Abstract
Stereoelectroencephalography (SEEG) has been widely used in the presurgical evaluation of patients with medically intractable epilepsy. In the past, SEEG was commonly used as a method for mapping and localizing the epileptogenic zone (EZ). Since 2004, several studies have been conducted to examine the effectiveness of SEEG-guided radiofrequency thermocoagulation (RF-TC) in treating refractory epilepsy. However, the seizure-free and responder rates varied greatly across studies. We aimed to analyze the outcome of 56 patients who were treated with SEEG-guided RF-TC to evaluate the effectiveness of this treatment. SEEG-guided RF-TC can be considered as a treatment for refractory epilepsy. However, due to its limited efficacy, SEEG-guided RF-TC might be regarded as a temporary treatment performed under SEEG rather than a promising treatment for refractory epilepsy.
1 Introduction
In spite of the development of new antiepileptic drugs (AEDs), 20%–30% of patients with epilepsy remain refractory to AED treatment, which is called drug-resistant epilepsy [1]. Some of these patients whose epileptogenic focus is well identified may have an excellent seizure control after surgical resection [2]. If the epileptogenic zone (EZ) is confirmed as focal, it can be destroyed in a minimally invasive manner to treat the disease, which is similar to the goal of resective surgery [3]. Two ablative therapies have been used in treating epilepsy in recent years: stereotactic laser ablation (SLA) or laser interstitial thermal therapy, and stereotactic radiosurgery (SRS) [4, 5]. Review of data from retrospective studies suggests that these two methods may be beneficial in treating drug-resistant epilepsy with mesial temporal sclerosis, hypothalamic hamartoma, cavernous hemangioma, and small cortical dysplasias and malformations [5, 6]. The problem of both SLA and SRS is how to identify the location of EZ, especially in patients with contradictory noninvasive presurgical evaluation results. Stereoelectroencephalography (SEEG) has been widely used for more than 50 years in the presurgical evaluation of patients with medically intractable epilepsy. In the past, SEEG was commonly used as a method for mapping and localizing the EZ. Since 2004, several studies have examined the effectiveness of SEEG-guided radiofrequency thermocoagulation (RF-TC) in treating refractory epilepsy. SEEG-guided RF-TC has been considered as a safe and effective method for treating intractable epilepsy [7, 8]. However, the seizure-free and responder rates varied greatly across studies, they were 23% (95% CI, 8–50) and 58% (95% CI, 36–77), respectively, as reported by a recent meta-analysis [9].
Our study aimed to assess the effectiveness of SEEG-guided RF-TC in treating patients with refractory focal epilepsy.
2 Materials and methods
2.1 Patient selection
A total of 62 consecutive patients who underwent SEEG evaluation in the Epilepsy Center of Tsinghua University Yuquan Hospital between March 2016 and December 2017 were examined retrospectively. 56 patients agreed to be treated with SEEG-guided RF-TC after SEEG evaluation. All 56 patients (30 males and 26 females; aged 2–43 years, mean aged 16.5 ± 10.6 years) were diagnosed with refractory focal epilepsy. Multiple noninvasive presurgical evaluations were performed, including long-term scalp video-EEG monitoring, neuropsychological testing, threedimensional 3T magnetic resonance imaging (3D- 3T MRI), phase contrast angiography (3D PCA), and metabolic imaging 18F-fluorodeoxyglucose positron-emission tomography (F-FDG-PET). Implantation of SEEG electrodes was recommended because the data obtained from noninvasive investigations were not sufficient to localize the EZ. After implanting the SEEG electrodes, cortico-cortical evoked potentials and cortical stimulation were performed. After SEEG evaluation, 56 patients agreed to undergo SEEG-guided radiofrequency thermocoagulation, while the localization of the epileptic zone of each patient was defined using SEEG. This study was approved by the local ethical committee of Yuquan Hospital, Tsinghua University, China. All 56 patients were informed about the aims and risks of SEEG-guided RF-TC procedure, and signed a medical informed consent form before undergoing treatment.
2.2 SEEG implantation and recording
Before surgery, 3D-3T T1-weighted MRI and 3D PCA images were transferred to the stereotactic neuronavigation software (Neurotech, Advanced Systems Development Co., Ltd., China) in which 3D brain images and intracranial vessels were reconstructed. Trajectories were designed according to the prognosis of the location of EZ and the circumvention of the main intracranial vessels [Figs. 2(D)–(E)]. The SEEG implantation schemes were designed depending on the location of the suspected EZ. Using SEEG electrodes [Beijing HKHS Healthcare Co., Ltd., China; Fig. 1(B)] of various lengths and number of contacts (8–16 contacts per electrode; with each contact having a length of 2 mm and a diameter of 0.8 mm, and the distance between adjacent two contacts being 1.5 mm). When a wide area was suspected to be the EZ (e.g., insular cortex), the oblique orientation method was chosen for preparing RF-TC as it might enlarge the area of the lesions [Figs. 2(B) and (C)]. In the morning of the day of SEEG electrode implantation surgery, the patient received focal anesthesia, and the Leksell stereotactic frame was applied using a standard technique. After that, a stereotactic computed tomography (CT) scan was performed. The preoperative MRI, stereotactic CT, and PCA images were digitally processed using a dedicated fusion software (Neurotech, Advanced Systems Development Co., Ltd., China) to calculate the coordinates of each target and trajectory parameters; then, the neurosurgeons determined the actual implantation parameter. The fused 3D images were used during the implantation to confirm the accurate entry point and puncture angle, simultaneously avoiding the puncturing vessels of the scalp. The electrodes were implanted using the traditional stereotactic technique through 3-mm-diameter drill holes. Using the Leksell stereotactic system, the implantation was performed with skull perforation, dura opening, placement of the guiding bolt, trajectory formation using a probe, and final insertion of the electrodes (orthogonal or oblique orientation). CT scans were performed immediately after the surgery, and the reconstructed images were fused with the MRI data using the previously described fusion software to confirm the actual location of the electrodes.
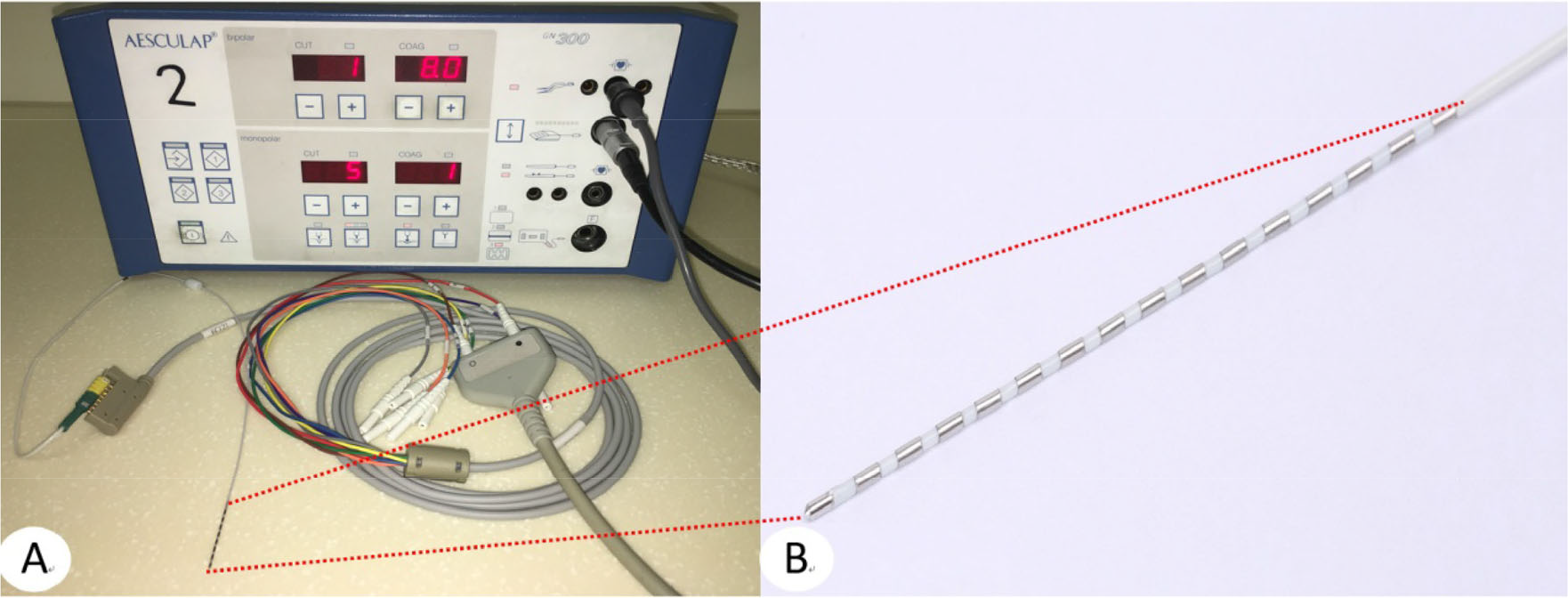
(A) Bipolar radiofrequency thermocoagulation (RF-TC) generator (GN300, Aesculap, Germany) and connecting lines between electrode and RF-TC generator. (B) SEEG electrode with 16 contacts. Each contact measured 2 mm in length and 0.8 mm in diameter, with a distance of 1.5 mm between two contacts.
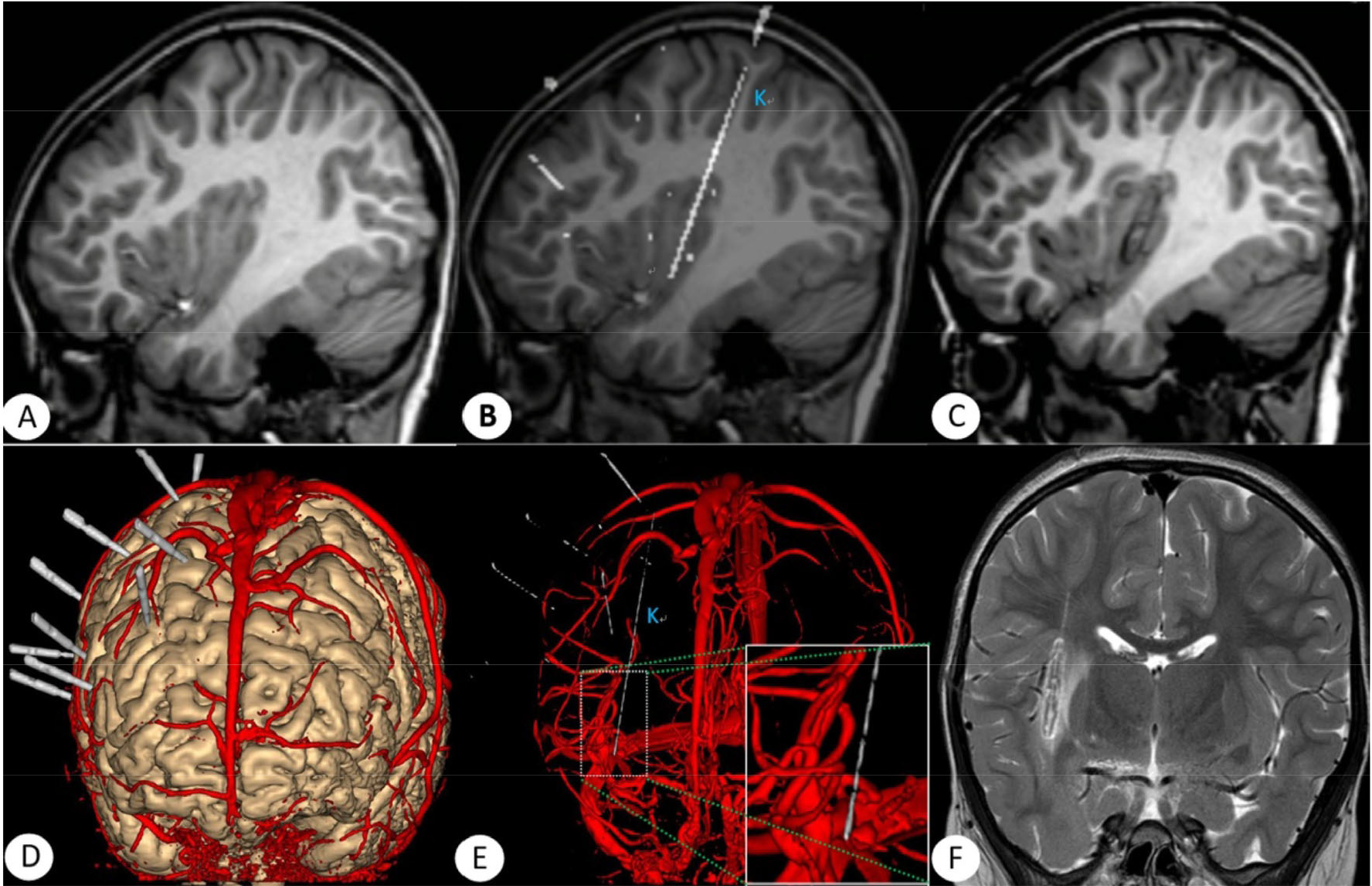
Images of patient 7. (A) MRI before SEEG electrodes implantation. (B) Fused images of postoperative CT and MRI of implanted electrodes. (C) MRI scans after RF-TC showing changes in brain tissue after coagulation in sagittal view. (D) Fused images of postoperative CT and MRI (T2 flair and PCA) using Neurotech software, three-dimensional images are shown. Electrodes were implanted as designed to avoid injury to the main intracranial vessels. (E) Fused images of postoperative CT and MRI (PCA) show that the minimal distance between electrodes (4–5 contacts on electrode K) and vessels nearby was about 2 mm. (F) MRI scans after RF-TC (coronal view along electrode K) show that the distance was safe.
Recordings were performed after surgery using SEEG electrodes (8–16 contacts). Signals were recorded on a 256-channel Nihon Kohden system or a 128-channel BioLogic system, and they were sampled at 2000 Hz or 256 Hz, respectively. Two hardware filters were present in the capture procedure: a high-pass filter (cutoff frequency, 0.16 Hz) and a low-pass filter (cutoff frequency, 100 Hz at 256 Hz, or 600 Hz at 2000 Hz).
2.3 Seizure-onset zone identification
The SEEG seizure-onset identification was performed independently by two epileptologists (Haixiang Wang and Bingqing Zhang) by reviewing the SEEG data to determine the seizure-onset zone (SOZ) and seizure-onset pattern. The seizure onset was defined as the first change in SEEG within a subsequent sustained rhythmic discharge and appearance of clinical signs using visual analysis. Seizure-onset patterns were classified according to previously published studies. Subclinical seizures were excluded from the analysis. The EZ site was classified as focal or multifocal according to the number of structure(s) involved in high-frequency oscillation (HFO) based on the perioperative image co-registration. Focal EZ was defined as a situation where only one structure had an HFO. Multifocal EZ was considered when at least two distinct structures disclosed HFO. The discrepancies were solved, and a consensus was reached after discussion with a senior epileptologist (Wenjing Zhou).
2.4 Thermocoagulation
Targets were considered eligible for RF-TC if some adjacent contacts were located in the cortical gray matter on postoperative CT-MRI registration and if SEEG showed evidence that those contacts were in the focal SOZ or adjacent to it. Each selected site for SEEG-guided RF-TC was initially tested using low and high-frequency bipolar electric stimulations between two adjacent contacts at increasing intensities up to a maximum of 8 mA. No important vessels were found around the lesion with a distance of less than 2 mm [Figs. 2(E) and (F)]. Ablation of the targets was performed immediately when the SOZ was confirmed by consultation because further recordings were required to observe SEEG changes after RF-TC (Fig. 3).
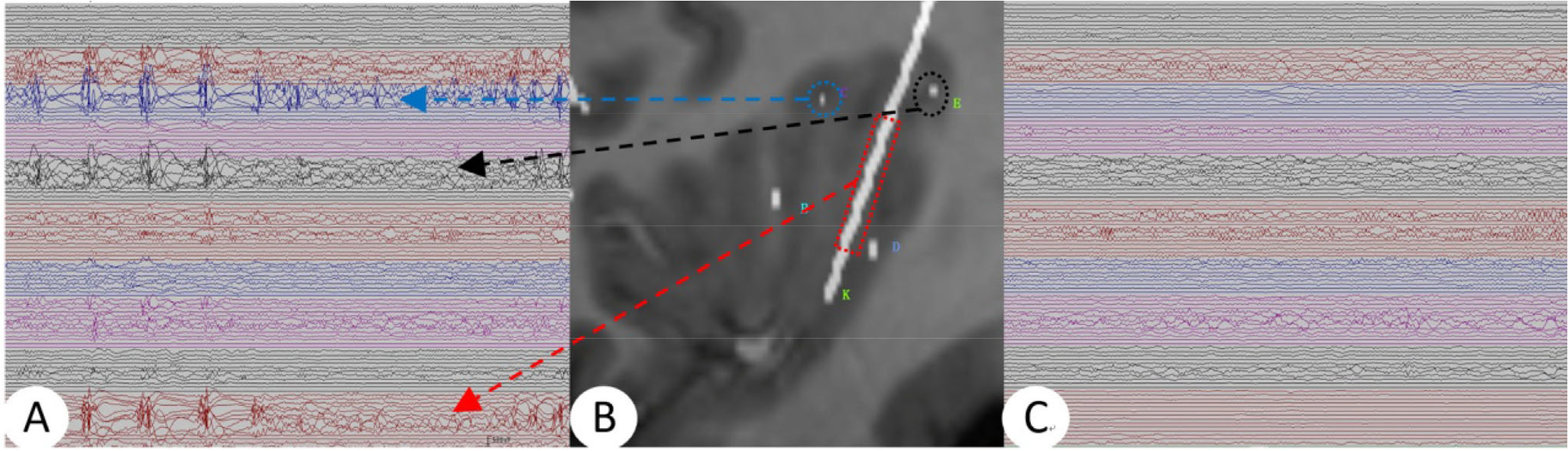
(A) SEEG of patient 7 before RF-TC. (B) Fused MRI and CT images after electrode implantation in the right insula lobe. (C) SEEG 2 days after RF-TC.
Lesions were produced between two adjacent contacts on an SEEG electrode using bipolar coagulation generator (GN 300, Aesculap; Fig. 1A), which could generate a 447-kHz radiofrequency current. The impedance between each pair of electrodes was measured before coagulation for the sake of safety. Moreover, a current of 8–10 W was used with a duration of 10–30 s, which could cause an increase in temperature up to 72–79 °C at the target (confirmed by
2.5 Seizure outcomes and statistical analysis
The outcome was assessed according to the Engel’s classification. For analysis, the outcome of the latest follow-up data (up to December 31, 2018) was used, in which 28 patients had a follow-up of more than 1 year after surgical resection. Patients with a seizure free or seizure reduction > 75% period beyond three times the longest pretreatment interseizure interval were considered to have a transient improvement after SEEG-guided RF-TC, although seizure reoccurred after that. After more than 1 year of follow-up after coagulation / surgical resection, patients with class I–II were classified as Group A (benefited from the coagulation / resection), while those who had class III–IV were classified as Group B (had no improvement). The Fisher’s exact test or Pearson chi-square test was used to compare the categorical data, while Student’s
3 Results
3.1 Number of SEEG electrodes, contacts, and coagulations produced
A total of 5–20 electrodes (12.8 ± 2.9) were implanted per patient (in the left hemisphere in 15 patients, in the right hemisphere in 20 patients, and both sides in 21 patients). Moreover, 3–47 lesions (14.7 ± 8.6) were produced after 1–3 times of coagulation. No significant difference was observed in the number of electrodes implanted and the number of coagulations produced between groups (12.3 ± 3.7 electrodes in Group A and 12.9 ± 2.7 electrodes in Group B,
3.2 Seizure outcome
After a follow-up of 12–32 months, 7 (12.5%) of 56 patients were seizure free and 2 (3.6%) of 56 had a decrease in seizure frequency of > 75%. Hence, 9 (16.1%) of 56 patients benefited from the treatment constantly, 47 are not. Furthermore, 28 (50%) of 56 patients had a transient reduction in seizure frequency or constant improvement after coagulation. Moreover, during 1–13 months after RF-TC, 42 of 47 patients who did not benefit from coagulation underwent surgical resection, and 1 underwent vagus nerve stimulation (VNS) surgery. In addition, 4 of 47 patients refused to undergo resection at the last follow-up. 1 patient who had a decrease in seizure frequency of about 80% underwent surgical resection 1 year after coagulation. Furthermore, 31 of 43 patients were seizure-free after surgical resection within 1-week to 31-month follow-up. Moreover, 19 of 28 patients who had a follow-up of more than 1 year after surgical resection were seizure-free. Patients who had a transient reduction of seizure frequency or constant improvement after SEEG-guided RF-TC had a higher probability of being seizure-free after surgical resection (
Data of 56 patients.
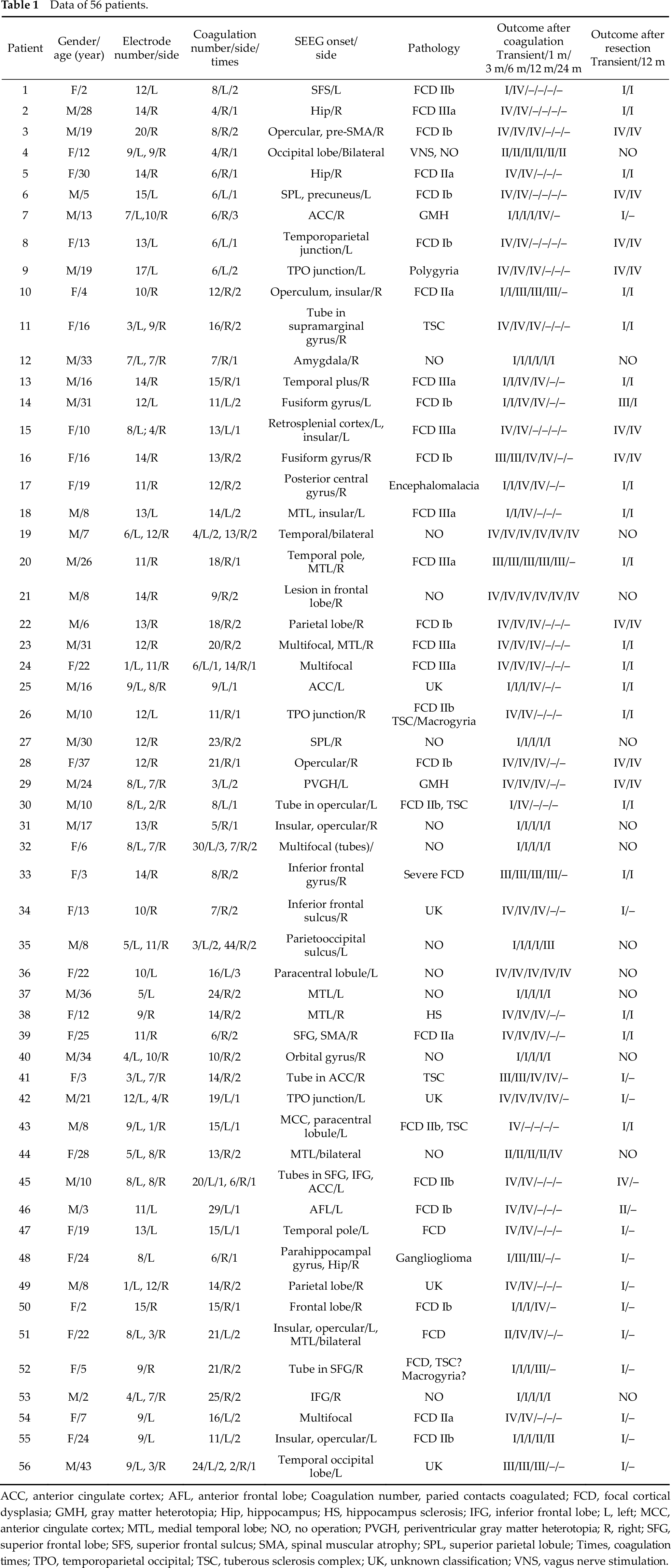
ACC, anterior cingulate cortex; AFL, anterior frontal lobe; Coagulation number, paried contacts coagulated; FCD, focal cortical dysplasia; GMH, gray matter heterotopia; Hip, hippocampus; HS, hippocampus sclerosis; IFG, inferior frontal lobe; L, left; MCC, anterior cingulate cortex; MTL, medial temporal lobe; NO, no operation; PVGH, periventricular gray matter heterotopia; R, right; SFG, superior frontal lobe; SFS, superior frontal sulcus; SMA, spinal muscular atrophy; SPL, superior parietal lobule; Times, coagulation times; TPO, temporoparietal occipital; TSC, tuberous sclerosis complex; UK, unknown classification; VNS, vagus nerve stimulation.
3.3 Side effects and morbidity after TC
Among 56 patients enrolled, 5 complained of transient headache, 9 complained of transient intracranial abnormal noise, and 4 had abnormal sensations, including nausea, sense of fear, and numbness of the limbs during the RF-TC procedure. However, these discomforts immediately disappeared when the treatment was discontinued. 3 patients had an aura, and usual attacks were induced in 4 patients during thermocoagulation. 2 patients (3.6%) suffered mild motor dysfunction at the last follow-up after coagulation. However, both patients were informed of the complications of motor dysfunction prompted by low and high-frequency bipolar electric stimulations before coagulation. None of the 56 patients complained of obvious cognitive impairments caused by coagulation.
4 Discussion
4.1 Efficacy and safety
In the past, a great deal of studies explored the use of stereotactic ablation in the treatment of epilepsy. However, these stereotactic ablation methods proved to have a less favorable outcome compared with standard ablation and hence were largely abandoned [3]. For patients with refractory focal epilepsy, surgical resection of the EZ showed more satisfactory outcome than other nonsurgical treatment [10, 11]. However, when discordance exists between electroclinical and presurgical imaging data, or when the SOZ is located close to functional areas, invasive presurgical evaluations are commonly required to determine the location of EZ and refine the boundaries of the surgical resection [12]. SEEG has been used to identify the exact location(s) of the epileptogenic area(s) and the pathways of discharge propagation for more than half a century. In the past, if the EZ was identified by SEEG, the electrodes were removed and surgical resection was subsequently performed. Nowadays, in addition to passive recording, SEEG electrodes are used to generate focal lesions of EZ identified by SEEG [7–9, 13 –27]. Patients may become seizure-free when the EZ is damaged according to the definition of Lüders [28].
Since the first publication of SEEG-guided RF-TC as a method for treating refractory epilepsy in 2004, this method was considered to be safe and effective. About 2.4%–80% of patients were seizure-free after undergoing SEEG-guided RF-TC. In the present study, 12.5% of the patients became seizure-free, and 16.1% benefited from this treatment during a follow-up of more than 1 year. Bourdillon et al. found that 67% of patients showed an improvement during a follow-up of 2 months, while 48% showed positive treatment response 12 months after RF-TC; after a follow-up of 10 years, only 13% of patients showed positive treatment response [8]. Although the outcome after SEEG-guided RF-TC is not quite satisfactory as that after surgical resection, and the rate of being seizure-free varies a lot among different studies, some patients do benefit from the treatment. When lesions overlap the EZ, patients may become seizure-free and avoid the need for surgical resection after the removal of electrodes.
On the contrary, if seizures are not under control, subsequent resective surgery may be considered. Bourdillon et al., based on a large sample study, reported that some patients who have benefited from SEEG-guided RF-TC may experience a recurrence in the future [8]. Therefore, the outcome should be monitored for a much longer follow-up period to evaluate the factors that may affect the outcome of SEEG-guided RF-TC.
The limited area of lesions produced by adjacent contacts in line of an SEEG electrode may lead to incomplete destruction of the EZ tissue limited to the width of the lesion produced, and that the short-term benefit may be due to the presence of edema or transient interruption of epileptic network, resulting in a decrease in satisfactory outcome in a long-term follow-up. In addition, presumed EZ identified by SEEG may be one of the independent EZs, and other foci may escape from implantation of SEEG electrodes. Given this, the prognosis of the location of EZ when an epileptologist makes the plan of implantation, as well as precise implantation of the electrodes during surgery may affect the result of SEEG-guided RF-TC, especially in patients who underwent controversial or negative presurgical evaluation. Even if the patients benefited from this treatment within a short term, it may indirectly verify the location of EZs. Bourdillon et al. showed that an improvement in the early months following a SEEG-guided RF-TC is predictive, with a high positive predictive value (93%), of a good outcome after conventional cortectomy in patients eligible for surgery [8]. In the present study, 10 of 10 (100%) patients who had a transient improvement after coagulation were seizure-free more than 1 year after resection, while 9 of 18 (50%) patients who did not benefit from coagulation were seizure-free 1 year after surgical resection. Therefore, a transient improvement after SEEG-guided RF-TC may be a positive predictor of the outcome after subsequent surgical resection. Patients with a transient improvement after RF-TC may have a greater chance of becoming seizure-free with subsequent resective surgery than those without transient improvement.
Considering the safety of the procedure, the distance between the center of planned coagulation site and vessels around it was examined in every patient before treatment. The diameter of lesions ranged from 5 to 7 mm. However, when the minimal distance between the vessels and the lesion was about 2 mm in patient 10, posttreatment MRI showed that it was still safe to perform RF-TC [Figs. 2(E) and (F)]. The coagulation generator used in the present study was GN 300, which was different from that used in other studies. Hence, it may be inappropriate to compare parameters among different studies and use parameters of the present study directly because different coagulation generator equipment may produce different coagulation sizes. In previous studies, most of the side effects were transient, and severe neurological or cognitive impairment was not frequently observed after SEEG-guided RF-TC, except when targets were close to the eloquent areas [7, 8, 22, 25]. In the present study, some transient discomforts were observed during treatment. Two patients (3.6%) had permanent neurologic impairments during the follow-up, while the rate of permanent neurologic impairment in previous studies was 1.2%–5.3%.
Compared with traditional methods of stereotactic ablation, SEEG-guided RF-TC had the following advantages: (1) the RF-TC could be performed directly between adjacent contacts on the recording electrode under the guidance of SEEG, especially in patients whose EZs were not clearly determined after undergoing a noninvasive presurgical evaluation; (2) the procedure did not require anesthesia; (3) changes in intracranial electroencephalogram (EEG) could be monitored after thermocoagulation, and another RF-TC could be performed if epileptic discharges were still observed at the EZ; in patients who refused to undergo surgical resection, electrodes might be added just for the purpose of coagulation; and (4) surgical resection could be performed in patients who were not seizure-free after SEEG-guided RF-TC because the lesions were located inside or near the resective area, and transient improvement after SEEG-guided RF-TC seemed to be a positive predictor of the outcome of resection.
4.2 Factors affecting the efficacy of SEEG-guided RF-TC
Some recent studies with a sample of more than 80 cases analyzed the factors affecting the outcome. They found that the factors with a positive impact were the etiology of nodular heterotopy, which showed significant differences compared with other etiologies, such as focal cortical dysplasia (FCD) [8, 9, 18, 24]. In our study, 2 patients with gray matter heterotopy did not become seizurefree after coagulation, which was inconsistent with the results of other studies. 1 patient diagnosed with tuberous sclerosis complex (TSC) became seizure-free more than 1.5 years after coagulation. Seizure outcomes were more favorable in patients with hippocampal sclerosis and those with a lesion detected on MRI, although these two factors had no significant impact on the efficacy [22]. Bourdillon et al. found that the MRI data, duration of epilepsy, and number of coagulations, which were thought to be the potential factors affecting the outcome of the treatment, were not considered important [8].
5 Conclusion
SEEG-guided RF-TC can be used for treating refractory epilepsy, especially in patients whose EZ has been identified as focal by SEEG in the presurgical evaluation; it does not add the risk of side effects. When patients become seizurefree after RF-TC, they may avoid the need for resective surgery; if not, it does not hinder the following surgical resection. However, due to its limited efficacy, SEEG-guided RF-TC might be regarded as a temporary treatment performed under SEEG recording rather than a promising treatment for refractory epilepsy. Patients with a transient positive response to the treatment have a greater chance of achieving a good outcome after undergoing a surgical resection.
Footnotes
Conflict of interests
The authors have no conflict of interests to declare.
Acknowledgments
The authors thank all participants, especially Professor Xun Wu and Professor Xiaoyan Liu for their help in our study.
