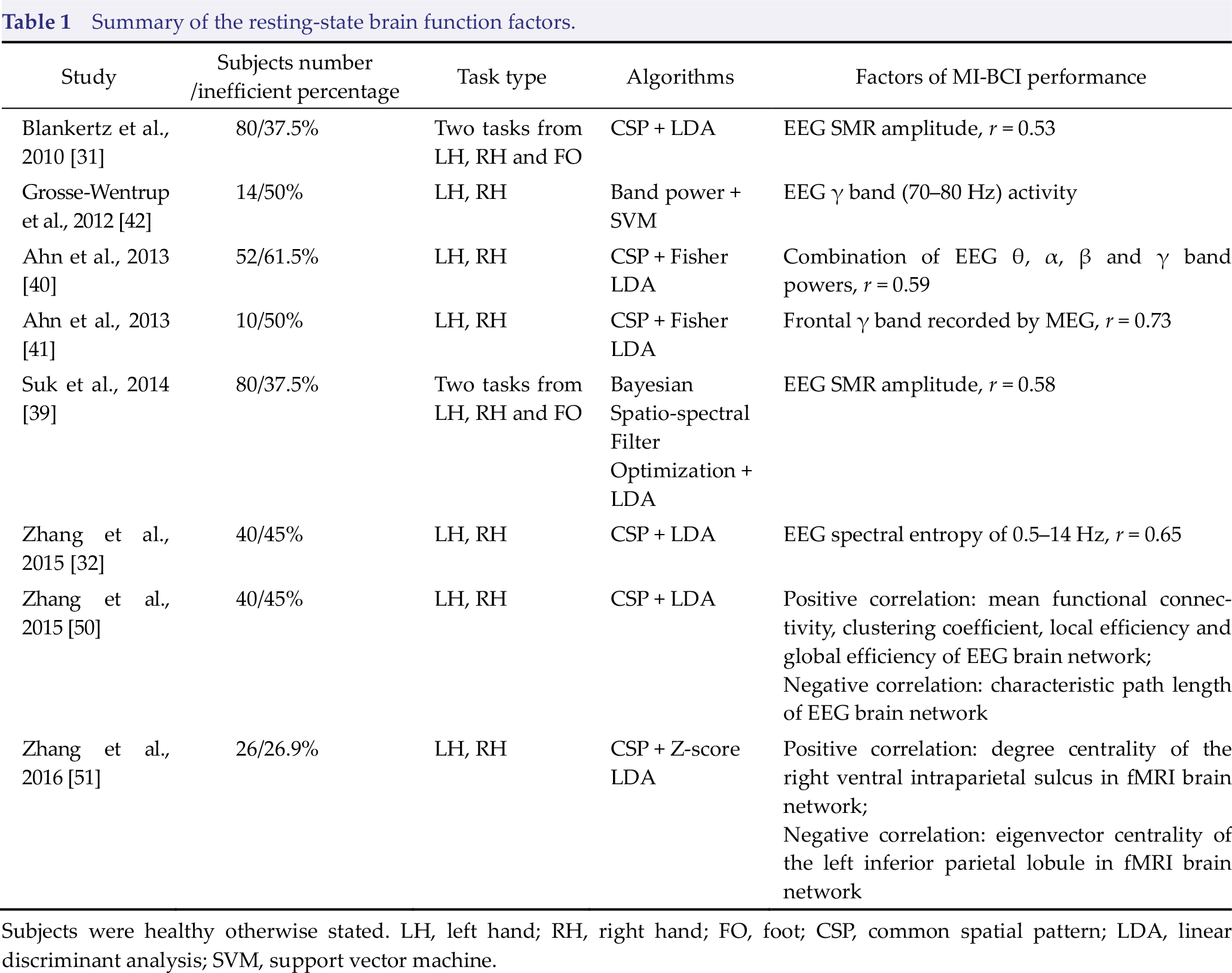
Subjects were healthy otherwise stated. LH, left hand; RH, right hand; FO, foot; CSP, common spatial pattern; LDA, linear discriminant analysis; SVM, support vector machine.
Summary of the motor imagery task‐related activity factors.
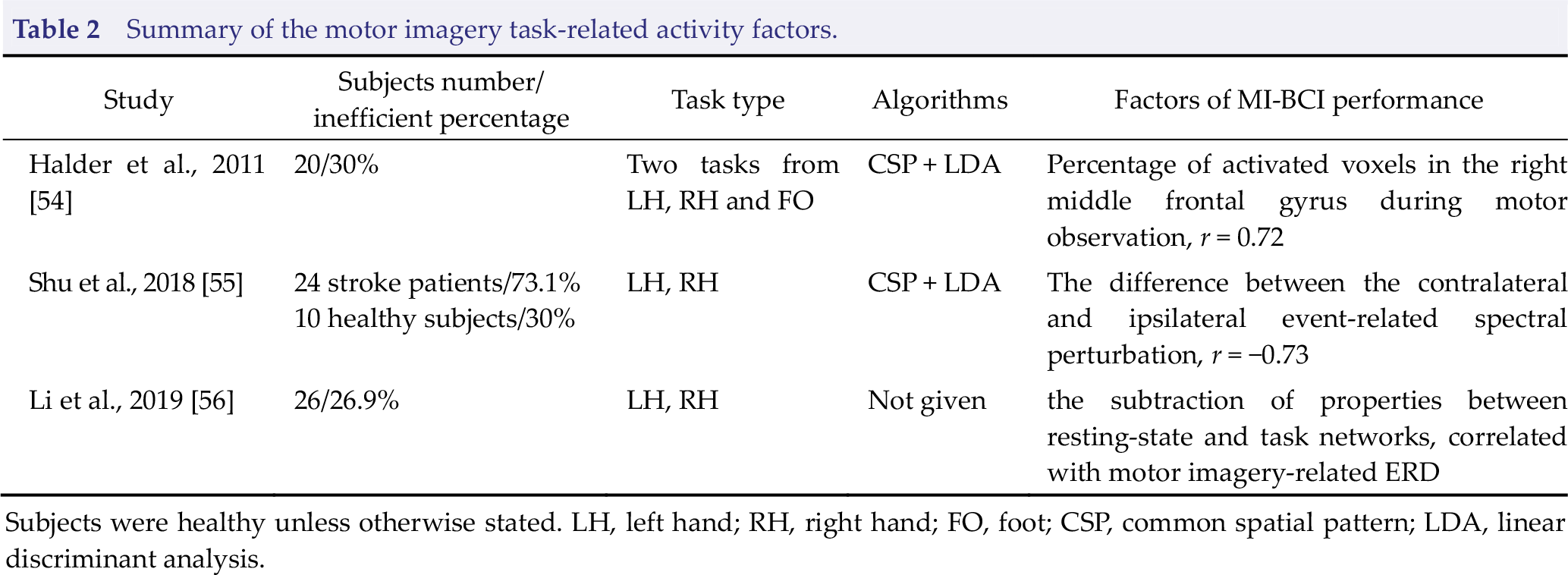
Subjects were healthy unless otherwise stated. LH, left hand; RH, right hand; FO, foot; CSP, common spatial pattern; LDA, linear discriminant analysis.
Summary of the brain structure factors.
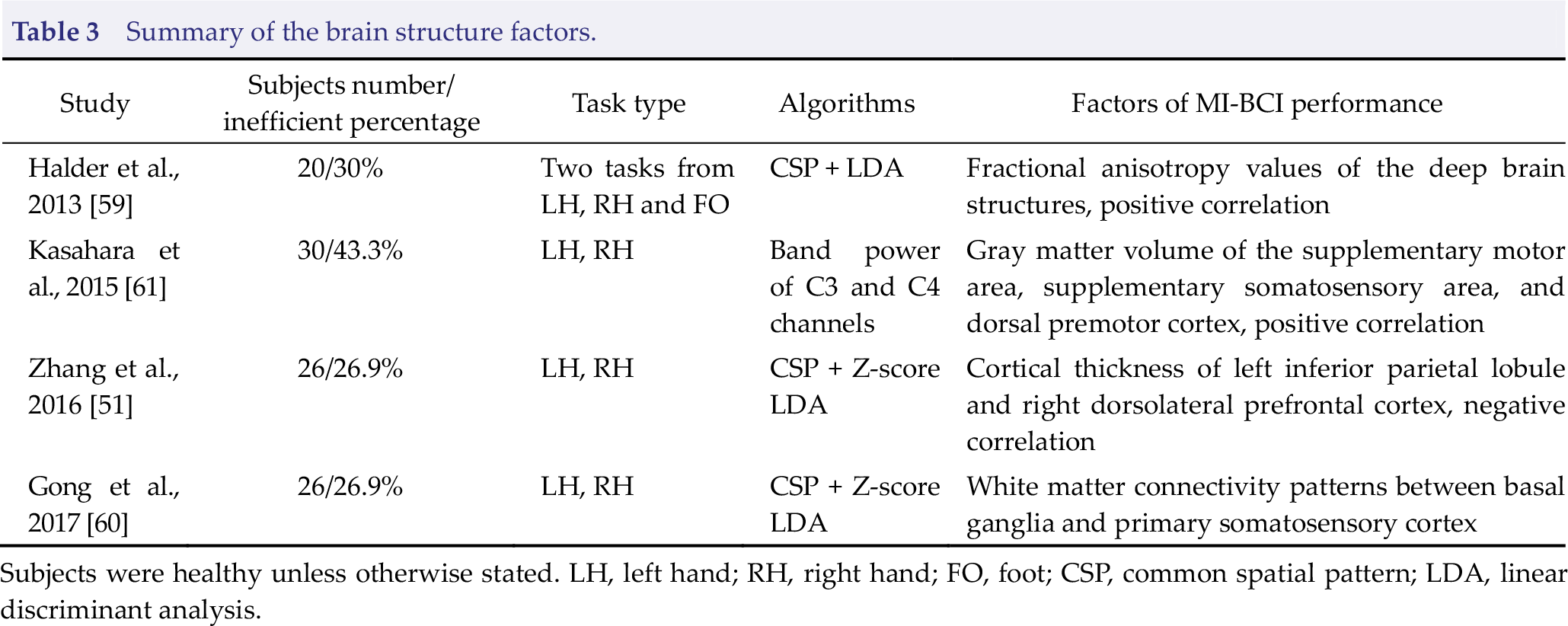
Subjects were healthy unless otherwise stated. LH, left hand; RH, right hand; FO, foot; CSP, common spatial pattern; LDA, linear discriminant analysis.
Summary of the psychological factors.
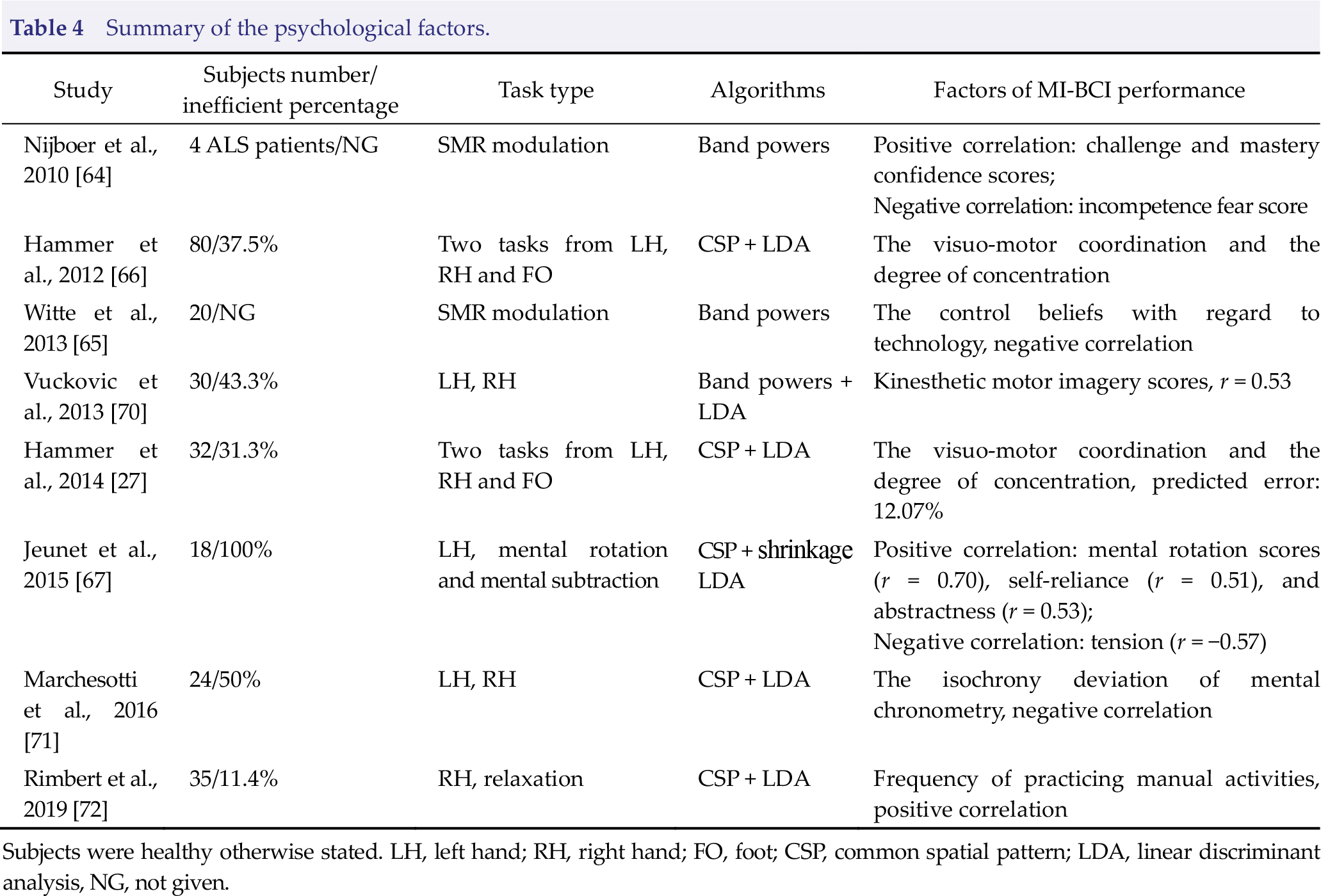
Subjects were healthy otherwise stated. LH, left hand; RH, right hand; FO, foot; CSP, common spatial pattern; LDA, linear discriminant analysis, NG, not given.
3.1 Resting‐state brain function factors
The human brain continues to function even during the resting state [38], and spontaneous neuronal activity in the resting state may affect task‐related brain signal changes. In 2010, Blankertz et al. first found that resting‐state EEG SMR amplitude is positively correlated with a subject’s MI‐BCI control accuracy. Then, a neurophysiological predictor derived from the “relax with eye open” condition recorded by two Laplacian EEG channels was calculated, and a correlation of
Motor imagery employs a large‐scale frontal‐parietal network and the sensorimotor region [13]. The human brain is a complex network comprising a large number of interconnected brain regions, and neural impulses are always transmitted and integrated across the spatially separated (but functionally connected) brain regions [44]. Many studies have revealed that the resting‐state brain network is closely related to human behavior and disease progression [45 –48]. Accordingly, the organizational structure of the resting‐state brain network may play an important role in SMR modulation. Wu et al. examined the relations between motor skill learning capacity and the brain network constructed by three‐minute resting‐state EEG, and the results demonstrated that stronger connectivity strength between the left primary motor area (M1) and left parietal cortex with weaker connectivity strength between the left M1 and left frontal–premotor areas predict greater motor skill acquisition [49]. Zhang et al. constructed resting‐state EEG network using the coherence and phase lag index, and the results of topology analysis demonstrated that MI classification accuracy is positively correlated with mean functional connectivity, node degrees, edge strengths, the clustering coefficient, local efficiency, and global efficiency of the rest‐state network, whereas the characteristic path length is negatively correlated with MI classification accuracy [50]. The positive correlation between the frontoparietal attention network and MI‐BCI performance was also confirmed by a resting-state functional magnetic resonance imaging (fMRI) study [51]. Thus, an inefficient background EEG network may be one cause for poor MI‐BCI control performance.
3.2 Motor imagery task‐related activity factors
The direct reason for poor control accuracy of MI‐BCI inefficient subjects is that the discriminative task‐related EEG features (primarily μ and β ERD in the sensorimotor area) cannot be produced by motor imagery. fMRI studies have revealed that contralateral somatosensory, contralateral premotor, bilateral supplementary motor area (SMA), contralateral thalamus, and ipsilateral cerebellum are activated during motor imagery [52, 53]. What is the brain activity difference between MI‐BCI efficient and inefficient subjects when performing motor imagery tasks? Halder et al. investigated the activations during motor imagery, motor observation, and motor execution tasks using fMRI. The group difference analysis demonstrated that MI‐BCI inefficient subjects exhibit significantly lower activation of the SMA for the motor imagery and motor observation tasks than MI‐BCI efficient subjects, and the number of activated voxels in the right middle frontal gyrus during motor observation task was significantly correlated (
performance in a dataset of 24 stroke patients and 10 healthy subjects [55]. Li et al. studied the brain network reconfiguration mechanism from the resting state to the motor imagery task state, and they found that the proposed reconfigured network index, which is defined as the subtraction of properties between resting‐state and task networks, is positively correlated with motor imagery‐related ERD [56]. In addition, the connections between motor‐related brain areas are significantly enhanced during motor imagery tasks, while the activity of the default mode network is suppressed. These findings suggest that MI‐BCI inefficient subjects may not be capable of switching the brain from resting to task states efficiently such that insufficient motor‐related brain regions are recruited during motor imagery task; thus, SMR suppression generated in the sensorimotor area is weaker.
3.3 Brain structure factors
Scalp EEG rhythms are associated with neuroanatomical characteristics. Specifically, white matter architecture and gray matter volume are correlated with the peak and power of the α band [57, 58]. The white matter network determines the conduction properties of the brain; thus, EEG rhythms are affected by these properties. The reduction of gray matter may reflect the reduction of neuropil and elimination of active synapses and thus cause EEG power reduction [58]. The EEG characteristics are closely related to the structure of the brain; therefore, it is reasonable to infer that MI‐BCI control performance is also related to white and gray matter properties. Halder et al. collected diffusion tensor imaging data including 16 subjects divided into MI‐BCI high and low aptitude groups. The results demonstrated strong correlations between MI‐BCI performance and the fractional anisotropy (FA) values of the deep brain structures involving the right cingulum, left superior fronto‐occipital fasciculus, body of corpus callosum, left cerebral peduncle, and right posterior corona radiate [59]. Local FA values reflect the myelinization quality, which is critical to the maintenance of appropriate conduction velocities for interregional communication in the brain; thus, this finding provides brain structural evidence to the inefficient resting‐state network transmission capacity of MI‐BCI inefficient subjects. The white matter connectivity pattern between the basal ganglia and primary somatosensory cortex was further found to significantly affect the ERD feature during motor imagery by Gong et al. [60], where an MI‐BCI inefficient subject showed more white matter connections between the sulci of primary somatosensory cortex and basal ganglia. For the efficient subject, more connections were located in the gyri of primary somatosensory cortex. Kasahara et al. revealed that the gray matter volume of motor‐related cortical areas, including the SMA, supplementary somatosensory area, and dorsal premotor cortex, are positively correlated with MI‐BCI performance [61], which indicated that increased gray matter in motor‐related regions facilitates distinct motor imagery‐related features recorded by scalp EEG. However, this conclusion may be unsuitable for other brain regions. For example, the cortical thickness of the left inferior parietal lobule and left dorsolateral prefrontal cortex demonstrated negative correlations with MI‐BCI performance [51].
3.4 Psychological factors
MI‐BCI control relies on efficient modulation of SMRs through motor imagery. It has been argued that learning to self‐regulate SMRs via motor imagery is similar to learning motor skills [62, 63]; thus, some psychological factors may play an important role in the learning process. Nijboer et al. investigated the correlation between mood and motivation in BCI paradigms and control performance with four ALS participants, which found that motivation rather mood is related to BCI performance [64]. They found that challenge and mastery confidence scores are positively related to SMRs regulation ability, whereas incompetence fear score is negatively related to SMRs regulation ability. However, Witte et al. studied the locus of control effect in 20 healthy subjects with 10 training sessions of SMR neurofeedback training, and they revealed that the control beliefs relative to technology are negatively correlated with the ability to increase SMR, and a possible explanation is that participants with high confidence in the control over technical equipment may consume more cognitive resources, and, in turn, this higher effort may interfere with the state of brain relaxation reflected in SMR [65]. Hammer et al. demonstrated that the psychological parameters they investigated played a moderate role in one‐session MI‐BCI control performance, where visuomotor coordination and the degree of concentration were significantly correlated with MI‐BCI accuracy [66]. Visuomotor coordination was measured by the overall mean error duration of two‐hand coordination tests that reflected the accuracy of fine motor skills and the exactness of information processing. Here, efficient MI‐BCI subjects demonstrated fewer mistakes and required less handling time. The degree of concentration indicates that the ability to concentrate on the task was measured by the performance level of attitudes towards work, and the corresponding result showed that this is important in MI‐BCI control. This is in line with the findings of Halder et al. because prefrontal areas are known to be important for allocation of attentional resources [54]. In 2014, Hammer et al. retested the prior MI‐BCI accuracy predictive model in a four‐session experiment (one calibration and three training sessions). The results indicated that the model explained approximately 20% of the variance of MI‐BCI performance, and the average prediction error was 12.07% [27]. Then, strong correlations between MI‐BCI performance and mental rotation scores, which reflect spatial abilities, were revealed by Jeunet et al. in 2015 [67].
Motor imagery could be divided into kinesthetic imagery and visual imagery [68, 69], where kinesthetic imagery recreates an experience as if the body was actually moving, while visual imagery experience is similar to watching a movie of themselves. Note that SMR modulation is more effective if the subject uses kinesthetic imagery. Vuckovic et al. tested kinesthetic and visual motor imagery questionnaire scores on the effect of MI‐BCI performance when subjects used two imagery strategies. Here, simple imagery and goal‐oriented imagery were selected to perform motor imagery tasks. The results demonstrated that kinesthetic imagery scores achieved higher correlations with motor imagery classification accuracy under both strategies compared to visual imagery scores, and the strongest correlation (
4 Solving the MI‐BCI inefficiency problem
According to the literature reviews on the influencing factors of MI‐BCI control performance, it can be concluded that resting‐state SMR is a key indicator to distinguish MI‐BCI inefficient and efficient subjects. Note that high resting‐state SMR does not a high MI‐BCI control accuracy because the achieved accuracy is also affected by psychological factors, e.g., mastery confidence, degree of concentration, and motor imagery strategies. Sannelli et al. proposed to divide MI‐BCI inefficient subjects into two categories according to their offline and online accuracies [29]; however, they did not consider the resting‐state SMR factor. To develop more accurate coping methods for the MI‐BCI inefficiency problem, we propose to further divide MI‐BCI inefficient subjects into the following three categories.
4.1 Transfer learning
Category I subjects obtained higher accuracy in the training set but lower accuracy in the test set, which indicates that they can modulate the SMR via motor imagery. However, the motor imagery‐derived feature distributions of the training and test data may differ significantly, although their conditional distributions remain unchanged [18, 73]. Thus, the classifier model calibrated based on the training data achieves worse prediction performance on the test data. Transfer learning technology from the machine learning field has been adopted to compensate for inter‐subject and inter‐session variability in motor imagery‐derived feature distributions [25, 74, 75]. When the distributions of the training and test sets change but their conditional distributions remain the same, transfer learning methods improve the classification accuracy of the test set by regularizing the model parameters estimated from the training set. Vidaurre et al. have demonstrated that one‐half of MI‐BCI inefficient subjects could perform MI‐BCI experiments with a good level of control within only 15 minutes [76]. Therefore, more efficient transfer learning algorithms for category I subjects may achieve remarkable results relative to reducing the percentage of inefficient subjects.
4.2 Mindfulness meditation
The inconsistent distributions of features between the training and test sets may be caused by unstable mental states due to anxiety, fatigue, frustration, or loss of concentration. Distraction caused by real‐time feedback may modify the EEG signal characteristics and introduce noise in a test session [77]. Mindfulness meditation involves non‐judgmental observation of sensations, thoughts, feelings, emotions, and environmental stimuli. It is a metacognitive process as it requires that both cognitive processes be controlled and stream of consciousness be monitored. Short term meditation training is known to improve attention and self‐regulation [78], and experienced meditators show more distinguishable EEG patterns than untrained subjects during motor imagery tasks [79]. Thus, mindfulness meditation training may help increase the degree of concentration and improve signal‐to‐noise ratios and thereby facilitate MI‐BCI control performance Previous studies have confirmed that MI‐BCI control accuracy could be improved by both long term (12 weeks) and short term (4 weeks) mindfulness meditation practice [80, 81]. Based on the above results, we can infer that MI‐BCI inefficient subjects, particularly category Ⅰand Ⅱ subjects, may benefit from mindfulness meditation practice.
4.3 New experimental paradigms
Generally, kinesthetic motor imagery induces stronger SMR modulation, making the EEG characteristics of the motor imagery of different limbs (e.g., left hand
4.4 Novel training strategy
Similar to other motor skills, generating stable and distinct brain activity patterns when performing different motor imagery tasks is an ability that can be learned [2]. Therefore, an appropriate training strategy is required to achieve MI‐BCI control. Meng et al. presented results that showed MI‐BCI control accuracy could be significantly increased even after only three MI‐BCI training sessions [88]. Simple motor imaging training, without real‐time EEG decoding, has also been found to help improve the control accuracy of poor BCI subjects [89]. Jeunet et al. investigated the training effects of simple motor tasks and demonstrated that there was no significant linear correlation between motor task performance and MI‐BCI control performance, whereas spatial abilities were found to be related to MI‐BCI control performance. Thus, they suggested using spatial ability training to improve MI‐BCI accuracy [90]. However, Botrel et al. revealed that visuomotor coordination and relaxation training does not increase MI‐BCI control accuracy, although the visuomotor coordination performance does improve [91, 92]. Different categories of subjects may require different types of training protocols. The inefficient MI‐BCI control performance of category Ⅰ and category Ⅱ subjects maybe partly due to unsuitable training protocols or the short training duration. Therefore, to expand the applicable MI‐BCI population, it is important to develop more effective training protocols and subdivide MI‐BCI inefficient subjects into appropriate categories.
4.5 New motor imagery‐related features
Because the resting‐state SMR of category Ⅲ subjects is low, it can be inferred that the SMR changes caused by motor imagery modulation may also be weak, resulting in insufficient discrimination of EEG signal characteristics for different tasks. In addition, most existing feature extraction algorithms are based on the motor imagery‐related ERD feature; therefore, it is reasonable that category Ⅲ subjects cannot achieve good MI‐BCI control performance. Motor imagery recruits a large fronto‐parietal network in addition to sensorimotor areas, and fMRI studies have revealed different brain connectivity patterns between left and right hand motor imagery [93, 94]. Such differences were also confirmed by EEG dynamic brain networks analysis [95]. Therefore, classifying motor imagery tasks using EEG network features is feasible [96, 97]. Zhang et al. showed that the topology features of a task‐related brain network achieved higher accuracy with 12 inefficient subjects compared to classical CSP features, and the average accuracy was improved by 7.9% when the network node degree and CSP feature combination were used. In that study, four out of the 12 subjects obtained accuracies higher than 70% [98].
Deep learning methods have been successfully used for MI‐BCI [9, 10]. Typically, deep learning methods do not require prior assumptions and involve fewer data preprocessing procedures. Therefore, deep learning methods can be applied to end‐to‐end training for motor imagery classification [99, 100]. Although it is difficult for inefficient MI‐BCI subject to produce discriminative ERD features, there may be other features that have not yet been discovered. Deep learning methods generate predictive models entirely based on training data and labels; therefore, deep learning has the potential to capture new features for MI‐BCI inefficient subjects. However, applying deep learning to BCI can be difficult. First, deep learning methods require a large number of samples to train and generate neural networks; however, with BCI tasks, the data sample size is often insufficient. In addition, when applied to EEG classification tasks, deep learning results are difficult to interpret.
The preferred solutions for different categories of MI‐BCI inefficient subjects are summarized in Table 5.
Suitable solutions for inefficient subjects by category.

5 Conclusion
Although MI‐BCIs have demonstrated some potential value in relation to prosthetics control, neurorehabilitation, and gaming, most systems still only operate in controlled laboratory environments due to their lack of reliability. In this paper, we have summarized the factors that have been found to affect MI‐BCI control performance from four perspectives, i.e., resting‐state brain function, task‐related brain activity, brain structure, and psychological factors. These factors can help us better understand the reasons for inter‐subject MI‐BCI control performance variability and can also help screen inefficient subjects in a short time, thereby avoiding long and frustrating training. To develop more accurate methods to alleviate the inefficiency problem, we propose dividing MI‐BCI inefficient subjects into three categories according to the resting‐state SMR and offline/online accuracy. Then, based on the three sub‐categories, we summarize five types of solutions to improve the accuracy of MI‐BCI inefficient subjects, thereby providing an innovative strategy to solve the inefficiency problem that hinders the practical application of MI‐BCI. Reducing the percentage of inefficient subjects will create opportunities to expand the value and influence of MI‐BCI, and this requires researchers in the BCI community to focus on developing effective algorithms, experimental paradigms, and training protocols.
Footnotes
Conflict of interests
The authors declare no conflict of interests in this work.
Acknowledgments
This research was supported by grants from the National Natural Science Foundation of China (NSFC; Grant No. 61603344, No. 61961160705, No. #U19A2082) and the Key Research Projects of Henan Higher Education Institutions (Project No. 16A120008).
